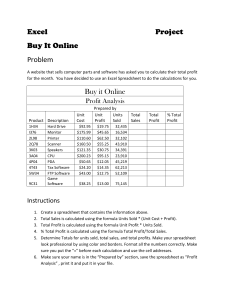
Physics Lab: Measurement & Error Analysis with Pennies
advertisement

Wednesday 11 January 2012 Class meeting Topics Textbook sections Ponderables Mini-labs (deliv.) Lab Demonstrations Mini-lectures Quiz Other PHYS 116 SCALE-UP 2 Measurement Chap. 1 Moon Pie Common Cents (spreadsheet) FCI Questions or concerns about class procedure, including Mastering Physics (first HW assignment due Monday) FCI (35 minutes): diagnostic, not part of grade, helps evaluate teaching Mini-lab: Common Cents (45 minutes): First example of a mini-lab. Explain deliverable. o Equipment: 3-5 digital balances, ~200 pennies, at least one Vernier caliper for each group (more is better) Ponderable: Moon Pie (20 minutes) 1 Wednesday 11 January 2012 PHYS 116 SCALE-UP COMMON “CENTS” In this exercise, you will investigate physical properties of the U.S. Lincoln one-cent coin, commonly known as a penny. By doing so, you will apply basic scientific reasoning skills and gain experience making measurements with common laboratory instruments and properly reporting results. Pre-lab (to be turned in at the beginning of next class): A few years ago a sample of pennies was collected from general circulation. A histogram showing the number of pennies from each year is provided below. 70 60 50 40 30 20 10 2002 2000 1998 1996 1994 1992 1990 1988 1986 1984 1982 1980 1978 1976 1974 1972 1970 1968 1966 1964 1962 1960 0 Answer the following questions in a few sentences each: What inferences can you make from this sample? Why does this histogram not follow the normal (Gaussian or bell-shaped) distribution curve? What year do you think the sample was obtained? Is there anything you find surprising about this sample? 2 Wednesday 11 January 2012 PHYS 116 SCALE-UP Explorations You will be shown the basics of using a vernier caliper (written instructions are available on the Sakai site in the document “Measuring Instruments” in the “Course Information” section of the site); you will also be instructed briefly on using the digital scale. Finally, you will be introduced to the idea of uncertainty and how you might propagate it by the upper/lower bound method when calculating an answer based on several uncertainties. All responses are to go in your lab notebook. Determining density: Determine a basic formula for density and calculate the density of a penny. Use vernier calipers and a digital balance to find the density of a typical penny as precisely and accurately as possible. Carefully consider the procedure you will use to accomplish this task. Report your intermediate and final results in your lab notebook, using the descriptions below as a guide. Be sure to propagate your uncertainties in the individual measurements to the final value of the density. Mass = _______ ± ______ g Diameter = ________ ± ______ cm Volume = ______________ ± ______________ Thickness = _______ ± ______ cm Density = _______________ ± ______________ Comparing values: Do your results agree with the findings of other groups? In thinking about why or why not, examine the physical properties of several pennies and think about how they are similar and different. Make brief notes about your observations. Of the three direct measurements you made for calculating the density, which was least precise and why? Record your findings in your lab notebook. Deliverable: Create an Excel-compatible spreadsheet and enter your measurements (and uncertainties) for mass, diameter, and thickness. Use the spreadsheet to calculate the density and its uncertainty using both the upper/lower bound approach and the propagation of uncertainty in quadrature (put the uncertainty values calculated by these two different methods into different columns of your spreadsheet). Format your results to an appropriate number of significant figures. Refer to the document “Measurement and Error Analysis” posted on the class Sakai site (in the “Course Information” section) for more information about the upper/lower bound method and propagation of uncertainty in quadrature (see pgs. 12-16). Submit your spreadsheet via the Sakai site before the next class meeting. Remember, “It is better to be roughly right than precisely wrong.” ( Alan Greenspan) 3 Wednesday 11 January 2012 PHYS 116 SCALE-UP MOON PIE A cup (U.S. measure) of cream cheese has a mass of 8.18 ounces. If the Moon were made of cream cheese, what would be its diameter (assuming it had the same mass as the real Moon)? What does that tell you about what the Moon is made of? 4