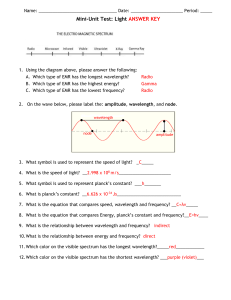
wavelength
advertisement

Planck’s constant in the light of an incandescent lamp Introduction The idea of light quanta • Planck (1900): emission of radiant energy by matter does not take place continuously, but in finite “quanta of energy” h (h= Planck’s constant 6.63x10-34 J.s, =frequency) • Einstein (1905): light quanta (photons) as inherent in the nature of radiation itself Distribution of intensity of heat radiation as a function of the wavelength : u c1 e 5 c2 T 1 Emissivity (=1 for perfect black-body radiation) C1 , C2 : Constant parameters Planck’s radiation law Radiation energy per time unit for the wavelength u c1 5 e c2 T Where 1 c2=hc/k h: Planck’s constant c: velocity of light k: Boltzmann’s constant Main objective of this experiment Report on the experiment Emission of a 12 V tungsten lamp u u wavelength, c1 c2 5 T e 1 The light spectrum emitted by the filament is continuous. u u wavelength, c1 c2 5 e T 1 A narrow band of the visible spectrum is selected with a combination of Orange II and Copper Sulphate solution (it absorbs infrared strongly). u u wavelength, 0 Liquid filter c1 c2 5 T e 1 We will assume that the selected band is nearly monochromatic. u u wavelength, 0 Liquid filter c1 c2 5 T e 1 The wavelength of the selected band is in the spectral response range of a Light Dependent Resistor (LDR) u u wavelength, 0 Liquid filter c1 c2 5 T e 1 From the formula: u c1 c2 5 T e 1 Block diagram h (1) c2 For small lnRldr u0 c1 c2 5 0T (2) 0 e 1/ T lllumination E on the LDR is proportional to the transmitted energy E u 0 ln R ln c3 (3) Resistance R of LDR is related to illumination as: R b E b: constant : parameter (4) Plotting c 2 1 0 T (6) Taking logarithms Combining (2), (3) and (4): R c3e c2 0T (5) Experimental setup GENERAL DIAGRAM Solution filter Potentiometer LDR Lamp Ohmeter Battery Voltmeter V A Ammeter COMPONENTS Battery Cover Potentiometer LDR Lamp Platform Solution filter Holder V A Grey filter Ruler Ohmeter Voltmeter Ammeter INSTALLING THE EQUIPMENT 1 Turn the potentiometer knob anticlockwise up to the limit 2 Turn slowly the tube holder aligning the lateral holes between the lamp and the LDR. 3 Move the LDR towards its lateral hole, positioning its surface as the figure shows. 4 Insert the solution filter tube in its holder. 5 Put the cover onto the platform to protect from the outside light. In order to ensure the correct initial conditions, LDR should keep in total darkness for at least 10 minutes before the measurements. Procedure Some previous measurements are needed before using Equation (6) c 2 1 ln R ln c3 T R (6) T 0 T Temperature of the emmitting filament Relation between the resistance of the filament (RB) and its temperature (T) RRB Experimental data fit T aRB RRB00 0 ,83 I a can be derived from the filament resistance (RB0) at room temperature (T0) T0 a 0.83 RB0 Using the multimeter as a thermometer. RB0 can be extrapolated to I = 0 from measurements of V and I, V A transmission of the filter Solution of: - Orange II. - CuSO4 (it absorbs the infrared light). 0 = 590 nm % transmitance 35 30 25 20 15 10 5 0 450 500 550 600 650 700 750 l/nm /nm Parameter of the LDR En Rn bE Rn Rn ln ' ln 0.512 Rn Grey filter 0.512En R ’ n R b(0.512 E ) ' n COLLECTING DATA R V A I V RB=V/I T = aRB0.83 RB-0.83 R lnR I1 V1 RB1 T1 RB1-0.83 R1 lnR1 I2 V2 RB2 T2 RB2-0.83 R2 lnR2 I3 V3 RB3 T3 RB3-0.83 R3 lnR3 In Vn RBn Tn RBn-0.83 Rn lnRn lnR m c 2 0 a RB-0.83 From the slope lnR c 2 m 0 a RB-0.83 We obtain c2 m0 a And finally the Planck´s constant: kc2 h c h: Planck´s constant. k: Boltzmann´s constant. c: speed of light. End of presentation