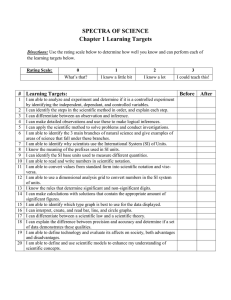
Significant figures

CHAPTER 2
Analyzing Data
National Standards for Chapter 2
– UCP.1 Systems, order, and organization
– UCP.2 Evidence, models, and explanation
– A.1 Abilities necessary to do scientific inquiry
– A.2 Understandings about scientific inquiry
– B.3 Chemical reactions
– B.6 Interactions of energy and matter
– E.2 Understandings about science and technology
– F.4 Environmental quality
– F.5 Natural and human-induced hazards
– G.2 Nature of scientific knowledge
– G.3 Historical perspectives
Vocabulary/Study Guide
• Define each term using the Glossary
• Either write on the handout, or use your own paper
• This is due on Test Day (tentatively, Thursday,
September 12)
Demonstration: Layers of Liquids
TITLE: LAYERS OF LIQUIDS
OBJECTIVE: To observe how liquids have different densities just like solids such as lead and plastic have different densities.
PREDICTIONS: (Do not write the questions)
1.
Which solids are less dense than water?
2.
Which solids are denser than water?
3.
Which liquids are less dense than water?
4.
Which liquids are denser than water?
DATA:
Lab: Determine Density, page 39
TITLE: DETERMINE DENSITY (Pg. 39)
OBJECTIVE: To calculate the density of an object by finding its mass and volume, using a scale and a graduated cylinder.
PREDICTIONS: (Do not write the questions)
1.
Why must you mass the object before immersing it in water?
2.
Some students have tiny pieces of magnetite (dull grey) and some students have large pieces of magnetite. Predict if they will have the same or different densities.
DATA:
Section 1: Units and Measurement
• National Standards:
– UCP.1 Systems, order, and organization
– UCP.3 Change, constancy, and measurement
– A.1 Abilities necessary to do scientific inquiry
– A.2 Understandings about scientific inquiry
– B.2 Structure and properties of matter
– G.1 Science as a human endeavor
– G.2 Nature of scientific knowledge
Objectives – Section 1
• Define SI base units for time, length, mass, and temperature.
•Explain how adding a prefix changes a unit.
•Compare the derived units for volume and density.
• REVIEW VOCABULARY:
– mass: a measurement that reflects the amount of matter an object contains
Units
• Chemists use an internationally recognized system of units to communicate their findings.
• Système Internationale d'Unités (SI) is an internationally agreed upon system of measurements.
• A base unit is a defined unit in a system of measurement that is based on an object or event in the physical world, and is independent of other units.
Base Units and SI Prefixes
Base Units and SI Prefixes
• Metric Chart
Base Units and SI Prefixes
Base Units and SI Prefixes
• The SI base unit of time is the second (s), based on the frequency of radiation given off by a cesium-133 atom.
• The SI base unit for length is the meter (m), the distance light travels in a vacuum in
1/299,792,458th of a second.
• The SI base unit of mass is the kilogram (kg), about 2.2 pounds
Base Units and SI Prefixes
• The SI base unit of temperature is the kelvin (K).
• Zero kelvin is the point where there is virtually no particle motion or kinetic energy, also known as absolute zero
• Two other temperature scales are Celsius and Fahrenheit.
Base Units and SI Prefixes
• Metric System Handouts
Derived Units
• Not all quantities can be measured with SI base units.
• A unit that is defined by a combination of base units is called a derived unit .
Derived Units
• Volume is a derived unit and is measured in cubic meters (m 3 ), but this is very large. A more convenient measure is the liter , or one cubic decimeter (dm 3 ).
Derived Units
• Density is a derived unit, g/cm 3 , the amount of mass per unit volume.
• The density equation is density = mass/volume
Density Problems
• Example Problem #1, Page 38
• Practice Problems #1-3, Page 38
• Density Handout with Chart
Homework, Section 1
• SECTION 1 REVIEW, Page 39
• Questions #4, 5, 7-10
• Answer with complete sentences
• Due tomorrow
Section 2: Scientific Notation and
Dimensional Analysis
• National Standards:
– UCP.1 Systems, order, and organization
– UCP.3 Change, constancy, and measurement
– A.1 Abilities necessary to do scientific inquiry
– A.2 Understandings about scientific inquiry
– E.2 Understandings about science and technology
Objectives – Section 2
• Express numbers in scientific notation.
• Convert between units using dimensional analysis.
• Review Vocabulary:
– quantitative data: numerical information describing how much, how little, how big, how tall, how fast, and so on
New Vocabulary
• scientific notation
• dimensional analysis
• conversion factor
• Scientists often express numbers in scientific notation and solve problems using dimensional analysis.
Scientific Notation
• Scientific notation can be used to express any number as a number between 1 and 10
(known as the coefficient) multiplied by 10 raised to a power (known as the exponent).
