Practice WS #3
advertisement

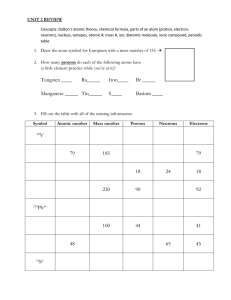
Atomic Structure Practice Name: ____________________________ Date: __________________Period: _____ 1. What can you always look up on the periodic table about an atom because it will never change? (atomic number, mass number, neutrons, electrons). 2. The atomic number is the number of ______ in an atom. 3. All atoms of the same element have the same number of _______. 4. Isotopes of the same element have different number of _________. 5. Determine the number of neutrons in 16O 8 6. What does the number 84 represent in the name krypton-84? 7. The sum of the protons and neutrons in an atom equals the __________. 8. The atomic number of an element is the number of ____. 9. Write the complete symbol for Carbon-14. 10.The particles that are found in the nucleus of an atom are ____ and ______. 11.How many protons, electrons, and neutrons does an atom with atomic number 50 and mass number 125 contain? Protons=___ , electrons= ______ , neutrons = ____ 12.Calculate the number of neutrons in Lead-210 13.When talking about the size, Protons and neutrons are ____amu and a electrons is ___amu. 14.The _______ is the identity of an atom. 15.Find each of these: a. number of protons 60 b. number of neutrons Fe 26 c. number of electrons d. Atomic number e. Mass Number 16.If an element has an atomic number of 20 and a mass number of 43, what is the: a. number of protons b. number of neutrons c. number of electrons d. complete symbol 17.If an element has 19 protons and 16 neutrons what is the a. Atomic number b. Mass number c. number of electrons d. complete symbol 18.Find each of these: a. number of protons b. number of neutrons c. number of electrons d. Atomic number e. Mass Number 14 C 19.What is the following for the isotope Lithium-10. a. b. c. d. e. f. Mass Number number of protons number of electrons number of neutrons Atomic number Complete symbol 20.Find each of these: a. number of protons b. number of neutrons c. number of electrons d. Atomic number e. Mass Number 16 7 N -3