atomic number - Cloudfront.net

Brief Timeline of Atomic Theory
Democritus
• 400BC
• Greek philosopher
Hard Particle (Cannonball)Theory
• Proposed that they world was made up of tiny, indivisible particles moving through a void of empty space
• “atom” comes from the Greek word “atomos”, meaning indivisible (cannot be divided)
John Dalton
• 1808 AD
• First modern atomic theory
Daltons Atomic Theory
1. All matter is composed of tiny, indivisible particles called atoms
2. All atoms of an element are identical
3. Atoms of different elements are all different
4. Atoms combine in simple ratios to form compounds
J.J. Thomson
• 1897-1904
• “Plum Pudding Model”
• Cathode Ray tube experiment
• demo
demo
Cathode Ray Tube
• Thompson showed that cathode rays
(electrons) were composed of negatively
charged particles that separated from the gas atoms inside the tube
• Significant because: this meant that atoms are not hard, indivisible particles. Atoms are composed of smaller “subatomic” particles
Thomson’s Plum Pudding Model
• The atom was a hard sphere that was positively charged with negatively charged electrons that “dotted” the atom like raisins in plum pudding
The discovery of radioactivity
• Henri Becquerel
– 1896
– Discovered that uranium ore released rays that could expose photographic film
The discovery of radioactivity
• Marie & Pierre Curie
– Extracted 2 new elements from uranium (U)ore: radium (Ra) and polonium (Po)
Marie Curie
Ernest Rutherford
•
Magnetic Field Experiment
• Was able to separate radioactive rays into 2 types: alpha ( a
) & beta (
B
)
Determined that a rays were composed of helium nuclei (He +2 charge)
Gold Foil Experiment (1911)
• Lead to discovery of the nucleus, as a positively charged center of atom, containing the mass
• Most of the atom is negatively charged empty space, electrons are outside the nucleus
Magnetic Field Experiment
Gold Foil Experiment
Gold Foil Experiment
Gold Foil Experiment
Gold Foil Experiment
Rutherford’s Atomic Model
Rutherford’s “Nuclear Model”
• Most of the atom is negatively charged empty space, surrounding a small, positively charged nucleus, containing most of the mass of the atom
Modern Theory of Atomic Structure
• Developed by Niels Bohr, based on the science of nuclear physics
• Bohr determined that an element's position on the periodic table was related to its
electron configuration.
Electron configuration
Electron configuration – shows how many electrons are in each energy level or “ring”
• Ex: Carbon 2-4
Bohr’s Planetary Atomic Model
• Niels Bohr (1922)
• Determined that electrons rotate around the nucleus in discrete paths or rings
Planetary Model of Atomic Structure
Wave-Mechanical Model
• Current (modern) theory of atomic structure
• Moseley used x-ray analysis to calculate an integer for each element: these integers are the atomic numbers
Wave-Mechanical Model
• There is a tiny, dense positively charged nucleus at the center of a huge negatively charged electron cloud
Wave-Mechanical Model
Orbital
• Region of probability of finding an electron
“The whole point:”
• The modern model of the atom is the result of many investigations that have been revised over a long period of time by many scientists
• Atomic theory song
Place the models of atomic structure in order from earliest to the modern theory:
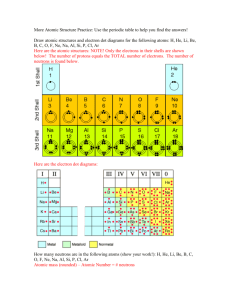
Basic Atomic Structure
• The nucleus occupies less than 0.01% of the total volume of an atom but accounts for
99.97% of its mass. Thus most of an atom is
EMPTY SPACE where the ELECTRONS are found, this is called an ELECTRON CLOUD .
• One atomic mass unit is 1/12 TH THE MASS OF
A CARBON-12 ATOM . This is the standard by which the masses of all other elements are determined. It is abbreviated “u”.
Start here with lecture notes!!
