water F.c college
advertisement

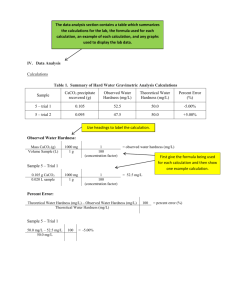
PCSIR, Labs Lahore Provide an overview of the types of chemical and microbial contamination of drinking water and their remedies Over two thirds of the earth's surface is covered with water 97.2% of which is contained in the five oceans. only less than 2.7% of global water is freshwater. Only less than 0.7% is available for human use. Surface Waters 1. Rain Water 2. River Water 3. Lake Water 4. Sea water Underground Water 1. Spring 2. Well water Over the last century Human population has increased 3 times Global water withdrawal has increased 7 times % of total water used 80 70 70% 60 50 40 30 22% 20 8% 10 0 Agriculture Domestic Water uses Industry Glacieres--- hamalayan Mountains (Chenab,Indus, Jehlum) Drinking water to Urban areas (Tehsil & District managemnt bodies—WASA & PHED) Ruler areas ---river, canal, sub soil,pond, rainwater) Agriculture 93% Industry 5% Domestic 2% A person needs about 1 gallon water for hydration Usage in Pakistan / person/day 6.0 gallon Over 1.1 billion people lack access to safe water supply, while 2.6 billion people lack adequate sanitation. Water associated to infectious disease claim upto 3.2 million lives each year, approx. 6% all deaths globally. Rs. 14 billion is spent on the water borne disease (world Bank) 33 % death are due to typhoid, cholera, Diarrhea, dysentery, and hepatitis (UNICEF) Pakistan rank 2nd among 31 countries in annual diarrhea episode o f children (WHO) 80% of all illness in developing countries is caused by water related diseases. 90% of wastewater in developing countries is discharged directly into rivers and streams without treatment. Poor health status of Pakistan----infant mortality rate 12.6% Physical. Suspended solid (SS), color, taste, smell, temp. Chemical. Dissolved substances, alkalinity, hardness, fluoride, heavy metal, organic compound, nutrient (nitrogen & phosphorus), pH, biochemical oxygen demand (BOD), chemical oxygen demand (COD). Biology. Bacteria, virus, protozoa, helminthes There is no pure water in nature, as it can contain up to 90 possible unacceptable contaminants Free from pathogenic organisms Clear Not saline Free from offensive taste or smell Free from compounds that may have adverse effect on human health Free from chemicals that cause corrosion of water supply systems Color may be due to the Presence of organic matter, metals (iron, manganese) or highly colored industrial waste Desirable that drinking water be colorless Taste & odor are mainly due to organic substances, ,Biological activity, industrial pollution Taste buds in the oral cavity specially detect inorganic compounds of metals like magnesium, calcium, sodium, copper, iron and zinc Water should be free from objectionable taste and odour. Pure water---colourless Iron salts ---afetr oxidation ---ferric hydrooxide—red brown colour Off flavour/off ordour -----decomposition of organic matter by non pathogenic bacteria Sulphate --- sulphide by the action of reducing bacteria Wells---proteolytic organisms Turbidity is caused by suspended matter High level turbidity shield and protect bacteria from the action of disinfecting agents Desirable pH limit 6.5-8.5 Beyond this limit the water will affect the mucous membrane and water supply system Acidic Industries Sugar - 5–6 Distillery Electro-Plating unit 3- 4 2.5-4 Pickle 2- 3 Basic Industries Paper 8 – 10 Textile 8.5-11 Fertiliser 6.5- 9 Oil Refineries 6.5-9.5 Capacity of water for reducing and destroying the lather of soap It is total concentration of calcium and magnesium ions Temporary hardness – Bicarbonates of Calcium