Chem. 31 – 9/15 Lecture
advertisement

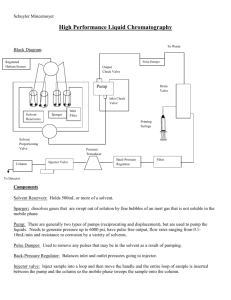
Chem. 231 – 3/4 Lecture Announcements I • Set 1 Lab Reports – Both Lab Reports due 3/13 – Only one of the lab reports needs to be detailed, the other can be a summary • Homework Set 3 – due Wednesday • Quiz 3 (last quiz) – next Monday • Set 2 Labs (two; need to do one of them) – SPE/HPLC lab – SPME/GC lab – More instructions coming Announcements II • Set 3 Labs – Hope to have instructions completed by Monday • Today’s Lecture – SPE Lab Information – SPME Lab Information – Practical Applications of HPLC SPE/HPLC Lab Information • Goal: Determine concentration of unknown phenol in aqueous solution • Background: – Phenols are possible soil contaminants (e.g. pentachlorophenol and nitrophenols) and can contaminate water supplies – Phenols are also in some beverages (e.g. vanillin, other guaiacols in mostly aqueous environment) – Solid Phase allows trapping of moderately non-polar compounds needed for sensitive analysis R OH OCH3 guaiacols SPE/HPLC Lab Information – Methodology • Overview – Unknowns will need to be trapped on the SPE cartridge, then eluted (with smaller volume) to concentrate samples – Analysis by HPLC (will need to develop method and determine elution order as in set 1 lab) • SPE Procedure – Clean SPE cartridges (first with removal eluent, then with sample solvent) – Flow rate must be slow enough for transfer – Apply sample to SPE cartridges SPE/HPLC Lab Information – Methodology • SPE Procedure – cont. – Wash sample with sample solvent (removes polar contaminants from pores) – Remove analytes with stronger solvent – If removing into volumetric flask, need to fill to line (more quantitative method) • Unknown Analysis – Will want to use SPE method on unknown to determine unknown compounds SPE/HPLC Lab Information – Methodology • SPE Efficiency Testing – To determine if unknown compound losses (in passing through SPE cartridge or in removal from SPE cartridge) are significant – Done by keeping and analyzing multiple fractions SPME/GC Lab Information • Goal – Determine the concentration of two flower scent monoterpene unknowns in a sample • Background – Many of the scent compounds in flowers are monoterpenes (consist of two isoprene units) – Traditional analysis would involve extraction and liquid injection – Alternative of SPME is solvent free H C 3 CH2 H3C myrcene H2C SPME/GC Lab Information - Methodology • GC – Methodology – Requires splitless injection (so can’t use Buck GCs) – We will be using an HP6890 and an Agilent 7890 GC-MS (this will also give qualitative analysis) – These have software control closer to the Agilent HPLCs – See 2/4 lecture, slide 16 for procedure using SPME SPME/GC Lab Information - Methodology • GC – Methodology – cont. – SPME equipment is fragile • SHEATH ALWAYS ON WHEN GOING IN OUR OUT OF SEPTA OR DAMAGE OCCURS • NO CONTACT WITH DICHLOROMETHANE – SPME fibers have limited lifetimes under normal use and may need replacing – Besides what is shown in past slide, SPME must be pre- and/or post- heated (can use injector for this if conditions are o.k.) SPME/GC Lab Information - Methodology • GC – Methodology – cont. – Quantitative Analysis by SPME/GC works best when: • SPME conditions are kept as consistent as possible (same exposure conditions in vial and same desorption conditions in GC inlet) • An internal standard can help control some variables so that if the transfer of two compounds is consistent, good results will occur Practical Applications of HPLC • Review of Instrument Components Chromatograph Components Chromatographic Column Flow Control Mobile Phase Reservoir Injector Signal to data recorder Detector Waste or fraction collection Chromatogram Practical Applications of HPLC • Mobile Phase Selection – HPLC class will determine main mobile phase requirements: • mostly non-polar solvent for normal phase • polar solvents for reversed phase • water plus ion exchanger for ion chromatography – Other Major Factors • desired retention (adjusting polarity of solvent in HPLC to get desired retention or eluent ionic strength in IC) • need for pH adjustment (to reduce or promote ion formation) Practical Applications of HPLC • Mobile Phase Selection – Cont. – Additional Considerations: • Selectivity (different solvents will have different solvent – analyte interactions; best to choose solvent that emphasizes analyte differences) • Solvent viscosity (low viscosity means smaller back pressure for given flow rate) • Solvent miscibility • Sample solubility • Detector limitations (e.g. wavelength cut-offs for UV detection) • Compatibility with column packing and tubing Practical Applications of HPLC To column • Solvent Selection – Single Pump • Manual Solvent Selection Mixing chamber pump – manual valve selection or replacement of mobile phase – not capable of gradient runs • Low Pressure Mixing – controlled valve for selection of eluents (4 possible with our Agilent system) – fast proportional opening of valves allows good control for gradients – Dual Pump Systems • each pump runs independently • better gradient control and retention time stability vs. low pressure mixing • also smaller dwell time (difference in time between switching valves and effects at columns) gradient starts Pressure dwell time time Effect seen To column pumps Practical Applications of HPLC • Pump Requirements – High pressures • Small packing material diameters require high pressures (up to 300-400 bar) so that one can obtain reasonable flow rates • Newest packing material diameters (sub 5 mm) require even higher pressures or shorter column lengths) – in UPLC – which can go to 1000 bar • Pumps require specific components capable of withstanding such high pressures (e.g. use of sapphire piston rods) – Stable Flow rates • Usually involve feedback control Practical Applications of HPLC • Pump – how they work – Most pumps use two piston heads 180º out of phase to reduce pressure fluctuations – Solvents go into and out of piston heads through oneway “check valves” – Exit check valve closes on “in” stroke and entrance check valve closes on “out” stroke Check valves In Stroke Out Stroke closed open closed open pistons Practical Applications of HPLC • Pump Failures – Common for check valves to fail – This results in a drop in pressure during one of the cycles resulting in an alternating pressure 200 Signal (uV) 100 0 -100 -200 -300 8 8.2 8.4 8.6 Time (min) Bad check valve leaking 8.8 9 9.2 Practical Applications of HPLC • Injection Valves 6 port valve – Fixed loop injector is by far the most common type • small loop volume with manual injection • with autosamplers, it is common to use partial filling (e.g. fill 10 mL of a 250 mL loop) • For large sample injections, the loop can be replaced with a trap – Specialized injectors • used for injecting very small samples in microbore columns • SPME injectors From pump To column Waste sample in LOAD POSITION INJECT POSITION Practical Applications of HPLC • Sample Injection – Injection Volumes • With sample solvent = eluent, volume injected shouldn’t contribute significantly to extra column broadening (rule of thumb injection volume < 0.25(early eluting peak volume)) – Sample Solvent • Ideal solvent is solvent weaker than eluent (or initial eluent in a gradient) • Then can use larger injection volume since analytes will trap on column • Stronger solvents can result in distorted peak shapes or require even smaller injection volumes width = 0.084 min. (or 5.1 s) @ flow = 1.2 mL/min or peak volume = 100 mL, so use vol < 25 mL 6.129 DAD1 D, Sig=230,16 Ref =400,25 (HERRERA\02201303.D) mAU 800 0 1 2 4 5 6 6.748 6.864 7.568 7.134 5.412 4.338 3 4.559 4.666 1.357 200 2.272 1.585 400 5.708 5.839 6.031 5.579 6.382 600 9 8 7 min