Particle
Subatomic Particles
Symbol Location Charge Mass (amu) Mass (g)
Proton p +
In
Nucleus +1 1 1
Neutron n 0
In nucleus 0
Electron e -
Outside nucleus -1
1
0
1
0
Nucleons:
Def: particles found in the nucleus
: protons & neutrons
Number of Nucleons = Atomic Mass
Nuclear Charge:
Def: equal to the number of protons
(b/c only protons have charge, neutrons are neutral)
For this atom the nuclear charge would be +6, because there are 6 protons
protons protons neutral neutrons protons
6
6
6 electrons
Signature Atomic
Number
Mass
Number
Nuclear charge
# of
PROTONS
# of
NEUTRONS
# OF
ELECTRONS
27
13
Al
35
17
Cl
1
1
H
207
82
Pb
Signature Atomic
Number
Mass
Number
Nuclear charge
# of
PROTONS
13 27 +13 13
27
13
Al
# of
NEUTRONS
14
# OF
ELECTRONS
13
17 35 +17 17 18 17
35
17
Cl
0 1 1 1 +1 1
1
1
H
207
82
Pb
82 207 +87 87 125 87
The only number that never changes for an element is
ATOMIC
NUMBER
!!
24
?
N
Atomic number:___________
# protons: ______________
# electrons: _____________
Mass number: ___________
# neutrons: _____________
Name-mass:_______________________________
64
?
Cu
Atomic number:___________
# protons: ______________
# electrons: _____________
Mass number: ___________
# neutrons: _____________
Name-mass:_______________________________
14
7
N
Atomic number:___________
# protons: ______________
# electrons: _____________
Mass number: ___________
# neutrons: _____________
Name-mass:______________________________
_
31
15
P
Atomic number:___________
# protons: ______________
# electrons: _____________
Mass number: ___________
# neutrons: _____________
Name-mass:_______________________________
27
13
Al
Atomic number:___________
# protons: ______________
# electrons: _____________
Mass number: ___________
# neutrons: _____________
Name-mass:_______________________________
108
47
Ag
Atomic number:___________
# protons: ______________
# electrons: _____________
Mass number: ___________
# neutrons: _____________
Name-mass:_______________________________
Atomic Structure Practice:
Determining Subatomic Particles
1.) Complete the following definitions: a. ATOM b. ATOMIC NUMBER – equals the number of ________ in an atom c. MASS NUMBER – equals the number of ________ + _________ d. Charge of a PROTON - ________ Mass of a PROTON- ________ e. Charge of a NEUTRON - ______ Mass of a NEUTRON -_______ f. Charge of an ELECTRON- _____ Mass of an ELECTRON- ______
WORD BANK: proton, neutron, electron, nucleus, electron cloud
For the atom shown above:
f. Number of Protons= _______________ g. Atomic Number =________________ h. Number of Neutrons= _____________ i. Mass Number = _______________
Using the ATOMIC NUMBER, identify the element name___________ and SYMBOL
_____
Name
Helium-4
Nitrogen-14
Manganese-60
Barium-137
Iodine-131
Iodine-127
64
29
Cu
60
25
Mn
137
56
Ba
131
53
I
127
53
I
Atomic Structure 1
Symbol Charge # of Neutrons # of electrons Atomic
Number
Mass
Number
# of
Protons
19
9
F 0
0
11 23
53
53
25
56
32
60
137
131
127
0
0
0
0
0
0
0
0
0
14
53
53
25
56
14
78
74
35
81
16
25
56
53
53
Name
Helium-4
Nitrogen-14
Atomic Structure 1
Symbol Charge # of Neutrons # of electrons Atomic
Number
Mass
Number
# of
Protons
19
9
F 0
0
11 23
64
29
Cu
25
53
32
131
0
0
0
0
0
0
0
0
0
14
53
14
35
81
74
16
56
Phosphorus-32
Potassium-39
14
6
56
26
C
Fe
24
12
Mg
18
16
40
79 197
0
0
0
0
0
0
0
0
0
29 35
**Shade the columns representing the nucleons light blue
8
Changes in number of subatomic particles
Isotopes
• Change in number of neutrons
• Same atomic number, different mass
• Same number protons, different number neutrons
Ions
• Change in number of electrons
• A ca t ion is positive ion, results from loss of electrons, reducing radius
• An a n ion is negative ion, results from gain of electrons, increasing radius
32
16
S
Atomic number:___________
# protons: ______________
# electrons: _____________
Mass number: ___________
# neutrons: _____________
Name-mass:_______________________________
23
11
?