– Carbon atoms in the Hope Diamond = 4.6 x 10 23
– 4.6 is the coefficient and 23 is the exponent.
Scientific Notation
• Count the number of places the decimal point must be moved to give a coefficient between 1 and 10.
• The number of places moved equals the value of the exponent.
• The exponent is positive when the decimal moves to the left and negative when the decimal moves to the right.
800 = 8.0
10 2
0.0000343 = 3.43
10 –5
Scientific Notation
• Practice Problems, page 41
– #11-12
– Write the problem, then the answer
– Show your work, if necessary
Scientific Notation
• Addition and subtraction
– Exponents must be the same.
– Rewrite values to make exponents the same.
– Ex. 2.840 x 10 18 + 3.60 x 10 17 , you must rewrite one of these numbers so their exponents are the same.
Remember that moving the decimal to the right or left changes the exponent.
2.840 x 10 18 + 0.360 x 10 18
– Add or subtract coefficients.
– Ex. 2.840 x 10 18 + 0.360 x 10 17 = 3.2 x 10 18
Scientific Notation
• Multiplication and division
– To multiply, multiply the coefficients, then add the exponents.
Ex. (4.6 x 10 23 )(2 x 10 -23 ) = 9.2 x 10 0
– To divide, divide the coefficients, then subtract the exponent of the divisor from the exponent of the dividend.
Ex. (9 x 10 7 ) ÷ (3 x 10 -3 ) = 3 x 10 10
Note: Any number raised to a power of 0 is equal to 1: thus, 9.2 x 10 0 is equal to 9.2.
Scientific Notation
• Practice Problems, page 42-43
– #13-16
– Write the problem, then the answer
– Show your work, if necessary
Dimensional Analysis
• Dimensional analysis is a systematic approach to problem solving that uses conversion factors to move, or convert, from one unit to another.
• A conversion factor is a ratio of equivalent values having different units.
Dimensional Analysis
• Writing conversion factors
– Conversion factors are derived from equality relationships, such as 1 dozen eggs = 12 eggs.
– Percentages can also be used as conversion factors. They relate the number of parts of one component to 100 total parts.
• Practice Problems, page 45
– #17-18
– Write the problem, then the answer
– Show your work, if necessary
Dimensional Analysis
• Using conversion factors
– A conversion factor must cancel one unit and introduce a new one.
• Practice Problems, page 45-46
– #19-23
– Write the problem, then the answer
– Show your work, if necessary
Dimensional Analysis
• Answer questions on Teaching Transparency 4
– Converting Units
Homework, Section 2
• SECTION 2 REVIEW, Page 46
• Questions #24-28, 30, 31
• Answer with complete sentences
• Due tomorrow
Section 3: Uncertainty in Data
• National Standards:
– UCP.1 Systems, order, and organization
– UCP.3 Change, constancy, and measurement
– A.1 Abilities necessary to do scientific inquiry
– A.2 Understandings about scientific inquiry
– E.2 Understandings about science and technology
– G.2 Nature of scientific knowledge
Objectives – Section 3
• Define and compare accuracy and precision.
• Describe the accuracy of experimental data using error and percent error.
• Apply rules for significant figures to express uncertainty in measured and calculated values.
• Review Vocabulary:
• experiment: a set of controlled observations that test a hypothesis
New Vocabulary
• accuracy
• significant figures
• percent error
• precision
• error
• Measurements contain uncertainties that affect how a result is presented.
Accuracy and Precision
• Accuracy refers to how close a measured value is to an accepted value.
• Precision refers to how close a series of measurements are to one another.
Accuracy and Precision
• Error is defined as the difference between an experimental value and an accepted value.
Accuracy and Precision
• The error equation is error = experimental value – accepted value.
• Percent error expresses error as a percentage of the accepted value.
Accuracy and Precision
• Practice Problems, page 49
– #32-34
– Write the problem, then the answer
– Show your work, if necessary
• Teaching Transparency 5 – Precision and
Accuracy
• Problem-Solving Lab: Identify an Unknown
– Page 50
Significant Figures
• Often, precision is limited by the tools available.
• Significant figures include all known digits plus one estimated digit.
Significant Figures
• Rules for significant figures
– Rule 1: Nonzero numbers are always significant.
– Rule 2: Zeros between nonzero numbers are always significant.
– Rule 3: All final zeros to the right of the decimal are significant.
– Rule 4: Placeholder zeros are not significant. To remove placeholder zeros, rewrite the number in scientific notation.
– Rule 5: Counting numbers and defined constants have an infinite number of significant figures.
Significant Figures
• Practice Problems, page 51
– #35-37
– Write the problem, then the answer
– Show your work, if necessary
Problem-Solving Lab: Identify an
Unknown
• Title: Identify an Unknown, page 50
• Objective: To observe how mass and volume data for an unknown sample be used to identify the unknown
• Data: Calculate the volumes and density for each of the six samples. Calculate the average density of the six samples. Be sure to use significant figure rules.
