File
advertisement

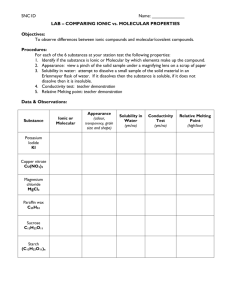
Section 8.1 Matter Pure Substances Elements Mixtures Compounds Molecular Homogenous Ionic Heterogeneous Common Molecules www.reciprocalnet.org/edumodules/commonmolecul es/element/list.html#simple Molecules and Molecular compounds Covalent bond- atoms held together by the sharing of electrons What is the difference between an ionic compound and a molecular compound? Examine this picture of NaCl in H2O Ionic compounds are made of + and – charged ions arranged in repeating 3-D patterns ex: NaCl Molecular compound is made of elements chemically combined ex: H2O Molecular compounds have lower melting and boiling points than ionic compounds Molecular formulas Molecular formula-chemical formula of a molecular compound that shows how many atoms of each element are in a molecule NOTE: this reflects actual # of atoms-not whole number ratio Ex: H2O Subscript (2) after the H indicates there are 2 hydrogen atoms and one oxygen atom How many carbon and oxygen atoms are in a molecule of CO2? Chocolate chip cookie recipe: 2 eggs 1 cup sugar 1 cup brown sugar 2 cups chocolate chips 1 tsp vanilla 2 cups flour If I change the recipe-add peanut butter chips instead of chocolate chips, the cookies are different In a molecular formula, the subscripts tell you the number of atoms of each element in a compound Diagrams and models Ex:Ammonia Molecular formula- NH3 Structural formula- Space filling molecular model Ball and stick molecular model Perspective drawing In plane Coming forward Going backwards Bond going across the back Molecule-a neutral group of atoms joined together by covalent bonds Diatomic molecule- a molecule consisting of two of the same atoms Carbon is black, oxygen is red…. What is the formula of this molecule? What are the molecular formulas for these molecules? TRUE OR FALSE: All compounds contain molecules. FALSE There are no elements that exist as molecules. FALSE Most molecular compounds are composed of two or more nonmetallic elements. TRUE Atoms in molecular compounds share electrons. TRUE Molecular compounds tend to have higher melting and boiling points than ionic compounds FALSE Which of the following formulas represent molecules? Br2 KBr CH4 SO3 N2H2 Ne2 Answer: Br2, CH4, SO3, N2H2 Which of the following formulas represent compounds? Br2 KBr CH4 SO3 N2H2 Ne2 Answer: KBr, CH4, SO3, N2H2