Atomic number:___________
# protons: ______________
# electrons: _____________
Mass number: ___________
# neutrons: _____________
Name-mass:_______________________________
?
?
Li
Atomic number:___________
# protons: ______________
# electrons: _____________
Mass number: ___________
# neutrons: _____________
Name-mass:_______________________________
?
?
I
Atomic number:___________
# protons: ______________
# electrons: _____________
Mass number: ___________
# neutrons: _____________
Name-mass:_______________________________
ISOTOPE
• Forms of the same element having different mass due to different number of neutrons .
• Indicated by “ element name-mass ”
15
8
O
Name: _______________
16
8
O
Name: _______________
Mass: ________________ Mass: ________________
Protons: ______________ Protons: ______________
Neutrons: _____________ Neutrons: _____________
Practice:
Name Symbol
235 U
Atomic # Mass # # Protons # Neutrons # Electrons
238 U
Carbon-12
Carbon-13
The mass on the periodic table is the
MOST ABUNDANT mass !
** You can estimate which isotope is found in the highest abundance as the one with a mass closest to the mass listed on the periodic table
Example:
Chlorine-35 mass 34.969g
Chlorine-37 mass 36.966g
Look on the periodic table for the mass of chlorine____________________________
The more abundant isotope has a mass closer to the mass given on the periodic table_____________
Practice:
Which isotope of silicon would be found in the highest percentage?
28
14
Si, mass 27.977
30
14
29
14
Si, mass 29.974
Si, mass 28.976
Why?
_______________________________________
_______________________________________
Atomic Structure 2
Isotopic
3.
4.
5.
1.Oxygen-16
2.Oxygen-18
10.
11.
12.
6.
7.
8.
9. Iron-
13.
14.Hydrogen-
15.
16.
Notation
O-16
Ar-40
Ne-20
H-2
16
34
57 Fe
3
O
S
H
Number of protons
Number of neutrons
Number of electrons
Mass number
18
16
19
19
26
10
18
20
32
32
41
22
1
2.) Calculate the weighted average of the following naturally occurring isotopes. SHOW ALL WORK!
a.) 95.50% 7 Li & 7.50% 6 Li d.) 99.63% 14 N & 0.37% 15 N b.)80.20% 11 B & 19.80% 10 B c.)95.02% 32 S, 0.75% 33 S, & 4.21% 34 S e.) 78.9% 24 Mg, 10.00% 25 Mg, & 11.01% 26 Mg f.) 92.23% 28 Si, 4.67% 29 Si, & 3.10% 30 Si
IONS
• A charged part of an atom, resulting from the loss or gain of electrons
• VALENCE electrons: outermost electrons, the last number in an electron configuration
• KERNEL electrons: all electrons except valance electrons
Electron configuration
Electron configuration – shows how many electrons are in each energy level or “ring”
• Ex: Carbon 2-4
Electron configuration of sodium:
2 diagrams of atomic structure :
Bohr diagrams Lewis electron dot diagrams
Bohr realized that the rows on the periodic table corresponded to the number of shells of electrons
Lewis realized that the groups/families on the periodic table correspond to the number of valence electrons
This model shows the nucleus, indicating the number of protons and neutrons, surrounded by rings, representing each energy level
This model shows the element symbol surrounded by dots, representing the valence electrons. You must place one dot at each (3, 6,9,12 o’clock) location before “doubling up” (exception:
Helium)
18
9
F electron configuration 2-7 F electron configuration 2-7
1
1
1
7
1
9
2
11
Bohr Atomic Structures
13
12
14
14
15 tables to fill in the electron configurations, as shown, then draw the Bohr Atomic Structure for each element 1-20.
