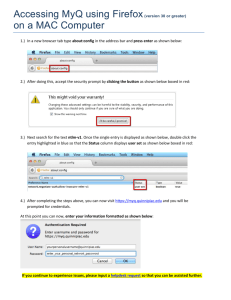
Boxed Warning Overview - New York State Council of Health
advertisement

Define Boxed Warning (BW) Legislature behind Boxed Warnings Describe and give examples of BW addition process Consequences of BW addition Provide list of top prescribed medications that have Boxed Warnings Evaluate frequency of physicians prescribing medications with BW and compliance with warnings List alert systems that notify prescribers and consumers of newly issued warnings List sources of information regarding medication alerts What is Boxed Warning? Boxed Warning – strongest warning that FDA requires on labeling of prescription medication Boxed Warning – brief summary of information critical for prescriber to be aware of, including restrictions on distribution or use ► Risk of serious adverse reaction ► Proper administration and monitoring requirements that can prevent or diminish serious adverse reaction ► Information on restriction of distribution and use of drug ► Important risk/benefit information unique to drug Only FDA can mandate addition of BW to labeling § 21 CFR 201.57(c)(1) Boxed Warnings in History 1938 - Federal Food, Drug, and Cosmetic Act (FDCA) - 60 day approval process 1912 – Sherley Amendment – prohibits labeling medicines with false therapeutic claims 1948 – Administrative Procedures Act – enforcement of labeling compliance 1962 - Kefauver-Harris Amendments to FDCA – drug has effects claimed by manufacturer; greater safety proof; FDA, not FTC regulates drug advertisement 1969 – Adverse Event Reporting System established 1970 – 1st patient package insert (oral contraceptives) 1979 - 21 CFR §201.56, §201.57 - Regulation of format and content of prescription drug labeling Boxed Warning Facts Nomenclature - “Boxed Warning”, sometimes referred to as ► “Black Box Warning” ► “Black Label Warning” ► Warning information displayed in box “black box” Boxed warning placement dictated by FDA ► Usually – at beginning of label Boxed warning information availability ► Post-marketing or at marketing FDCA – “drug is misbranded if it lacks adequate warning of possible danger” Adequate warning must appear on all drug labeling Results of Survey of 206 Drugs: Categories Identification and avoidance of high risk patients – 25% (95/375) ► E.g., PO contraceptives in smokers Important drug dosing and drug interactions – 20% (74/375) ► E.g., ketorolac Need for special training or special setting for drug administration – 18% (66/375) ► E.g., chemotherapeutic agents Drug administration information – 15% (57/375) Early detection of reversible condition – 12% (47/375) Benefits > Risks in certain situations – 10% (47/375) Beach JE et al. Food and Drug Law Journal 1998;53:403-411 What are BW Based on? “Ordinarily based on clinical data” ► Trials that were part of new drug application (NDA) – 28.7% (73/206) −E.g., zalcitabine ► Postmarketing reports – 52.4% (133/206) −E.g., felbamate ► Epidemiological surveys – 9.4% (24/206) −E.g., PO contraceptives in smokers Serious animal toxicity data in absence of clinical evidence Other miscellaneous sources – 9.4% (24/206) ► E.g., potential for abuse, not studied in certain patient groups (e.g., children) Beach JE et al. Food and Drug Law Journal 1998;53:403-411 Process of Boxed Warning Addition 1. Clinical data from post-marketing surveillance reported to MEDWATCH 2. FDA may refer ADE reports to Center for Drug Evaluation and Research (CDER) 3. CDER may refer information to FDA Advisory Committee for drug under evaluation 4. FDA Commissioner makes final decision ► Note: FDA Commissioner not obligated to follow FDA Advisory Committee − E.g., droperidol case – 2001 BW added − Anesthetic and Life Support Drugs Advisory Committee within the Center for Drug Evaluation and Research not consulted Dershwitz M. Journal of Clinical Anesthesia 2002;14(8):598-603 Repercussions of BW Addition Prescriber ► Ensure to follow BW advisory to avoid patient harm ► May lead to significant reduction of drug use for approved indications ► May lead to prejudice against drug ► Increased potential for litigation Pharmacist ► Ensure adequate counseling ► Perform adequate “checks and balances” ► Dispense medication guide (in some cases) Repercussions of BW Addition Pharmaceutical manufacturer ► Restricted advertisement (e.g., no “reminder ads”) ► Negative effects on drug sales −Seldane® sales dropped from $700 $450 million ► Decrease in prescribing ► Protects from product liability Patient ► Decreased compliance due to fear Boxed Warning Limitations Criteria and steps for BW placement not welldefined Compliance with BW Requirements Study Overview Objective ► To assess frequency of use of medications containing boxed warnings in ambulatory care and prescribing compliance with BW recommendations Design ► Retrospective study ► Data drawn from automated clams of members of HMO Research Network’s Center for Education and Research on Therapeutics ► 10 geographically diverse plans in US ► Data collection 01.01.1999 – 31.06.2001 Study Methods …cont’d Assessment of prevalence of dispensing BW medications ►N = 929,958 health plan members −Continuously enrolled −Had ongoing drug benefits Assessment of prescribing compliance with BW ►N = 216,694 ► Received 1 of 19 selected BW medications or groups of medications Sources of BW information ► Physician’s Drug Reference 1999 – 2001 ► Published lists of BW medications from Hosp Pharm 2002;37 issue BW Definition and Inclusion Drugs grouped by pharmacologic class if they all carried same BW BW classification ► Applies to all patients ► Applies to children (0 – 14 y/o) only ► Applies to women only ► Applies to women of childbearing age (15 – 44 y/o) only Other BW included ► Drugs requiring laboratory monitoring ► Contraindications of coadministration ► Specified pregnancy as contraindication Study Definitions …cont’d Definition of non-compliant prescribing; noncompliance with: ► BW requiring laboratory monitoring before therapy initiation or during continued therapy ► BW listing contraindicated co-medications ► BW with pregnancy as contraindication Drugs with BW excluded from study if ► BW pertained to storage, handling, distribution, compounding directions, administration, or dosing guidelines Study Results: Potentially Relevant BW Results: 11 Most Common Drugs with BW ► BW warning against abrupt discontinuation − Atenolol − Metoprolol ► BW warning for restricted use for specific indications − Clindamycin − Levothyroxine − Metronidazole ► BW indicating drugs not to be used in some cases − Propoxyphene − Medroxyprogesterone BW Drugs Prescribed without Baseline Lab Tests ► No claims for baseline tests noted 49.6% prescribed in violation of BW recommendation! Results: Other Findings Missing claims for recommended laboratory tests with continuous therapy – 6,605 (12.8%) ► Valproic acid (29.5%) ► Isoniazid (25.2%) ► Various diuretics Dispensing drugs with BW against coadministration – 5,199 (9.3%) ► Methotrexate + NSAIDs ► Ketorolac + NSAIDs Drugs with BW contraindicated in pregnancy – 95 (0.3%) BW Drugs with Excellent Compliance Warning explicitly states pregnancy as contraindication Warnings listing contraindicated drug coadministration Concise and focused wording = better compliance Potential Barriers of Compliance with BW Recommendations Poor lay public education regarding boxed warning meaning and implications Inconsistent format of BW text Unclear message and/or non-specific requirements ► E.g., not listing specific contraindicated medication ► E.g., not specifying how often laboratory test should be done Criteria for BW to be added Timing of New BW Addition and Drug Withdrawal from Market Objective ► To determine frequency and timing of new BW addition or withdrawal of prescription medications PDR review 1975-1999 ► 548 new drugs approved ► 56 (10.2%) new Boxed Warning added or withdrawn −45 (8.2%) 1 Boxed Warning −16 (2.9%) withdrawn (5 with new BW) Lesser KE et al. JAMA 2002;287(17):2215-2219. BW Addition or Market Withdrawal …cont’d Kaplan-Meier analysis ► 20% chance for withdrawal or new BW for drug over 25 years ► ½ package insert changes within 7 years of approval ► ½ withdrawals within 2 years of approval Boxed Warnings inconsistencies noted ► Asynchronous appearances of BW among drugs in same class ► Not all drugs in same class have BW −E.g., Beta blockers (atenolol vs. bisoprolol) ► Asynchronous appearances of BW among interacting medications Lesser KE et al. JAMA 2002;287(17):2215-2219. U.S. Drugs on Market >4,300 drugs on U.S. market 428 drugs have Boxed Warning ► ~ 10% drugs carry Boxed Warnings Top prescribed drugs with BW from 2007 1. Fluticasone+salmeterol (Advair® Diskus) 2. Quetiapine (Seroquel®) 3. Venlafaxine (Effexor XR®) 4. Escitalopram (Lexapro®) 5. Pioglitazone (Actos®) 6. Risperidone (Risperdal®) 7. Aripiprazole (Abilify®) 8. Duloxetine (Cymbalta®) 9. Lamotrigine (Lamictal®) 10. Olanzapine (Zyprexa®) www.drugs.com/top200.html New Boxed Warnings 02/26/2009 – new BW for metoclopramide December 2008 ► Avandamet ► Avandaryl ► Myozyme November 2008 ► Premarin vaginal cream ► Truvada ► Viread ► Fluoroquinolone antibiotics ► Ontak IV ► Raptiva for injection ► Regranex gel Self Assessment Question Which statement is true regarding Boxed Warnings? A. 17% of approved medications on US market have boxed warning B. Boxed warning can only be placed in package insert C. Boxed warning labeling does not always apply to all patients D. If manufacturer is aware of severe adverse drug event of their medication, it may place Boxed Warning initially, but FDA may recommend deleting it later on