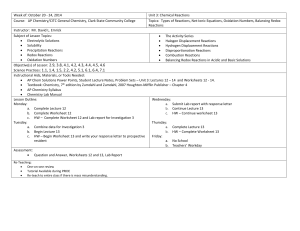
Unit 1 Introduction to Chemistry

Unit 1
Introduction to Chemistry
Outlin e
PowerPoint Presentation adapted from Mr. John Bergmann
Whether you believe you can do a thing or believe you can’t,
Don’t be afraid to take a big step if one is indicated.
You can’t cross a chasm in two small jumps.
All through my life,
Believe that life is worth living, and your belief will help create the fact.
Chemistry is the study of matter and the transformations it can undergo…
…Matter is anything that has mass and occupies space.
Chemistry
with a
Purpose
Interactive Periodic Table
1
2
3
4
5
6
7
H
S
16
P
15
Ar
18
Ta
73
The Human Element
N
7
S
16 H
1
He
2
1
Li Be
3
Na
4
Mg
B
5
Al
C
6
Si
N
7
P
O
8
S
F
9
Cl
Ne
10
Ar
11
K
19
Rb
12
Ca
20
Sr
Sc
21
Y
Ti
22
Zr
V Cr Mn Fe Co Ni Cu Zn
13
Ga
14
Ge
15
As
23
Nb
24
Mo
25
Tc
26
Ru
27
Rh
28
Pd
29
Ag
30
Cd
31
In
32
Sn
33
Sb
16
Se
34
Te
17
Br
35
I
18
Kr
36
Xe
37
Cs
38
Ba
39
*
55
Fr
56
Ra
W
87 88
40
Hf
41
Ta
42
W
43
Re
44
Os
45
Ir
72
Rf
73
Db
74
Sg
75
Bh
76
Hs
77
Mt
104 105 106 107 108 109
46
Pt
47
Au
48
Hg
49
Tl
50
Pb
51
Bi
52
Po
53
At
54
Rn
78 79 80 81 82 83 84 85 86
La Ce Pr Nd Pm Sm Eu Gd Tb Dy Ho Er Tm Yb Lu
57
Ac
89
58
Th
59
Pa
90 91
60
U
92
61
Np
62
Pu
63
Am
64
Cm
65
Bk
66
Cf
67
Es
68
Fm
69
Md
70
No
71
Lr
93 94 95 96 97 98 99 100 101 102 103
Physical Science
Natural Science
Earth and Space Science Life Science
Physics Chemistry Geology Astronomy Botany Zoology
Meteorology Ecology
Oceanography
Natural science covers a very broad range of knowledge.
Wysession, Frank, Yancopoulos, Physical Science Concepts in Action, 2004, page 4
Genetics
Evaluation
Synthesis
Analysis
Application
Comprehension
Knowledge
The Six Levels of Thought
“Success is a journey, not a destination.”
-Ben Sweetland
“Successful students make mistakes, but they don’t quit. They learn from them.”
-Ralph Burns
“Success consist of a series of little daily efforts.”
-Marie McCuillough
Job Skills for the Future
• Evaluate and Analyze
• Think Critically
• Solve Math Problems
• Organize and Use References
• Synthesize Ideas
• Apply Ideas to New Areas
• Be Creative
• Make Decisions with Incomplete Information
• Communicate in Many Modes
Chemistry will develop ALL of these skills in YOU!
Dual
Perceptions
Dual
Perceptions
Stack of Blocks
Perception of Motion
A C o l o r f u l Demonstration: The Remsen Reaction
Click to see
VIDEO
Safety
Basic Safety Rules
#1 Rule: Use common sense.
Others:
No horseplay.
No unauthorized experiments.
Handle chemicals/glassware with respect.
Safety Features of the Lab
safety shower fire blanket fire extinguisher eye wash fume hood circuit breaker switch
Material Safety Data Sheet
(MSDS)
-gives information about a chemical
-lists “Dos” and “Don’ts;” emergency procedures
Chemical Exposure
acute exposure a one-time exposure causes damage e.g., reaction to drugs or medication chronic exposure damage occurs after repeated exposure e.g., smoking, asbestos
Government Regulation of
Chemicals
The government regulates chemicals to reduce the risk to the…
• Consumer
FDA, USDA, Consumer Product Safety Commission
• Worker
OSHA
• Environment
EPA
Chemical Stewardship
Chemical Burns
Flammable
Health Reactive
Special
Chemical burns on feet
.