16
4
16
19
17
2
2
20
18
3
2-1
23
4
2-2
24
5
2-3
27
6
2-4
28
7
31
11
2-8-1
39
12
2-8-2
40
19
2-8-8-1
20
2-8-8-2
13
2-8-3
Rules:
1.) Show placement of ALL electrons
*use atomic #
OR the entire electron configuration
14
2-8-4
2.) The nucleus is represented by a center circle showing the
# of protons & the
# of neutrons
15
3.) Indicate the number of electrons in each energy level, by writing the number on each ring.
** closest to nucleus is 1 st
16
8
32
9
35
17 18
10
40
1
3
1
1
4
2
LEWIS Electron Dot Structures
5
13
6
14
7
15
Directions:use your reference tables to fill in the electron configurations, as shown, then draw the Lewis Dot Structure for each element 1-20.
8
16
9
17
2
18
10
2-1
11
2-2
12
2-3
13
2-4
14 15 16 17 18
2-8-1
19
2-8-8-1
2-8-2
20
2-8-8-2
2-8-3
Rules:
1.) Only show outermost(VALENCE) electrons
*use group #
or the last # in the
electron configuration
2-8-4
2.) Electrons are represent- ex: ed as dots, placed at the
12
12,3,6,9
around the element symbol.
3.) You must place 1 dot(e ) at each location before
you double up.
4.) Exception is row 1: for element #2, indicate both electrons at the 12 o'clock location.
2 Main Types of Ions:
a n ion
A negative ion
Ex: Cl , O -2 ca t ion
A positive ion
Ex: Na + , Al +3
The octet rule
Atoms will gain or lose electrons in order to have a full valence shell of 8 electrons.
Exception: Helium can have a maximum of 2 valance electrons
When an atom gains 1 or more electrons
It becomes a negative ion and it’s radius
increases. A negative ion is an anion.
When an atom loses 1 or more electrons
It becomes a positive ion and it’s radius
decreases. A positive ion is a cation.
Definition
Results from
Indicated by
What happens to radius???
CATION
Na Na +
Naming
Lewis Dot
Structure
ANION
Definition
Results from
Indicated by
What happens to radius???
CATION positive ion
Loss of electron(s)
(+) charge
Gets smaller
Naming
Lewis Dot Structure
Na Na +
“Element name ion”
[Na] +
ANION negative ion
Gain of electron(s)
(-) charge
Gets bigger
Change ending of element to “ ide”
..
[:.F.:] -
How to predict if an element will form an anion or cation:
The “electron clock”:
6
8/0
7
5
1
3
4
# valance electrons
2
Al
N
Mg
O
Fr
C
Atomic Structure 3: Predicting Ions
Radius increase or decrease?
Lewis dot structure of atom
How many electrons lost or gained?
Element
Electron configuration
Lose or gain electrons?
Ionic Charge
**
Lewis dot structure of ion
F 2-7 F gain 1 -1 F increase
2-8-2 Mg lose 2 +2 Mg decrease
Element
Electron configuration
2-8-8-1
Lewis dot structure of atom
Lose or gain electrons?
How many electrons lost or gained?
Ionic Charge
**
Lewis dot structure of ion
Radius increase or decrease?
