The pH Scale - Lake Elkhorn Wiki Home
advertisement

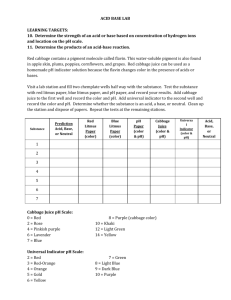
The pH Scale Is a substance an acid or a base? ACIDS An acid is a compound that increases the number of hydrogen ions when dissolved in water (H+) An acid solution tastes sour An acid can change the color of certain compounds What is an ion? **An ion is an atom that has lost or gained an electron giving it a positive (+) or negative (-) charge. ** Charged Atom USES OF ACIDS Citric acid and ascorbic acid (Vitamin C) are found in orange juice Carbonic acid and phosphoric acid give a “bite” to soft drinks Acids in your stomach aid in digestion Sulfuric acid is the most widely used industrial acid in the world. It is used in making metals, paper, paints, and fertilizers. BASES A base is any compound that increases the number of hydroxide ions (OH-) when mixed with water A base solution tastes bitter A base solution feels slippery A base can change the color of certain compounds Uses of Bases Bases are used in soaps (remember, bases are slippery) Bases are used to make paper, in oven cleaners, and to unclog drains Ammonia is a base and is used in many household cleaners Detecting Acids and Bases You can detect an acid using an indicator. INDICATOR - A substance that changes color in the presence of an acid or a base. INDICATORS Litmus paper is an indicator An acid turns blue litmus paper red A base turns red litmus paper blue Basic 7 Acid 14 Neutral pH Scale Acidic 0 Base Zumdahl, Zumdahl, DeCoste, World of Chemistry 2002, page 515 [H+] pH 10-14 14 10-13 13 10-12 12 10-11 11 10-10 10 10-9 9 10-8 8 10-7 7 10-6 6 10-5 5 10-4 4 10-3 3 10-2 2 10-1 1 100 0 1 M NaOH Ammonia (household cleaner) Blood Pure wate Milk Vinegar Lemon juice Stomach acid 1 M HCl pH of Common Substances Timberlake, Chemistry 7th Edition, page 335 Did we Miss something?? What happens when the pH of a substance is 7? Ans: A pH level of 7 indicates a Neutral Substance i.e: Water! Why Learn about pH? What do you think is the pH level of NYC tap water? The pH of a swimming pool must be checked periodically. Why? Is it important for Lakes & Rivers to maintain a certain pH? How do Antacids Work? Normal pH in your stomach is 3 If your stomach pH drops below 3 you don’t feel so well An antacid neutralizes the acid in your stomach and brings the pH up to a 3 or 4 Antacids A base can't neutralize your acid all by itself. A base needs some chemical "helpers” All antacids contain at least one of these four primary ingredients: * Sodium * Calcium * Magnesium * Aluminum. HOW DO WE MEASURE pH? N E U T R A L ACID 0 1 2 3 4 5 6 7 BASE 8 9 10 11 12 13 14 The pH Scale N E U T R A L ACID 0 1 2 3 4 5 6 7 BASE 8 9 10 11 12 13 14 The pH Scale Let’s see what you’ve learned! Professor Know-ItAll wants to know what you’ve learned. Answer the following questions carefully. Question #1 Lemon juice has a pH of 2.2. Is it an acid or a base? Question #2 Detergents such as Tide have a pH of about 10. Is Tide an acid or a base? Question #3 Seawater has a pH of 8.2. Is seawater an acid or a base? Question #4 The inside of your mouth has a pH of 7. Is it an acid or a base? Question #5 How do you think we came up with the term “acid rain”? Question #6 409 cleaner contains ammonia that has a pH of 12. What will 409 do to red litmus paper? Question #7 A soft drink has a pH of 3.2. What will it do to a piece of red litmus paper?