Chemistry of Life/ Carbon Compounds
advertisement

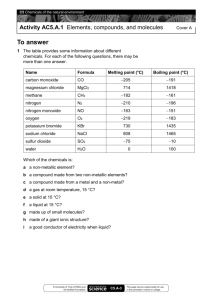
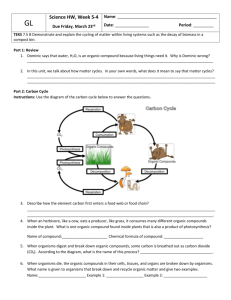
Chemistry of Life/ Carbon Compounds Chapter 10, Lessons 1 & 2 Elements of Life • Biomass is the total mass of all living matter on Earth, which is made up mostly of the elements sulfur, phosphorus, oxygen, nitrogen, carbon, and hydrogen (SPONCH) – 96% of which is hydrogen, oxygen, nitrogen, carbon (HONC) • Living organisms obtain these elements from their environments. Cycles in Life • All of the elements essential to life flow through food webs that are part of natural cycles. • The carbon cycle describes how carbon moves from the atmosphere into plants (where it is used to provide energy) , into animals that eat plants, and into the environment as waste from animals. Carbon Cycle • Plants obtain carbon from atmospheric carbon dioxide to make sugar molecules to store energy. • Animals eat the plants or other animals to obtain carbon. • Carbon dioxide is a waste product of breathing. • When fossil fuels are burned, they release carbon dioxide into the atmosphere. Carbon Cycle (cont’d) Nitrogen cycle • Plants cannot use atmospheric nitrogen. • Bacteria in the soil ‘fix’ nitrogen, changing it to a form that plants can take up through their roots. • Plants use the nitrogen for building cells. • Animals eat the plants and nitrogen is passed on. • Nitrogen is also passed on when one animal eats another. Nitrogen Cycle (cont’d) • The cycle is complete when decomposers break down dead organisms and return the nitrogen to the soil. Phosphorus Cycle • Natural processes break down rocks containing phosphorus. • Plants absorb phosphorus through their roots. • Consumers obtain phosphorus by eating plants or animals that have eaten plants. Water and Living Organisms • In addition to the elements that make up living tissue, living organisms cannot survive without the compound water. • Essentially, all of life’s processes occur in a water mixture. – The fluid properties of water allow blood to flow and carry substances through the body. The liquid portion of blood, called plasma, is 90% water. – Water requires more heat than other liquids to change temperature, which protects living organisms from temperature changes. – Solid water is less dense than liquid water. Ice, therefore, floats on liquid water and provides a protective layer for organisms living in water. Polar Molecule • Water is a polar molecule, which makes it an excellent solvent for other polar molecules necessary for living organisms. – A polar molecule is a molecule that has a positive end and a negative end because of an unequal sharing of electrons between its molecules. – A nonpolar molecule is a molecule that shares electrons equally and does not have oppositely charged ends. Polar Molecule (cont’d) • Individual water molecules act like magnets. • The positive end attracts the negative end of other water molecules. • This weak bonding is known as hydrogen bonding. Life-Sustaining Properties of Water Organic Compounds • An organic compound is one of a large group of compounds that always contain carbon. • Most of the compounds in living cells are organic compounds, but not all carbon compounds are organic compounds. Carbon Bonding • Carbon has atomic number 6 = 4 valence electrons. • Carbon can bond to itself and other elements in many different ways (chain, branched, ring). • Carbon can always make up to 4 bonds. • Carbon is unusual because it can form double and triple covalent bonds. Hydrocarbons • Molecules that contain only carbon and hydrogen atoms are called hydrocarbons. • They are flammable and burn easily – used for fuels. • Isomers – different arrangement of a formula. *Butane (C4H10) *Isobutane (C4H10) Butane Isobutane Hydrocarbons- Saturated vs. Unsaturated • Saturated CH have all single bonds and are saturated with hydrogens. (names end in –ane) • Unsaturated CH contain double or triple bonds and fewer hydrogens. (names end in –ene or –yne) Ethene Substituted Hydrocarbons • A functional group is a group of atoms that replace a hydrogen atom in organic compounds. – A functional group changes the properties of an organic compound from the original hydrocarbon on which it is based. • Organic compounds that contain a functional group are called substituted hydrocarbons. Hydroxyl Group • A hydroxyl group contains an oxygen atom and a hydrogen atom covalently bonded to one another (-OH). • A hydrocarbon substituted with a hydroxyl group is called an alcohol • Nonpolar hydrocarbons do not dissolve in water, but by adding a hydroxyl group, water-soluble compounds are formed. Carboxyl Group • Carboxylic acids contain the carboxyl group, –COOH, where the carbon is double-bonded to one of the oxygen atoms. – Aspirin is a carboxylic acid, as is lactic acid. Amino Group • Amino groups contain a nitrogen atom and two hydrogen atoms, –NH2. • Compounds that contain the amino group are called amines. – Many amines, such as histamine and thiamine, end with the suffix –amine. Amino Acids and Proteins • Substituted hydrocarbons with both an amino functional group and a carboxylic functional group are called amino acids. – An amino acid is a member of a class of organic compounds that are the basic building blocks of proteins. – Twenty common amino acids are used in your body to create different protein molecules. Human cells can create 11 of the 20 common amino acids, but you must get the other nine common amino acids from the foods you eat. Shapes of Molecules • Models can be used to understand the shapes of molecules. • The three-dimensional shapes of molecules help explain the properties of the compounds that consist of those molecules. • Tetrahedral molecules are shaped like pyramids. Shapes of Molecules (cont’d) • Planar molecules are flat like sheets of paper. Shapes of Molecules (cont’d) • Linear molecules are arranged in a line. Lesson Review Questions 1) Which is one of the six elements that make up most of Earth’s biomass? A helium B iron C nitrogen D sodium Lesson Review Questions 2) Water is a polar molecule because ____. A it has a negative charge B it has a covalent bond with unequal sharing of the electrons C it has a positive charge D it has an ionic bond Lesson Review Questions 3) Where do plants obtain nitrogen for cellular activities? A from the air B from decaying organisms C from animal waste D from nitrogen-fixing bacteria in the soil Lesson Review Questions 4) Which of the following is the amino group? A –OH B –NH2 C –COOH D CH4 Lesson Review Questions 5) Which type of molecule is like a flat piece of paper? A planar B linear C cubic D tetrahedral Lesson Review Questions 6) How many possible covalent bonds can carbon form? A one B two C three D four