and Magnesium Permanent hardness – Sulphates, chlorides and nitrates of calcium and magnesium Surface water is softer than ground water Causes encrustations in water supply structures Capacity to neutralize acid Presence of carbonates, bi-carbonates and hydroxide compounds of Ca, Mg, Na and K Alkalinity = hardness, Ca and Mg salts Alkalinity > hardness - presence of basic salts, Na, K along with Ca and Mg Alkalinity < hardness – neutral salts of Ca & Mg present One of the earth’s most plentiful resource High iron causes brown or yellow staining of laundry, household fixtures Metallic taste, offensive odour, poor tasting coffee Cause iron bacteria Acceptable limit – 0.3 mg / l Dissolution of salt deposit Discharge of effluents Intrusion of sea water Not harmful to human beings Regarding irrigation – most troublesome anion Acceptable limit - 250 mg/l Increasing level of nitrate is due to Agricultural fertilizers, manure,animal dung, nitrogenous material ,sewage pollution (blue baby diseases to infants) Maximum permissible limit 45 mg / l Occurs naturally Long term consumption above permissible level can cause – dental flurosis (molting of teeth) Skeletal flurosis Acceptable limit – 1 mg / l Remedy – 1) Deflouridation 2) Mixing Fluride free wate 3) Intake of vitamin C,D, calcium,antioxidants Non degradable and are toxic to human health Common heavy metals are Lead, Mercury, cadmium, arsenic Produced by industrial activities and are slowly deposited in surrounding water and soil Transported by food, water ,air, absorption, adsorption on materials Heavy Metals Health effects Cadmium Lung, liver ,kidney damage Lead Lung, liver ,kidney damage Arsenic Lung, liver ,kidney damage Mercury (inorganic) Lung, liver ,kidney damage Zinc Skin irritation Copper liver ,kidney damage Insecticides – kill insects Herbicides – kill weeds Fungicides – suppress or kill fungi Effects Cancer Birth defects Blood disorder Nervous disorder Genetic damage A Types of water systems that serve as breeding grounds for potentially pathogenic species: Surface Water Ground Water Pipes and storage tanks Waterborne pathogens are present in large numbers Largely due to problems with inadequately maintained pipes, low pressure, poor hygiene standards and intermittent delivery, even municipal well water is unsafe E.coli Salmonella typhimourium Legionella neumpphili Pseudomonos Shigella Vibrio cholera WATER BORN VIRAL DISEASES VIRUS HEPATITIS- A ROTA VIRUS POLIO VIRUS DISEASE VIRAL HEPATITIS INFANTILE DIARRHEA POLIO Protozoa Entamoeba histolytica - amoebic dysentery Giardia lamblia - intestinal disease Cryptosporidium - intestinal disease When sources of water are Rivers,Streams,Lakes etc. then water is Purified by Storage Sedimentation Filtration (rapid sand & slow sand) Disinfection (chlorination) When sources of water are wells,Springs,Tanks etc.Then water is purified by the addition of Bleaching powder/Chlorinated Lime as it is Cheep Easy to use Reliable and safe Water filters( R.O, UV ) BOILING: for 5-10 minutes kills almost all organisms & removes temporary Hardness Bleaching Powder: 5% solution is used Dose: 3-6 drops/L contact time of ½ hour. Chlorine Tablets/Halazone Tablets. one tablet/litre. Iodine Solution: 02 drops of 2% Soln./litre KMnO4: an amount that gives just pink coloration to the Water. Alum: used for turbid water in a dose of 0.1-0.4 grains/5 litres of Water. Boiling: for 5-10 minutes. High Test Hypo chlorite: used in granular form Dose: 01gm./10 litres of water. Liquid Bleach/5% Sodium Hypo chlorite solution: Dose: 14 ml./1000 litres of water. Bleaching Powder: used in a dose of 2.3gm./1000 