Skin burned by chemicals
SAFETY in the Science
Classroom
Obey the safety contract
– Use common sense
– No unauthorized experiments
– Wear safety glasses
– Safety is an attitude!
– Don’t take anything out of lab
– Read and follow all instructions
LD
50 the lethal dosage for 50% of the animals on which the chemical is tested
There are various ways an LD
50 can be expressed. For example, acetone has the following LD
50 s:
ORL-RAT LD
50
: 5,800 mg/kg
IHL-RAT LD
50
: 50,100 mg/m 3 -h
SKN-RBT LD
50
: 20 g/kg
Example
Chemical A: LD
50
Chemical B: LD
50
= 3.2 mg/kg
= 48 mg/kg
Which is more toxic?
Chemical A is more toxic because less of it proves fatal to half of a given population.
Science
The Functions of Science
pure science the search for knowledge; facts applied science using knowledge in a practical way e.g., aluminum strong lightweight good conductor
Science attempts to establish cause-effect relationships.
risk-benefit analysis weigh pros and cons before deciding
Because there are many considerations for each case, “50/50 thinking” rarely applies.
How does scientific knowledge advance?
1.
curiosity
2.
good observations
3.
determination
4.
persistence
The Scientific Method
** Key: Be a good observer. observation uses the five senses inference involves a judgment or assumption
Types of Data
Observations are also called data .
qualitative data
-descriptions quantitative data
-measurements e.g., 55 L or 83 o C
Parts of the Scientific Method
Identify an unknown.
Make a hypothesis : a testable prediction
Repeatedly experiment to test hypothesis.
procedure : order of events in experiment (i.e., a recipe) variable : any factor that could influence the result
A Scientific
Experiment
Experiments must be controlled : they must have two set-ups that differ by only one variable conclusion: must be based on the data
Scientific Law vs. Scientific Theory
law:
--
-states what happens does not change never violated
-- e.g., law of gravity, laws of conservation theory: tries to explain why or how something happens
-based on current evidence
-- e.g., Theory of Gravity,
Atomic Theory
Phlogiston Theory of Burning
1. Flammable materials contain phlogiston.
2. During burning, phlogiston is released into the air.
3. Burning stops when…
… object is out of phlogiston, or
… the surrounding air contains too much phlogiston.
(superceded by combustion theory of burning)
Chemistry
The Beginning
early practical chemistry: household goods, weapons, soap, wine, basic medicine
___
D
The Greeks believed there earth were four elements.
D D wind
~ fire water
Alchemy
(~500 – 1300 C.E.) the quest for the Philosopher’s Stone
(the elixir, the Sorcerer’s Stone)
Allegedly, this substance would turn cheap metals into gold.
Alchemical symbols for substances…
.
. .
. . .
. . . .
. . . . .
GOLD SILVER COPPER IRON SAND
transmutation: changing one substance into another
Philosopher’s
Stone
COPPER GOLD
In ordinary chemical reactions… we cannot transmute elements into different elements.
Alchemy was practiced in many regions of the world, including China and the Middle East.
Alchemy arrived in western Europe around the year 500 C.E.
Modern chemistry evolved from alchemy.
Contributions of alchemists:
• experimental techniques
• new glassware
• information about elements
• developed several alloys
What is Chemistry?
the study of matter and its changes
Areas of Chemistry
organic the study of carboncontaining compounds biochemistry the chemistry of living things inorganic studies everything except carbon e.g., compounds containing metals physical measuring physical properties of substances e.g., the melting point of gold
Careers in Chemistry
• research (new products)
• production (quality control)
• development (manufacturing)
• chemical sales
• software engineering
• teaching
The skills you will develop by an earnest study of chemistry will help you in any career field.
The Scope of Chemistry
bulk chemical manufacturing acids, bases, fertilizers
**sulfuric acid (H
2
SO
4
) = #1 chemical petroleum products fuels, oils, greases, asphalt pharmaceuticals
1 in 10,000 new products gets FDA approval synthetic fibers nylon, polyester, rayon , spandex
All fields of endeavor are affected by chemistry.
