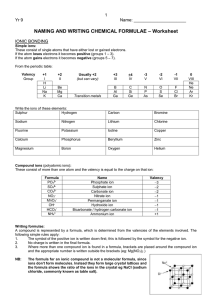
Study your notes! Quiz Ch.8
•
1.
2.
3.
•
4.
5.
Write the formula and name the
following:
Ca + Se
Sr + Br
Al + P
Write the formula from the name:
Chromium(III)oxide
Cesium iodide
Naming Ternary Ionic
Compounds
Na + NO3 NaNO3
Sodium nitrate
How? (use the handout!)
Polyatomic Ions
(many atom ions)
• Polyatomic ions are covalently bonded atoms
with a collective charge.
• EX:
H
..
N H + H+ H
H+
N H
H
NH4+ is the ammonium ion ( +1 charge)
Review the List of Polyatomic Ions
Treat a Polyatomic ion as a Single ion;
don’t break it up !!
You know some of these ions by name.
Nitrates (NO3-)
Nitrites (NO2-)
Phosphates ( PO4-3)
Writing Formulas w/Polyatomics
Li + CO3-2 Li2CO3
Mg + PO4-3 Mg3(PO4)2
NH4 + Cl NH4Cl
NAMES!!
Lithium carbonate
Magnesium phosphate
Ammonium chloride
The Rules for Ternaries
(very easy!!)
Write the correct formula
Write the name of the Cation first
Write the full name for the Anion second
That’s it !!
Al + SO4-2
Al+3 + SO4-2
Al2(SO4)3 * use parens ( ) if you need more than 1 ion!
Aluminum sulfate
You have 5 min to review notes
from last class.
Write the formulas for :
1. Ca + N
2. Al + SO3
-2
3. Mg + phosphate
Write the names for the above.
Write the formula for:
1. Magnesium carbonate 2. Nickel(II)sulfate
Naming Covalent Compounds
( really easy)
CO2
CO
carbon
carbon
dioxide
monoxide
P2O4
diphosphorous
tetraoxide
SO3
sulfur
trioxide
Covalent Compounds consist of two non-metals!!
There are no charges to balance because no
electrons are lost or gained.
So, how do you get the ratios?
Know the prefixes
Prefixes: only for Covalent Cmpds.
Non-metal, Non-metal combinations
Mono
Di
Tri
Tetra
Penta
1
2
3
4
5
Hexa
Hepta
Octa
Nona
Deca
6
7
8
9
10
Try These !!
Name the following:
PO4
NO2
NO3
N2O3
P2O5
SiO2
Write the formula:
Sulfur trioxide
Boron dioxide
Dinitrogen
pentoxide
Carbon monoxide
Carbon
tetrachloride
Handout !!
 0
0