Vulva,_Cervix,_Uterus_Handout
advertisement

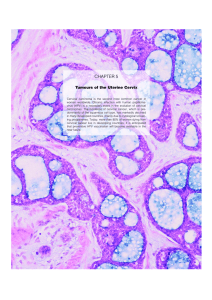
VULVAR DYSTROPHY (leukoplakia) • Age: any but most menopausal • Gross: smooth-surfaced, dry, stiff, white, atrophic vulva • Micro: − lichen sclerosus: epidermal atrophy, dermal fibrosis − squamous hyperplasia: epithelium thick, hyperkeratotic (now called lichen simplex chronicus) • Effects: fissures, uclers, infections, pruritis; few ca • Dx: biopsy • Differential Dx: lichen sclerosus, lichen simplex chronicus, chronic dermatitis, Paget Disease, VIN, vulvar cancer • Take home message – you have to biopsy these legion to check for malignancy VULVAR NEOPLASMS: BENIGN • Papillary hidradenoma − localized, benign, sweat gland tumor − structure: papillae of ductal lining cells − papillary structure − well circumscribed, localized mass • Condylomas (STD-related warts) − accuminatum: caused by human papilloma virus infection (HPV) koilocytosis (distinct cell type that is different histologically) − lata: syphilitic wart VULVA CARCINOMA • Precursor: vulvar intraepithelial neoplasia (VIN) − some pts present with vulvar dystrophy (leukoplakia) − Progressive grades of dysplasia I, II, III (III also called Bowen’s disease or carcinoma in situ; high grade or severe dysplasia) − HPV associated (especially types 16 & 18) − Concurrent vaginal and/or cervical CA in ~20% • Extramammary Paget disease − Presents as pruritic, red, crusted, welldelineated lesion usually on labia majora − Unlike Paget disease of breast - rarely associated with underlying invasive cancer − Confined to epidermis, hair follicles and sweat glands, believed to rise from these adenexal structures Becky Stepan − − Long survival, but may recur following surgical excision Paget cells contain mucin VULVA: CARCINOMA • Invasive carcinoma: may be preceeded by vulvar intraepithelial neoplasia (VIN) − squamous cell ca = 88% − spread pelvic and inguinal nodes − Rx: radical resection vulva, pelvic + groin nodes − Prognosis: no nodes + = 85% 5 yr survival Groin nodes + = 66% pelvic nodes + = 25% − Dx often delayed • Verrucous squamous cell carcinoma: − rarely metastasizes − histologically bland & grossly mimics condyloma − tend to be cured by surgery, usually a localized problem • Adenocarcinoma: few − from sweat glands? − Bartholin’s? • Melanoma: − 5% of vulvar cancer − delay in Dx 30% 5 yr survival VULVA: BARTHOLIN’S GLAND • Normal: − columnar-lined − mucus-secreting cyst − narrow neck • Chronic inflammation scarring obstructs neck cyst • Acute inflammation (often gonococcus, chlamydia) abscess VULVO-VAGINAL INFLAMMATION • Gonorrhea in children – presents as a purulent discharge • Trichomonas (strawberry mucosa — red) – very inflamed mucosa; strawberry cervix, but can involve the vagina and vulva • Moniliasis (candidiasis): white patches – loaded with fungus • Herpes (vesicles) – presents as papules then ulcers • Senile vaginitis (drynessulcers, fissures) – as the estrogen tapers off, the epithelium changes and it dries out. It can itch and be very easily irritated. May get ulcers, fissures. Very common problem. Treated by estrogen creams. Froberg – Vulva & Vaginal Path, Cervical Path, & Uterine Path Page 1 VAGINA: CONGENITAL LESIONS • Imperforate hymen: − Hematocolpos (blood filling uterus) − reflux • Septate (“double”) vagina • Blood is very irritating to the CNS and the peritoneum. Blood in these areas can be a problem. VAGINA: VAGINAL ADENOSIS • Islands of cervical glands beneath squamous ectocervix − cervical eruption • Cause: − rarely congenital − most = maternal exposure to diethylstilbesterol (DES) (synthetic estrogen) • Asymptomatic but 0.1% adenocarcinoma • Actually common finding VAGINAL: CANCER • Types: squamous cell carcinoma; adenocarcinoma; sarcoma botryoides; yolk sac tumor • Squamous cell carcinoma: − precursors: ca cervix or vulva; intraepithelial neoplasia (VIN) − spread: upper pelvic nodes, lower vagina inguinal − staging: like cervix − prognosis is stage dependent: stage 1 = 80% 5 yr; 3-4 = <20% • Adenocarcinoma: clear cell type = DES-related; young • Embryonic rhabdomyosarcoma (sarcoma botryoides) − age: <5yr − gross: rounded, grape-like (Greek: Botrys = grape) bulky mass − histology: small blue cell tumor may show myoblastic or strap cells; striations in some − behavior: locally destructive; large ones metastasize − Prognosis good if found and treated early Becky Stepan • Endodermal sinus (yolk sac) tumor − like same tumor in ovary − histo: sheets & Schiller-Duval body (central blood vessel surrounded by 2 layers of