litres of water. CHEMICAL METHODS / GERMICIDES CHLORINE: is used for purification of water on large scale KMnO4: used for disinfection of water in wells especially during epidemics CuSO4/CaCl2: used for the removal of algae Lime: used for muddy water Ozonization & U.V. irradiation: both methods are used for disinfection of water in swimming pools Silver: sand coated with silver used in slow sand filters instead of plain sand Charcoal: used as adsorbent to remove color,odor & taste Killing of pathogenic bacteria by inhibiting their protein synthesis. Oxidizes Iron, Manganese & Hydrogen Sulphide. Destroys taste & odor producing constituents. It controls algae growth. It helps in coagulation and suspension of the organic matter. Efficiency of chlorine increases, when it combines with Ammonia It decreases, when it combines with Sulphides, Ferrous salts & inert matter in suspension form Advantages Effective and Economical Stable and does not deteriorate on storage No impurities such as salts are introduced. Can be used at high as well as low temp. Disadvantage THM(Trihalomethanes) are carcinogenic Choroamine disinfection reduce THMs production due to preferential reaction of chlorine with ammonia Sr. # Parameter (A.P.H.A., 2005) PS:46392004(R) 1 pH at 25oC 2 Total Alkalinity as CaCO3 (meq/L) 3 Carbonates (mg/L) 4 Bicarbonates (mg/L) 5 WHO Limits 2008 6.5 – 8.5 6.5 – 8.5 --- ---- Total Hardness as CaCO3 (mg/L) 250 500 6 Calcium Hardness as CaCO3 (mg/L) --- --- 7 Magnesium Hardness as CaCO3 (mg/L) --- 8 Total suspended solids (mg/L) Nil Nil 9 Total dissolved solids (mg/L) 500 1000 10 Calcium as Ca2+ (mg/L) 100 ---- 11 Magnesium as Mg2+ (mg/L) 50 Sr. # Parameter (A.P.H.A., 2005) PS:46392004(R) WHO Limits 2008 12 Potassium as K+ 10 ---- 13 Sodium as Na+ 50 200 14 Chloride as Cl- 250 250 15 Sulphate as SO42- 250 250 16 Conductivity at 25oC (µS/cm) --- --- 17 Nitrate as NO3- (mg/L) 10.0 50 18 Nitrite NO2- (mg/L) 1.0 0.5 19 Fluoride F- (mg/L) 0.7 1.5 20 Arsenic (µg/L) 10.0 10.0 Drinking Water No coliforms contamination acceptable Recreational water 200 fecal coliforms /100 ml Fish and wildlife habitat 5000 fecal coliforms/100 ml Shellfish 14 fecal coliforms/100 ml Microbiology Heavy Metals Physical Testing Pesticide residue Water/Drinks testing (APHA) Enumeration of Heterotrophic Plate Count Enumeration of Total Coliforms Enumeration of Fecal coliforms Enumeration of E.coli Enumeration of Pseudomonas spp. Ion Chromatogram HPLC ELISA PCR Atomic absorption GC ICP Spectrophotometer Flame photometer LCMS Fertilize your garden and yard plants with manure or compost instead of commercial inorganic fertilizer. Minimize your use of pesticides. Never apply fertilizer or pesticides near a body of water. Grow or buy organic foods. Compost your food wastes. Do not use water fresheners in toilets. Do not flush unwanted medicines down the toilet. Do not pour pesticides, paints, solvents, oil, antifreeze, or other products containing harmful chemicals down the drain or onto the ground. Prevent groundwater contamination Greatly reduce nonpoint runoff Reuse treated wastewater for irrigation Find substitutes for toxic pollutants Work with nature to treat sewage Practice four R's of resource use (refuse, reduce, recycle, reuse) Reduce resource waste Reduce air pollution Reduce poverty Reduce birth rates Establishment ground water regulatory framework. Proper disposal& sanitary inspection of waste water Mapping of ground water quality Monitoring of water quality at District level Up gradation of analytical labs.& equipments Strengthening of R&D institutions to develop appropriate water purification techniques.