Government Regulation of Chemicals
The government regulates chemicals to protect the… worker
OSHA environment
EPA
FDA
USDA
FAA
CPSC consumer
Manipulating Numerical Data
Graphs
Bar Graph
shows how many of something are in each category
Chemistry Grades
10
8
6
4
2
0
A B C D F
Pie Graph
shows how a whole is broken into parts
Percentage of
Weekly Income
Entertainment (40%)
Food (25%)
Clothing (20%)
Savings (15%)
Line Graph
shows continuous change
Stock Price over Time
60
50
40
30
20
10
0
Jan Feb Mar Apr
Month
In chemistry… you will always use a line graph.
Elements of a “good” line graph
1.
title
2.
axes labeled, with units
3.
neat
4.
use the available space
3
2
1
0
6
5
4
10
9
8
7
Temp. v. Vol. for a Gas at Constant
Pressure
120 140 160 180 200 220 240
Temp. (K)
Essential Math of Chemistry
Scientific Notation
-- used to express very large or very small numbers, and/or to indicate precision
(i.e., to maintain the correct number of significant figures)
Form: (# from 1 to 9.999) x 10 exponent
800 = 8 x 10 x 10
= 8 x 10 2
2531 = 2.531 x 10 x 10 x 10
= 2.531 x 10 3
10
10
= 1.4 x 10
–3
Put in standard form.
1.87 x 10
–5
= 0.0000187
3.7 x 10 8 = 370,000,000
7.88 x 10 1 = 78.8
2.164 x 10
–2
= 0.02164
Change to scientific notation.
12,340 = 1.234 x 10 4
0.369 = 3.69 x 10 –1
0.008 = 8 x 10
–3
1,000,000,000 = 1 x 10 9
6.02 x 10 23 = 602,000,000,000,000,000,000,000
Using the Exponent Key
EE
EXP
The EE or EXP or E key means “times 10 to the…”
How to type out 6.02 x 10 23 : :
.
.
not…
6 .
or…
6 .
0 2 and not…
6 .
0 2
0 2 x 1 y x
0
2
EE
3
WRONG!
WRONG!
2 3 x 1
TOO MUCH WORK.
0 y x 2 3
Also, know when to hit your ( –) sign.
(before the number, after the number, or either one)
1.2 x 10 5
2.8 x 10 19
Type this calculation in like this:
1 .
2 EE 5
2 .
8 EE 1 9 =
Calculator gives… or…
4.2857143 –15
4.2857143 E–15
This is NOT written… 4.3
–15
But instead is written… 4.3 x 10
–9 or 4.3 E –9
7.5 x 10
–6
( –8.7 x 10 –14
) =
–6.5 x 10 –19
4.35 x 10 6 (1.23 x 10
–3
) = 5.35 x 10 3 or 5350
5.76 x 10
–16
9.86 x 10
–4
= 5.84 x 10
–13
8.8 x 10 11 x 3.3 x 10 11 = 2.9 x 10 23
Essential Math of Chemistry
Units must be carried into the answer, unless they cancel.
5.2 kg (2.9 m)
(18 s)(1.3 s)
= 0.64 kg-m s 2
4.8 kg (23 s)
(18 s)(37 s)
= 0.57 kg s
Solve for x.
x + y = z x and y are connected by addition. Separate them using subtraction. In general, use opposing functions to separate things.
The +y and –y cancel on the left, leaving us with… x + y = z
– y – y x = z – y
Numerical Example
Solve for x.
x – 24 = 13 x and 24 are connected by subtraction. Separate them using the opposite function: addition.
The –24 and +24 cancel on the left, leaving us with… x – 24 = 13
+24 +24 x = 37
Solve for x.
x and k are connected by multiplication. Separate them using the opposite function: division.
F = k x
( )
F = k x
(or)
The two k’s cancel on the right, leaving us with…
F = k x k
k x =
F k
Numerical Example
Solve for x.
x and 7 are connected by multiplication. Separate them using the opposite function: division.
8 = 7 x
( )
8 = 7 x
(or)
The two 7’s cancel on the right, leaving us with…
8 = 7 x
7
7 x =
8
7
Solve for x.
