Chapter 11 review Unsolved
advertisement

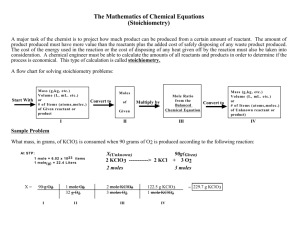
Chapter 11 Review Define the following: • • • • • • • Stoichiometry Mole ratio Excess reactant Limiting reactant Theoretical yield Actual yield Percent yield Find the following molar masses: • • • • • • O2 O AlPO4 NaCl C6H5Cl CuO Determine the number of steps for the following problems (1, 2, or 3): Given the following equation: 2 KClO3 –> 2 KCl + 3 O2 • How many moles of O2 can be produced by letting 12.00 moles of KClO3 react? • How many grams of O2 can be produced by letting 34.7g of KClO3 react? • How many moles of O2 can be produced by letting 14.6g of KClO3 react? Create the following mole ratios: • __Ag(s) + __H2S(g) + __O2(g) __Ag2S(s) + __H2O(l) (Equation must first be balanced.) • Ag : H2S • O2 : Ag2S • • • • Ag2S : H2O O 2 : H 2S Ag : O2 H2O : H2S How many ratios can this equation form? What is the molar ratio of all elements/compounds shown? Solve the following: Given the following equation: 2 KClO3 –> 2 KCl + 3 O2 • How many moles of O2 can be produced by letting 12.00 moles of KClO3 react? Solve the following: Given the following equation: 2 KClO3 –> 2 KCl + 3 O2 • How many grams of O2 can be produced by letting 34.7g of KClO3 react? Solve the following: Given the following equation: 2 KClO3 –> 2 KCl + 3 O2 • How many moles of O2 can be produced by letting 14.6g of KClO3 react? Solve the following: Given the following equation: Al2(SO3)3 + 6 NaOH 3 Na2SO3 + 2 Al(OH)3 • If 10.0 g of Al2(SO3)3 is reacted with 10.0 g of NaOH, determine the limiting reactant Solve the following: Given the following equation: 2 FePO4 + 3 Na2SO4 1 Fe2(SO4)3 + 2 Na3PO4 • What is the percent yield of this reaction if takes place with 25g of FePO4 and an excess of Na2SO4, and produces 18.5g of Fe2(SO4)3