germ cells) − synthesizes alpha fetoprotein (hyaline droplets that stain + by IHC; also in blood) and alpha-1-trypsin − prognosis: awful CERVIX: NORMAL ANATOMY • External os • Internal os • Endocervical canal • Transitional (transformation) zone CERVIX: BENIGN • Acute cervicitis: gonococcus or chlamydia; also herpes et al. • Chronic cervicitis: squamous metaplasia of endocervical mucosa. This obliterates mouth of mucus glands Nabothian cysts erosions and uclers, simulate cancer • Endocervical polyps: soft, edematous stroma, epithelial-covered may erode bleed. They are another cause of vaginal bleeding. • “Pill” cervix: microglandular hyperplasia (progesterone effect?) CERVIX: CANCER • Epidemiology: − 50,000 precancerous cases/yr, but 13,000 invasive/yr (75% prevented-Rx or spontaneous regression) − 13,000 invasive cases/yr, but <5,000 die (60% cure rate) Papanicolaou smear = effective one of few examples of value of early cancer detection • Risk factors − early age of first sexual intercourse − multiple sexual partners − male partner who has multiple other partners − penile condylomas • HPV virus (Human papillomavirus) − causes condyloma accuminatum & warts (virus remains episomal) − Types 16, 18, 31, 33 stainable in precancerous cervical mucosa (virus integrates into host genome), therefore capable of transforming it to a malignant phenotype Froberg – Vulva & Vaginal Path, Cervical Path, & Uterine Path Page 2 − − − Transforms squamous epithelial cells in vitro E6 oncoprotein product of HPV binds with and degrades p53 E7 oncoprotein binds hypophosphorylated pRb, frees E2F transcription factor to drive cell cycle CERVIX: CANCER • HPV virus (Human papillomavirus) found in ~90% of tumors • HPV infected cells called koilocytes − nuclear size of the cell, Nuclear/Cytoplasmic ratio with the nucleus dominating the cell, irregular nuclear contours, hyperchromasia and perinuclear clearing • Dysplastic cells also show loss of polarity which means they have a disorganized growth, & atypical mitoses • HPV types 16 & 18 are found in high-grade cervical lesions; over 90% of invasive cancers will have 16 & 18 • HPV 6 & 11 – these are commonly found in condylomas • HPV 31, 33, & 35 are also found in cancers but not as much as 16 & 18 Cervical Cancer Classification of precancerous lesion • Cervical intraepithelial neoplasia: CIN • Progresses through succession (CIN I II III) of increasing degrees of dysplasia without invasion mild, moderate, severe (same as CIN III) • CIN III also called “carcinoma in situ” BIOLOGICAL features of Cervical Cancer: • % of cases progressing to next-highest grade with the grade (i.e., % CIN II III > I II) • Most mild dysplasia regresses; stage 2 goes to stage 3 more than stage 1 progresses to stage 2 • lesions begins @ squamo-columnar junction (“transformation zone”) • initial lesions may be any grade • time in any grade varies from months to many years • detection: grossly visualizing the lesion or an iodine (Schiller’s) test—stains glycogen in normal cells DIAGNOSIS of Cervical Cancer: • screening: “pap” smear • definitive diagnosis: − pap smear repeat − colposcopy Schiller test biopsy visible lesion conization • follow: with repeat smears & colposcopy CERVIX: CANCER • Invasive carcinoma − type: 80% = squamous cell carcinoma remainder: undifferentiated, adenosquamous & adenocarcinoma In DES-Rxed patients = clear cell carcinoma • Staging of invasive cervical cancer: − Stage 0: CIN III − Stage I: limited to cervix − Stage II: beyond cervix, upper 1/3 vagina, does not reach pelvic wall − Stage III: lower 1/2 vagina & reaches pelvic wall − Stage IV: beyond pelvis or invaded bladder or colon; distant metastases *If we can stop the virus, we can stop the progression to cancer. Becky Stepan Froberg – Vulva & Vaginal Path, Cervical Path, & Uterine Path Page 3 • • • Treatment of Cervical Cancer: − CIN I, II, III: pap, cryoRx, laser, conization, wire loop − invasive: hysterectomy and radioRx Prognosis: stage-dependent − Stage I = 90% 5 yr − Stage III = 10% Complications: local invasion bladder, ureters, colon UTERUS: INFLAMMATION • Acute bacterial − only in puerperium (strep, staph, clostridium, mixed) − NOT gonococcal, chlamydia or STD’s • Chronic bacterial − chronic PID (pelvic inflammatory disease) − tuberculosis − IUD − retained placental products − actinomycetes are found in these infections Uterus: ADENOMYOSIS (Endometriosis interna) • Non-malignant, non-neoplastic • Islands of endometrial glands and stroma deep in myometrium probably in continuity with endometrium • Benign but may cause − Menorrhagia − Dysmenorrhea − Dyspareunia − Pelvic pain • Seen in up to 20% of uteri Becky Stepan UTERUS: ENDOMETRIOSIS (EXTERNAL) • Endometrium at ectopic sites (tubes, ovary [most common site], sigmoid wall, uterine serosa, bladder, vulva, peritoneum, umbilicus, eye). • Found under the serosa, found in the muscle • Causes: mostly unknown, many theories − one theory is menstrual reflux, − metaplasia − lymphatic dissemination? − induction? Pre-existing epithelium − Iatrogenic (laproscopic hysterectomy) • Problem: they respond to hormones of menstrual cycle bleed (pain) − Infertility − intestinal obstruction − “chocolate” cysts infertility (chocolate cysts are old, dark blood clots) − dysmenorrhea, pelvic pain • Dx: biopsy endometrial glands, stroma, and/or hemosiderin pigment (need 2 of 3 for Dx) • Rx: hormonal; surgical. Often unsatisfactory because it can be difficult to find these because there are multiple lesions that cause obscure pain. Patients have chronic pain related to the menstrual cycle Dysfunctional Uterine Bleeding • Excessive bleeding during or between menstrual periods • Differential Dx includes polyps, endometrial hyperplasia, trophoblastic disease, adenomyosis, & carcinoma • Bx endometrium to rule out malignancy • Most often due to anovulatory cycle which leads to prolonged estrogenic stimulation. Froberg – Vulva & Vaginal Path, Cervical Path, & Uterine Path Page 4 Other Endometrial Changes • Oral contraceptives: − Inactive glands − Predecidualized stroma (abundant cytoplasm as in pregnancy) • Postmenopausal: − Senile cystic atrophy − Atrophic endometrium with cystic dilatation of glands UTERUS: ENDOMETRIAL HYPERPLASIA • Clinical presentation: abnormal uterine bleeding, usually post-menopausal • Mechanism: prolonged estrogenic stimulation (polycystic ovary, estrogen-producing tumors, estrogen Rx, etc.) • Histo: simple or complex cystic hyperplasia, adenomatous or atypical hyperplasia (25% of endometrial hyperplasia leads to cancer) • Dx and Rx: D&C; hormonal, surgery if postmenopausal then they will have a hysterectomy UTERUS: POLYPS • Pathology: cystic endometrial glands with stroma; benign (rarely cancer may arise within polyp), arise from the fundus, tend to bleed • Age: any but especially perimenopausal • Sx: ulceration on the surface bleeding • They infaract more than cervical polyps do. It is not unusual to be partially or entirely infaracted. • Rx: D&C UTERUS: LEIOMYOMA (“FIBROID”) • Location: submucosal, intramural, subserosal, broad ligament, cervix • Most common soft tissue tumor other than a lipoma; benign; estrogen-responsive. • CSx: bleeding; large obstruct birth (dystocia); spontaneous abortion; if large, can cause malformation of the fetus. • Histo: whorled fascicles of smooth muscle cells; clear cell and pleomorphic variations; also “intravenous leiomyomyosis ” which is not malignant even though it invades the lymph nodes • Leiomyosarcoma: arise de novo; they do NOT arise from Leiomyoma. − Sarcomatous histo − usually fatal *polyps and leiomyomas are the most common Becky Stepan UTERUS: ENDOMETRIAL ADENOCARCINOMA • Epidemiology: risk factors − postmenopausal − obesity (related to estrogenic effects) − diabetes − hypertension − infertility − hyperestrinism nulliparous anovulatory cycles estrogenic tumors estrogen therapy • Histology: − most endometrioid (adenocarcinomas with villoglandular histology) − some adenoca with foci of squamous metaplasia (adenoacanthoma) − some adenoca with malignant squamous foci (adenosquamous ca) − variations: clear cell ca papillary serous ca variations are very aggressive • Staging: similar to cervix • Grading: FIGO system, I < 5% solid, II = 5-50% solid, III > 50% solid (serous papillary & clear cell are grade III tumors) • Sx: postmenopausal bleeding • Dx and Rx: D&C; hysterectomy; radiation • Prognosis: stage 1 = 90%; III = 20% 5 yr UTERUS: Carcinosarcoma (Malignant Mixed Mullerian Tumor) • Histo: endometrial adenocarcinoma with malignant stromal differentiation − Muscle, cartilage, osteoid • Otherwise similar to poorly differentiated endometrial adenocarcinoma • Prognosis: overall = 25% 5 yr survival Endometrial Stromal Tumors • Two classes: − Benign stromal nodules wellcircumscribed aggregate of stromal cells within myometrium − Stromal sarcoma neoplastic endometrial stroma invading myometrium: Diffuse between muscle bundles, or Intralymphatic High recurrence rate (80% for stage III/IV) 5 yr survival~ 50% Froberg – Vulva & Vaginal Path, Cervical Path, & Uterine Path Page 5