Chapter 10 - People Server at UNCW
advertisement

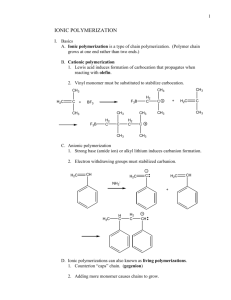
CHM 585 / 490 Chapter 10 • • • • • • PVC PP Polyurethanes Phenolic resins PS ABS 1 PVC • • • • • • Second largest (after polyethylene) Globally 25 million tonnes 14B pounds in U.S. Very versatile Strength, transparency, low cost ½ of the polymer is not based upon petrochemical feedstock Cl ( H H C C H Cl ) n 2 PVC Processes • All from free radical reactions • Most common is batchwise aqueous suspension process – Up to 50,000 gallons of monomer agitated as a suspension in water, centrifuged and dried – High bulk density porous particles obtained 3 •Vinylchloride monomer is dispersed in water by agitation. Polymerization starts by adding monomer-soluble initiators and addition of suspension stabilizers and suspending agents minimizes coalescence of the grains. •The reaction temperature is used for the control of the Mw and varies between 45 - 75 °C. Reactor pressure is between 800 1200 kPa. •The remaining monomer is stripped from the polymer with steam. The waste water is separated in a centrifuge. The PVC resin is dried with hot air and stored. 4 •Emulsion polymerization; batch, semi-continuous or continuous. Vinylchloride monomer is emulsified in water by means of surface-active agents. The monomer is thus present as droplets and a small fraction is dissolved in micelles. Water-soluble initiator is added and polymerization starts in the micelles. 5 Micelles • Are spherical aggregates in a dispersion • Are formed when molecules that possess a hydrophobic region and a hydrophilic region are placed in water • Are responsible for the cleansing action of soaps and detergents • CH3(CH2)16CO2-Na+ 6 Emulsion Polymerization of PVC •Monomer is added to the latex particles (=micelles) by diffusion from the emulsion droplets through the aqueous phase. Batch: all components in reactor; polymerization is stopped when the yield is reached. Semi-continuous: emulsifier is continuously added during the polymerization. Continuous: water, initiator, monomer and emulsifier are added at the top of the reactor. The PVC latex is removed at the bottom of the reactor. The latex is degasified and dried; the resulting solid PVC is stored. 7 Plasticizers • About 2 billion pounds of plasticizers used in U.S. • About 90% for PVC • Most are esters – Commonly esters of phthalic anhydride, adipic acid, phosphoric acid O O O O 8 PP • Used to make things like dishwasher-safe food containers. It can do this because it doesn't melt below 160 oC. Polyethylene will melt at around 100 oC, which means that polyethylene dishes will warp in the dishwasher. • As a fiber, polypropylene is used to make indooroutdoor carpeting, the kind that you always find around swimming pools and miniature golf courses. It works well for outdoor carpet because it is easy to make colored polypropylene, and because polypropylene doesn't absorb water, like nylon does. 9 Polypropylene • • • • Ziegler-Natta Typical system: TiCl4 / MgCl2 / Et3Al 90 – 97% isotactic PP Remainder is atactic and is removed in a separate step • North America PP consumption = 17B pounds / year 10 Stereoregular Polypropylene Isotactic polypropylene Syndiotactic polypropylene Atactic Polypropylene 11 Polyolefin Evolution M(CH3)2 Kaminsky and Sinn- late70s Atactic PP 12 Polyolefin Evolution 1985 Ewen at Exxon Isotactic PP M(CH3)2 13 Polyolefin Evolution M(CH3)2 Ewen 1988 Syndiotactic PP 14 Today • Estimated that > $ 4 Billion spent to date on polypropylene research • Many exciting developments in metallocene catalysis - Changing views about the need for cyclopentadienyl type ligands and extension beyond Group IV metals. • Very active research area! 15 16 17 18 Polyurethanes 19 •The urethane linkage looks like this: (looks like ester) •A wide variety of molecular sections which may be inserted between urethane links provides highly diversified polyurethane properties. 20 21 Here are some commonly used isocyanates: Isocyanate Abbreviation Structure Toluene 2,4-diisocyanate TDI Toluene 2,6 diisocyanate TDI Naphthalene diisocyanate NDI Diphenyl methane diisocyanate MDI Hexamethylene diisocyanate HDI 22 Polyurethane uses • • • • • • • Upholstered furniture Mattresses Automobiles Footwear Paints Adhesives Spandex 23 24 Industry sources peg DuPont's annual sales of Lycra at more than $1.5 billion. DuPont's has the lion's share of world spandex capacity at about 200 million lb annually. In the U.S., those other producers include Fall River, Mass.based Globe Manufacturing, maker of Glospan and Cleerspan spandex, and Bayer Corp. --the U.S. affiliate of Germany's Bayer--maker of Dorlasten brand spandex. Bayer also makes spandex in Germany. Other world producers include Japan's Asahi Chemical Industry and Toyobo, and South Korea's Tae Kwang Industrial Co. 25 Spandex Type 26 27 Phenolic Resins Phenol-formaldehyde ("phenolic") resins are the oldest completely synthetic polymer. (1907 Leo H. Baekeland patents). These thermoset polymers exhibit: •Excellent thermal stability •High char yield •Low smoke generation •Low smoke toxicity 28 Most phenolic resins are made using phenol (P) and formaldehyde (F). They are divided into two classes: •resoles (alkaline conditions...F > P) •novolac (acidic conditions ....F < P) 29 30 Cure of resole prepolymer proceeds under neutral or acidic conditions and at elevated temperature. •Crosslinking occurs via the continued formation of methylene links and the formation of dibenzyl ether linkages. •Higher temperatures favor the formation of methylene bridges •Both are condensation reactions and produce water 31 • About 150 million pounds sold in North America • Ideal for applications where physical properties and structural integrity must be maintained at elevated temperatures • Automotive transmission spacers and brake pads, kitchen range temperature controls, rocket nozzles and heat shields 32 Polystyrene • Free radical polymerization – Preferred industrial route – atactic • Anionic polymerization – atactic • Cationic Polymerization – atactic • Organometallic polymerization – Ziegler-Natta / metallocene – Syndiotactic or isotactic 33 Radical Polymerization R . R . R. + R . ( H2C Atactic CH ) n 34 Anionic Polymerization R - CH3CH2CH2CH2- Li+ + R - ( H2C CH ) n 35 Cationic + + BF3or AlCl3 trace water + ( H2C CH ) n 36 Organometallic • Ziegler-Natta (TiCl3 / triethylaluminum) – Isotactic • Melting point of 240C • Slowly crystallizes • Opaque • Metallocene – Syndiotactic • Melting point 270C • Crystallizes rapidly • Opaque 37 GPPS • • • • General purpose polystyrene Clear, amorphous polymer Lower density than PC Noncrystalline so less mold shrinkage than polyethylene, polypropylene, or PET • Relatively high modulus 38 HIPS • High Impact Polystyrene • Styrene polymerized in the presence of rubber ( polybutadiene) particles to give polystyrene with rubber dispersed throughout the polymer 39 • Some styrene grafts onto butadiene polymer and compatibilizes the blend 40 41 ABS • Terpolymer of acrylonitrile / butadiene / styrene CN A B S 42 What do they bring to the party? • Acrylonitrile – Heat stability – Chemical resistance • Butadiene – Toughness – Impact resistance • Styrene – Stiffness – Processability 43