Chapter 11 PowerPoint
advertisement

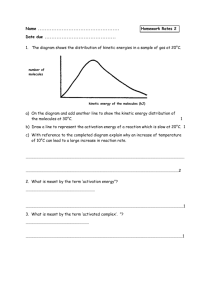
To accompany Inquiry into Chemistry PowerPoint Presentation prepared by Robert Schultz robert.schultz@ei.educ.ab.ca Chemistry 30 – Unit 1C Activation Energy and Catalysts Section 11.1 Reaction Pathways Chapter 11, Section 11.1 • Reaction rate • Theory: Collision Theory • To be an effective collision, need: • Correct collision geometry • Sufficient collision energy Chapter 11, Section 11.1 • Correct collision geometry • Textbook illustration for • NO(g) + NO3(g) 2 NO2(g) Chapter 11, Section 11.1 • Activation energy, Ea – the minimum collision energy required for a successful reaction (given that collision geometry is correct) • As temperature increases (recall: temperature is a measure of average Ek of the particles) more particles have sufficient energy • See figure 11.4 page 405 Chapter 11, Section 11.1 • As a result at a higher temperature, reactions tend to be faster Chapter 11, Section 11.1 • Recall Ep diagrams, Chapter 9 • More complete Ep diagrams: Chapter 11, Section 11.1 • In general, reactions with low activation energies tend to be faster than reactions with higher activation energies • It is impossible to predict reaction rates (or whether or not a reaction is spontaneous) from enthalpy changes Chapter 11, Section 11.1 • Note that forward and reverse activation energies are both shown on the graphs • Your book gives the following relationships: Exothermic: Ea (rev ) Ea ( fwd ) H Endothermic: Ea (rev ) Ea ( fwd ) H Book has r H ; this is a mistake Rather than memorize these I would encourage you to merely look at the graphs: Chapter 11, Section 11.1 • To get Ea (fwd), measure difference from reactants to transition state • To get Ea (rev), measure difference from products to transition state Ea (rev) Ea (fwd) Ea (fwd) ∆H ∆H Can you justify the relationships on the previous page? Ea (rev) Chapter 11, Section 11.1 • The chemical species that exists at the transition state in an Ep diagram is called the “activated complex” • Figure 11.8, page 408 in your text, shows an Ep diagram with a “reaction snapshot sequence” • This is theoretical, these are not actual photos Chapter 11, Section 11.1 Chapter 11, Section 11.1 • Try Practice Problems 2 and 4 page 409 Chapter 11, Section 11.1 • Do 11.1 Review, page 410, • questions 1, 2, 5, 6 (hint: draw a rough diagram), 8 Chapter 11, Section 11.2 • Catalyst: a substance that increases the rate of a chemical reaction without being consumed by the reaction • Considering what you have learned about reaction rates can you think of how a catalyst might accomplish this? • Catalysts provide an alternate reaction pathway with a lower activation energy figure 11.10, page 412 Chapter 11, Section 11.2 Review question 4 at top of page 413 Chapter 11, Section 11.2 • Question 4 lowers • a) A catalyst • b) A catalyst lowers • c) A catalyst has no effect increases • d) A catalyst of the forward reaction increases • e) A catalyst of the reverse reaction Ea (fwd) Ea (rev) on ΔH the speed the speed Chapter 11, Section 11.2 • Note the series of 4 photographs in figure 11.11 on page 413 Chapter 11, Section 11.2 • Read pages 413 to 414 about catalysts in industry and catalysts in car exhaust systems • Discuss • Read about enzymes, pages 415 to 416 • Do 11.2 Review, page 418, questions 1-6 Chapter 11, Section 11.2