2-8-7
2-8-18-18-8-2
2-8-6
2-8-5
2-3
**In the “ ionic charge ” column only: shade the cation charges red and the anion charges blue
ex 35
17
Cl
1 23
11
Na
2
3
9
4
Be
65
30
Zn
4
5
6
7
14
7
N
32
16
S
20
10
Ne
127
53
I
8
9
108
47
Ag
70
31
Ga
10 12
6
C
# of
Protons
17
# of
Neutrons
Atomic Structure 4
# of
Electrons
Nuclear
Charge
Bohr Diagram of
Atom
Lewis Dot of Atom
Predict
Ionic Charge
Lewis Dot of Ion
18 17 +17 Cl -1
Name of Ion
Chloride
Atomic Spectra
Radiant Energy
• Energy that travels through space as electromagnetic waves at the speed of light
Electromagnetic Spectrum
• Includes all types of radiant energy from gamma rays (hi E) to radiowaves (lo E)
• Visible light is only a small portion of the spectrum
1 photon = 1 quantum
Quanta: tiny packets of energy released or absorbed by objects
*Einstein and Plank determined that energy is released or absorbed in a continuous flow of small packets or quantum/photons
Release or Absorption of Energy:
Higher energy levels
(excited state)
Electrons release energy when falling to
Electrons absorb energy when jumping to
Lower energy levels
(ground state)
Bohr used the emission spectrum as proof of planetary model
But his model only works for hydrogen because he didn’t account for electrons moving between energy levels
Spectral Lines
Characteristic wavelengths ( l
) of photons of energy released as electrons fall from hi to lo energy
Spectral lines demo:
Salt of
Element
Strontium Chloride
Barium Chloride
Copper (II) Chloride
Lithium Chloride
Potassium Chloride
Color of Flame
Identity
Unknown Element
Unknown Mixture
Emission Spectrum :
Each element has it’s own characteristic spectrum:
Compare H & He:
hydrogen helium
Because electrons do move between energy levels, emitting “spectral lines”, we had to change our view of atomic structure:
Excited State Electron Configurations
Occurs when elements absorb energy and jump to a higher energy level.
** it will not look like it is written on periodic table, be sure they add to the correct number!
Ground state: 2-8-1
Excited state : 2-7-2
“Crib Sheet”
• #p + = atomic number *#n 0 = mass-atomic number
• #e = #p + - charge (use the sign of the charge)
• Isotope : same #p + , different #n o OR same atomic number, different mass
• To calculate weighted average: (%/100 x atomic mass)
+ (%/100 X atomic mass) + …..
• * Ion : same # p + , different #e -
• Charge= #p + - #e -
Atomic Structure Review p. 17
1. 11
2. 9
3. 43
4. 92
5. 118
6. 13
7. 11
8. 4
9. Br
10. C
11. Sn
12. Zn
13. Cl
14. 40
15. 16
16.)
Atomic Structure Review p. 17
17)
=(.925x7) + (.0750x6)
=(.789x24)+(.10x25)+(.1101x26)
=6.475 + .45
= 18.936 + 2.5 + 2.8626
=6.925
= 24.2986
=6.93g
= 24.30g
Atomic Structure Review p. 18
18.) 2-8-1
19.) Na
20.) 2-7-2
21.) 19
22.) 1
23.) Y
24.) Ar
25.) Not possible
27.) as electrons fall from excited state to ground state energy is released as radiant energy
(spectral lines).
28.) you can ID the gas element using spectral line analysis.
29.) electrons are negatively charged particles. B has 5 e , its e config. is 2-3, with 2 e in the 1 st energy level and 3 e in the 2 nd
(valence) level
Atomic Structure Review MC?s
1.) 2
2.) 4
3.) 1
4.) 1
5.) 3
6.) 1
7.) 4
8.) 2
9.) 4
10.) 3
11.) 4
12.) 3
13.) 1
14.) 4
15.) 1
16.) 3
17.) 4
18.) 3
19.) 2
20.) 2
21.) 3 pg 19-20
1.) 4
2.) 3
3.) 2
4.) 3
5.) 2
6.) 3
7.) 1
8.) 2
9.) 3
10.) 1
11.) 3
12.) 1
13.) 2
14.) 4
15.) 3
16.) 3
17.) 2
18.) 1
19.) 1
20.) 4
21.) 4
22.) 2
23.) 3 pg 21-22
Atomic Structure Review p. 23
1.) 19p, 20n, 18e
3.) 5p,6n,2e
5.) 16p,16n,18e
7.) 7p,7n,10e
9.) 37p,48n,36e
11.) 30p,35n,28e
2.) 9p,10n,10e
4.) 15p,16n,18e
6.) 14p,14n,10e
8.) 20p,20n,20e
10.) 53p,75n,54e
12.) 6p,6n,10e