• Think Critically:
Rounding Numbers
• Calculators are not aware of significant figures.
• Answers should not have more significant figures than the original data with the fewest figures, and should be rounded.
Rounding Numbers
• Rules for rounding
– Rule 1: If the digit to the right of the last significant figure is less than 5, do not change the last significant figure.
– Rule 2: If the digit to the right of the last significant figure is greater than 5, round up the last significant figure.
– Rule 3: If the digits to the right of the last significant figure are a 5 followed by a nonzero digit, round up the last significant figure.
Rounding Numbers
• Rules for rounding
(cont.)
– Rule 4: If the digits to the right of the last significant figure are a 5 followed by a 0 or no other number at all, look at the last significant figure. If it is odd, round it up; if it is even, do not round up.
Rounding Numbers
• Addition and Subtraction
– Round the answer to the same number of decimal places as the original measurement with the fewest decimal places.
• Multiplication and Division
– Round the answer to the same number of significant figures as the original measurement with the fewest significant figures.
Rounding Numbers
• Practice Problems, page 53-54
– #38-44
– Write the problem, then the answer
– Show your work, if necessary
Homework, Section 3
• SECTION 3 REVIEW, Page 54
• Questions #45, 47-51
• Answer with complete sentences
• Due tomorrow
Forensics Lab: Use Density to Date a
Coin
• Title: Use Density to Date a Coin, page 60
• Objective: To observe how density data can be used to determine whether a penny was minted before 1982.
• Data: Find the mass and volume for a group of pennies. Graph your data. Calculate the average density of each group of pennies.
• Analyze and Conclude:
Section 4: Representing Data
• National Standards:
– UCP.1 Systems, order, and organization
– UCP.3 Change, constancy, and measurement
– A.1 Abilities necessary to do scientific inquiry
– A.2 Understandings about scientific inquiry
– B.2 Structure and properties of matter
– E.1 Abilities of technological design
– E.2 Understandings about science and technology
– F.1 Personal and community health
– G.2 Nature of scientific knowledge
Objectives – Section 4
• Create graphics to reveal patterns in data
• Interpret graphs.
• Graphs visually depict data, making it easier to see patterns and trends.
• Review Vocabulary:
• independent variable: the variable that is changed during an experiment
• New Vocabulary:
• graph
Graphing
• A graph is a visual display of data that makes trends easier to see than in a table.
Graphing
• A circle graph, or pie chart, has wedges that visually represent percentages of a fixed whole.
Graphing
• Bar graphs are often used to show how a quantity varies across categories.
Interpreting Graphs
• Teaching Transparency 6 – Interpreting Graphs
• Math Skills Transparency 1 – Interpreting and
Drawing Graphs
Graphing
• On line graphs, independent variables are plotted on the x-axis and dependent variables are plotted on the y-axis.
Graphing
• If a line through the points is straight, the relationship is linear and can be analyzed further by examining the slope.
Interpreting Graphs
• Interpolation is reading and estimating values falling between points on the graph.
• Extrapolation is estimating values outside the points by extending the line.
Interpreting Graphs
• This graph shows important ozone measurements and helps the viewer visualize a trend from two different time periods.
Homework, Section 4
• SECTION 4 REVIEW, Page 58
• Questions #52-58
• Answer with complete sentences
• Due tomorrow
Key Concepts
• SI measurement units allow scientists to report data to other scientists.
• Adding prefixes to SI units extends the range of possible measurements.
• To convert to Kelvin temperature, add 273 to the
Celsius temperature. K = ° C + 273
• Volume and density have derived units. Density, which is a ratio of mass to volume, can be used to identify an unknown sample of matter.
Key Concepts
• A number expressed in scientific notation is written as a coefficient between 1 and 10 multiplied by 10 raised to a power.
• To add or subtract numbers in scientific notation, the numbers must have the same exponent.
• To multiply or divide numbers in scientific notation, multiply or divide the coefficients and then add or subtract the exponents, respectively.
• Dimensional analysis uses conversion factors to solve problems.
Key Concepts
• An accurate measurement is close to the accepted value.
A set of precise measurements shows little variation.
• The measurement device determines the degree of precision possible.
• Error is the difference between the measured value and the accepted value. Percent error gives the percent deviation from the accepted value.
error = experimental value – accepted value
Key Concepts
• The number of significant figures reflects the precision of reported data.
• Calculations should be rounded to the correct number of significant figures.
Key Concepts
• Circle graphs show parts of a whole. Bar graphs show how a factor varies with time, location, or temperature.
• Independent (x-axis) variables and dependent (y-axis) variables can be related in a linear or a nonlinear manner.
The slope of a straight line is defined as rise/run, or
∆y/∆x.
• Because line graph data are considered continuous, you can interpolate between data points or extrapolate beyond them.