PPT - Gmu

Isolation of Caffeine
References:
Slayden: pp. 47- 49
Pavia: pp. 24, 73 – 84
Note: These pages are not in the GMU version of the Pavia text sold in the book, only in the primary text that we do not use anymore .
1/17/2015 1
Isolation of Caffeine
Overview
An aqueous Vivarin/Sodium Carbonate mixture is extracted with Dichloromethane (Methylene
Chloride)
The organic fraction is separated from the aqueous solution and evaporated to dryness
The product is recrystallized from Acetone
The mass and % recovery of the product are determined
Product identity is verified by Melting Point
1/17/2015 2
Isolation of Caffeine
Background
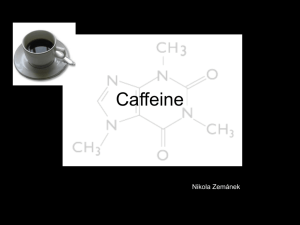
Caffeine (a stimulant) is a bitter, white crystalline alkaloid derivative of Xanthine (note methyl substitution on primary amino groups)
1/17/2015
Caffeine is synthesized from the purine nucleotides AMP, GMP, and IMP in plants
These in turn are transformed into Xanthosine and then Theobromine, the latter being the penultimate precursor of Caffeine
3
1/17/2015
Isolation of Caffeine
The two Amide groups exist as zwitterionic resonance structures where the Nitrogen and
Carbon atoms are double bonded to each other so that both of the Nitrogen atoms are essentially planer (in sp 2 orbital hybridization)
A zwitterion is a neutral molecule with a positive and a negative electrical charge (not dipoles) at different locations within that molecule
Zwitterions are sometimes also called inner salts
The fused ring system therefore contains a total of ten pi ( ) electrons, thus, according to
Huckel’s rule, is aromatic
4
Isolation of Caffeine
Zwitterionic Resonance
1/17/2015 5
Isolation of Caffeine
Chemical Properties
Caffeine acts as a general nervous system stimulant, warding off drowsiness
Caffeine is considered a “psycho active” substance, but is generally legal and unregulated
Caffeine is found in varying quantities in the seeds, leaves, and fruit of some plants, acting as a natural pesticide by paralyzing and killing insects
Sources for human use include coffee plants, tea leaves, and kola nuts
1/17/2015 6
Isolation of Caffeine
Procedure
Obtain 2 Vivarin tablets noting the actual mass of
Caffeine in each tablet
Determine the mass of the tablets
Obtain and determine mass of approximately 8 g of Sodium Carbonate
Crush the tablets with a pestal & mortor
Add the crushed sample to a 150 mL beaker
Add 60 mL of Distilled Water
Add the Sodium Carbonate (Na solution
2
CO
3
) to the
Add a Teflon boiling chip
Heat solution to boiling on a hot plate – maintain boiling point for 2 minutes
1/17/2015 7
Isolation of Caffeine
Procedure (Cont’d)
Allow the hot mixture to settle slightly
- some solid particles will remain undissolved
Attach an iron ring to a ring stand
Insert a 125 ml Separatory Funnel into the iron ring using a clay triangle if necessary to support funnel
Adjust height of iron ring so that stem of separatory funnel fits just inside a
150 ml beaker
Place a glass funnel into the neck of the separatory funnel
Make a boat of glass wool and place in the bottom of glass funnel
1/17/2015 8
Isolation of Caffeine
Procedure (con’t)
Decant the sample solution into the bottom of the glass wool boat in the glass funnel allowing the liquid to drain into the 125 mL Separatory Funnel
Rinse the beaker with a small amount of distilled water and pass the water through the glass funnel into the separatory funnel
Allow the filtrate in the Separatory Funnel to cool to room temperature
1/17/2015 9
Isolation of Caffeine
Procedure (con’t)
Liquid–liquid Extraction, also known as solvent extraction and partitioning, is a method to separate two immiscible liquids, usually an aqueous component and an organic solvent component
The process involves the extraction of a substance from one liquid phase into the other liquid phase utilizing a separatory funnel
Use the following steps to extract the Caffeine from the aqueous phase into the organic phase consisting of Methylene Chloride solvent
(density – 1.33 g/ml)
1/17/2015
Three (3) extractions will be performed
10
Isolation of Caffeine
Procedure (Cont’d)
Add 8.0 mL of Dichloroethane (Methylene
Chloride) to the mixture in the separatory funnel
Place the stopper in the top opening and make sure it is secure, i.e. won’t leak
Invert the funnel pointing the stopcock end away from your face
Use “gentle”, (not vigorous swirling to avoid formation of an emulsion) for 15-20 seconds
Slowly open the stopcock to relieve any pressure
1/17/2015 11
Isolation of Caffeine
Close the stopcock and continuing swirling with venting every 15 seconds until no more gas is released (two or three times should be sufficient)
Continue the swirling for a total of 5 minutes
Replace the separatory funnel in the ring stand and allow the mixture to settle for a few minutes
Remove the stopper from the Separatory Funnel
Open the stopcock slowly and drain the clear bottom (organic) layer into a clean 150 mL beaker
1/17/2015
Repeat the extraction process two more times with new 8.0 mL aliquots of Methylene Chloride, but reduce the total swirling time to 2 minutes
Each time, add the clear bottom layer to the 150 mL beaker
12
Isolation of Caffeine
1/17/2015
Procedure (Cont’d)
With the help of the instructor, add Anhydrous
Sodium Sulfate as a drying agent
Leaving the particles settled on the bottom of the beaker, slowly pour (decant) the liquid into a clean, dry, 50 mL beaker
Rinse the beaker containing the drying agent particles on the bottom with one additional small portion of Dichloromethane and add to the sample beaker
Evaporate the mixture to dryness in the hood
Note: Set hot plate setting to about 3
Note: Crude yield at this point is roughly 90%
13
Isolation of Caffeine
Procedure (Cont’d)
Recrystallization using a mixed solvent of Acetone and
Petroleum Ether
Dissolve the dried product in 12-15 mL cold Acetone
(from ice/water bath)
Heat solution gently on hot plate
If solution is incomplete, remove beaker from hot plate and add more Acetone in 2 mL increments
Up to 20 mL of additional Acetone may be required until a clear pale-yellow solution is obtained
Remove solution from hot plate and allow to cool slightly
1/17/2015 14
Isolation of Caffeine
Procedure (Cont’d)
Add Petroleum Ether, drop by drop, with swirling from a full plastic pipet until the first appearance of pervasive cloudiness throughout the solution
Continue cooling until crystallization is complete
Note: Maximum yield is obtained when the addition of
Petroleum Ether is stopped at the first appearance of a pervasive cloudiness throughout the solution
It may be necessary to evaporate some of the solution to effect recrystallization
Separate the crystals from the solution by Vacuum
Filtration rinsing the beaker and crystals in the funnel with small amounts of Petroleum Ether
1/17/2015 15
1/17/2015
Isolation of Caffeine
Immerse beaker in an ice-water bath for about a minute
Air-dry the sample from the evaporation procedure or the vacuum filtration procedure on a pre-weighed weighing tray for one week
Compute the mass of the purified sample
Compute the percent recovery of the product relative to the amount of caffeine indicated in the tablets
Determine melting point 236 o C
Note: Decomposition of this crude material occurs beginning at 236 o C over a range of 5 o C or so
16