20100326-EastHighAcidRain
advertisement

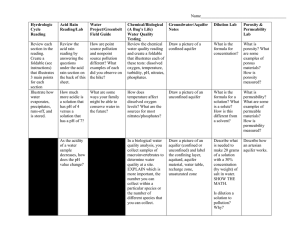
Line up the solutions in order of pH. Acidic ------------------neutral-------------------Basic pH 1 pH 7 pH 14 Lemon Juice (pH 2.3) Vinegar (pH 2.9) Mouthwash (pH 6) Water (pH 7) Baking Soda (pH 8.4) Detergent (pH 9.8) Windex (pH 11) Bleach (pH 13) Any substance, which when dissolved in water, tends to increase the amount of H+ HCl H+ (aq) + Cl- (aq) Properties: -corrosive, burns your skin -pH less than 7 -sour taste Any substance, which when dissolved in water, tends to increase the amount of OH- NaOH (s) Na+ (aq) + OH- Properties: Corrosive, burns your skin pH greater than 7 Soapy feel A form of pollution Has pH lower than 5.6 Can damage plants, animals, soil, water, building materials and people Scientists have discovered that air pollution from the burning of fossil fuels is the major cause of acid rain Can be transported over long distances Natural resources, including coal, fuel oil, and natural gas Created from the remains of dead animal and plant life that lived up to 300 million years ago. Found in deposits beneath the earth Over 85% of our energy demands are met by the combustion of fossil fuels. Power our cars, buses, airplanes, produces heat, light, tv, electricity Sulfur dioxide, nitrogen oxide Forms high in the clouds when reacts with water, oxygen, oxidants Write 4-6 sentences How do acids and bases affect your everyday life? When do you find yourself using/interacting with them? What is acid rain? How does acid rain affect our environment?