BA = TR x
___
H
One way to solve this is to cross-multiply.
Then, divide both sides by TR.
BAH = xTR
( )
BAH = xTR
The answer is… x =
TR
Solve for T
2
, where…
P
1
= 1.08 atm
P
2
= 0.86 atm
V
1
= 3.22 L
V
2
= 1.43 L
T
1
= 373 K
T
2
=
T
1
=
T
2
P
1
V
1
T
2
=
P
2
V
2
T
1
T
2
=
P
1
V
1
=
132 K
(1.08 atm)(3.22 L)
SI Prefixes
kilo(k) 1000 deci(d) 1 /
10 centi(c) 1 /
100 milli(m) 1 /
1000
Also,
1 mL = 1 cm 3 and 1 L = 1 dm 3
Conversion Factors and
Unit Cancellation
How many cm are in 1.32 meters?
equality: 1 m = 100 cm conversion factors:
(or 0.01 m = 1 cm) or
100 cm
1.32 m
( )
1 m
1 m
= 132 cm
We use the idea of unit cancellation to decide upon which one of the two conversion factors we choose.
How many m is 8.72 cm?
equality: 1 m = 100 cm conversion factors: or
100 cm
8.72 cm
( )
100 cm
1 m
= 0.0872 m
Again, the units must cancel.
How many kilometers is
15,000 decimeters?
15,000 dm
( )
10 dm
( )
1,000 m
= 1.5 km
How many seconds is 4.38 days?
4.38 d
( )
1 d
( )
1 h
( )
1 min
= 378,432 s
If we are accounting for significant figures, we would change this to… 3.78 x 10 5 s
Simple Math with
Conversion Factors
Find area of rectangle.
A = L .
W 4.6 cm
= (4.6 cm)(9.1 cm)
9.1 cm
= 42 cm
2
. cm cm .
cm
Convert to m 2 . 42 cm 2
( )
100 cm
2
= 0.0042 m 2
Convert to mm 2 . 42 cm 2
( )
1 cm
2
= 4200 mm 2
For the rectangular solid:
Length = 14.2 cm
Width = 8.6 cm
Height = 21.5 cm
Find volume.
V = L .
W .
H
= (14.2 cm)(8.6 cm)(21.5 cm)
= 2600 cm 3
Convert to mm 3 .
2600 cm 3
( )
1 cm
3
= 2,600,000 mm 3
= 2.6 x 10 6 mm 3 mm and cm differ by a factor of……….
10 mm 2 “ cm 2 “ “ “ “ “ ……….
100 mm 3 “ cm 3 “ “ “ “ “ ……….
1000
Basic Concepts in Chemistry
chemical: any substance that takes part in, or occurs as a result of, a chemical reaction
All matter can be considered to be chemicals or mixtures of chemicals.
chemical reaction: a rearrangement of atoms such that…
“what you end up with” products differs from
“what you started with” reactants
methane + oxygen carbon dioxide
+ water
CH
4
(g) + 2 O
2
(g) CO
2
(g) + 2 H
2
O(g)
sodium + water hydrogen + sodium hydroxide
2 Na(s) + 2 H
2
O(l) H
2
(g) + 2 NaOH(aq)
Law of Conservation of Mass total mass total mass
= of products of reactants
P mass
= R mass synthesis: taking small molecules and putting them together, usually in many steps, to make something more complex
JENNY
How many feet is 39.37 inches?
equality: 1 ft = 12 in applicable conversion factors: or
12 in
X ft = 39.37 in
( )
12 in
=
1 ft
3.28 ft
Again, the units must cancel.
Resources - Intro. to Chemistry
Worksheet - vocabulary
Worksheet - material safety data sheet (acetone)
Activity - checkbook activity
Worksheet - graphing
Worksheet - real life chemistry
Worksheet - conversion factors
Worksheet - scientific notation
Worksheet - metric article (questions)
Worksheet - significant digits
Worksheet - math review
Worksheet - math of chemistry
Worksheet - article on the metric system
Lab – introduction to qualitative analysis
Episode 1 - The World of Chemistry
Episode 3 – Measurement:
The Foundation of Chemistry
Episode 4 - Modeling The Unseen
Textbook - questions
Outline ( general )