Chem B Packet 8
advertisement

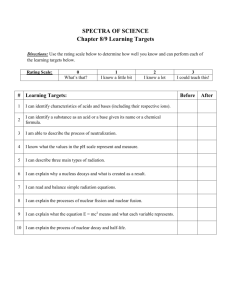
Nuclear Chemistry ALPHA-Chemistry I-B Chapter 28 Wilhelm Roentgen • Wilhelm Roentgen • Discovered X-rays • Was awarded a Nobel Prize for the discovery of the mysterious rays Henri Becquerel • Henri Becquerel was a part of the group that made a the early discoveries of radiation. Marie Curie • Marie Curie and her husband made the discovery of radiation. • She was awarded two Nobel Prizes for her work. The Discovery of Radiation • Henri Becquerel (1895) discovered that pitchblend (Uranium ore) would expose film. He called it radioactivity. • Marie and Pierre Curie worked long difficult hours and discovered new elements-Radium and Polonium Nuclear vs. Ordinary Reactions • Ordinary chemical changes (the ones that display our indicators of chemical reactions) only involve electrons. • Nuclear changes involve the nucleus and create new atoms. – Larger atoms are from fusion – Smaller atoms are from fission Radiation • Radioactivity or Radiation is the spontaneous emission of alpha, beta, or gamma rays the disintegration of the nuclei of atoms. Types of Radiation • Alpha particles – A helium atom doesn’t have any electrons; positively charged. • Beta Particles – A very fast moving electron – Negatively charged • Gamma Rays – A form of energy lots of energy that moves in waves – The rays go through a lot of materials. Going for the Block • Alpha particles can be blocked by a piece of paper; however, once inside the body the most immediate damage. • Beta particles can be stopped with a piece of Al foil. They do more damage over a great area of the body. • Gamma rays penetrate most things and do the most damage to DNA. Radiation Protection 1. Distance 2. Time 3. Shielding Nuclear Protective Gear Nuclear Equations-Know your particles and all is easy 4 • Alpha particles -changes • Alpha Particle 2 the mass number by four and the atomic number 0 by two. It’s a Helium • Beta Particle 1 nucleus • Beta Particles -changes 1 the atomic number by • Neutron 0 one. No change to the mass number. • Neutron -no change to the mass number, but the mass changes by one. These are the main ones !!!!!! He e n The other particles • Proton • Positron 1 1 H 0 1 e Five types of nuclear change 1. 2. 3. 4. 5. Alpha Decay Beta Decay Gamma Decay Fission (spitting) Fusion (joining) Nuclear Equations • Both sides of the equation must be equal in both mass number and atomic number! • It’s just very easy algebra! Examples: 10 5 B n He ? 39 19 1 0 K n 1 0 4 2 36 17 Cl ? Word Problem Example • Thorium-225 undergoes alpha decay. Write a balanced equation for the reaction. Decay Series • A continuous flow of nuclear decay. Fission reaction • Nuclear fission is when nucleus is split into two smaller nuclei. Fusion • Fusion is when two nuclei join to form a larger nucleus. The same reaction occurs in the sun! A Fission Reactor • Cannot explode like a nuclear bomb. Uses a different form of uranium-238 • There is actually less pollution that goes into the air. • Nuclear weapons use uranium-235 Half-Life • Half life is defined as the time it takes for one half the mass of the radioactive isotope to become stable (non-radioactive) • Substances with short half-lives can cause more health hazards than isotopes with long half-lives (radiation is given off much more slowly.) • We are going to view an animation: • http://library.thinkquest.org/27948/decay.html Half-Life Terms • The portion that remains radioactive is called the parent • The decayed portion is called the daughter. • The daughter is not usually radioactive, but it can be. Examples • Carbon-14 has a half-life of 5700 years. How much remains of a 600 gram sample after 17,100 years? • Polonium-210 has a half-life of 138.4 days. How much remains of a 1000 gram sample after 98.86 weeks. Calculating half-life • • The fraction of the original amount that remains is still radioactive. The easy way: 1. Determine the number of half-lives 2. Divide the mass by 2 until you have divided as many times as you have number of half-lives – The Formula: 1 2 n ( mass of substance) amt. left Carbon Dating Carbon-14 and Nitrogen-14 are used to date items based on the half-life of carbon-14. We know it takes 5730 years to decay carbon-14. The amount present in the atmosphere is constant. So you just measure what’s in the artifact and work backwards. Measuring Radiation-read only • Rad=Radiation Dose • Rankin=rad/hour • REM=roentgen equivalent man – 1/1000 rem = 1 mrem • LET=Linear energy transfer • A rad is the amount of energy that is released in tissue when it is irradiated. One rad=100 ergs (energy uints) deposited in one gram of tissue. REM-read only • A REM measures the biological effect of radiation on the human body without having to worry about the type of radiation involved. • What’s allowed? 5-10 rems per year. It’s very rare When radiation hits a cell-read only • It may pass through the cell w/o damage. • It may damage the cell, but the cell fixes the damage. • It may damage the cell; the cell reproduces the damaged form. • The cell dies-serious problems occur if too many cells are killed in a certain organ. Guidelines-read only • From WWII Studies: – 650 rads – 300 rads – 50-250 rads Death within a few hours Lethal-death in 60 days Radiation sickness • Symptoms of radiation sickness – Nausea, vomiting, diarrhea, hair loss, sore throat, reduction in blood platelets, bone marrow damage – Delayed effects: Cancer, leukemia, cataracts, sterility, decreased lifespan