Progress Report
advertisement

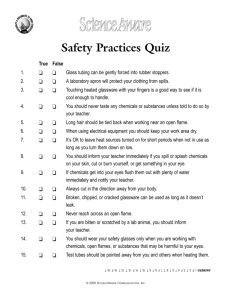
Fall Semester 2012 Jabin Miller, Rick Tarkka, and Robert Mauldin Compound: Name State CAS Number Lot # Container: Size Type Condition Percent remaining Date of Arrival Stability and storage information Manufacturing Company NFPA Hazard Codes Stockroom Schematic Key: Fire extinguisher Flammables and Flammable Waste Attic/Glassware Storage Bases Inorganic Waste Organic Resins glassware Air Conditioning Bulk Inorganic Organic Waste Metals Dumbwaiter Sink Base solns Empty bottles Temporary waste station Sink Acid solns Attic Organic Chemicals (A-S) Oxidizers Waste Household goods General Chemistry Solns Plasticware/ equipment Glassware/ equipment Fume Hoods/ Acids Stockroom Office Eye Wash/ Safety Shower Exit Exit Safety Glass Shower Tubing Sink Files Oxidizers Prepped Organic Organic Chemicals Labs (T-V) Sink Refrigerator Bulk Organic Indicators Inorganic Chemicals Chem demos Attic First aid kit HEALTH HAZARD 4 – Deadly 3 – Extreme danger 2 – Hazardous 1 – Slightly hazardous 0 – Normal material 2 3 SPECIFIC HAZARD Oxidizer OX Acid ACID Alkali ALK Corrosive COR Use NO WATER Radiation Hazard FIRE HAZARD Flash Point 4 – Below 73 F 3 – Below 100 F 2 – Below 200 F 1 – Above 200 F 0 – Will not burn 1 OX REACTIVITY 4 – May detonate 3 – Shock and heat may detonate 2 – Violent chemical change 1 – Unstable if heated 0 – Stable The MSDS for every chemical in your lab must be available to you. We have them available in paper form, but the quickest and easiest way to access MSDS is via the Internet. A computer is available in the stockroom office specifically for this purpose. Material Safety Data Sheets (MSDS) for any chemical give information regarding hazards, reactivity, and disposal. MSDS are available in hard copy upon request, or electronically from the following web-sites: www.hazard.com www.fisherSci.com www.sigmaaldrich.com “A student using research to meet the ACS certification requirements must prepare a well-written, comprehensive, and welldocumented research report including safety presentations.” Address MSDS safety information regarding handling and disposal; address safety issues particular to your research project. Safety Equipment Your eyes are important. Please wear safety glasses! When using gloves, make sure that the composition of the glove will not allow the chemicals I am using to penetrate the glove. Latex gloves work well for aqueous solutions and pathogens, but the blue nitrile gloves have better resistance to organic solvents. CAUTION For the safe operation of this safety shower and eyewash station, please remove any electronics from the vicinity and do not store anything below them. The main function of a fume hood is to exhaust gases that are generated in the hood to the outside. Make sure the exhaust blower is operating. Do not put your face inside! Minimize storage of chemicals. Close sash when not in use. Clean spills immediately. Work with the sash at the proper operating level. Use emergency purge if needed. "Picture a pasture open to all. It is to be expected that each herdsman will try to keep as many cattle as possible on the commons.... As a rational being, each herdsman seeks to maximize his gain. Explicitly or implicitly, more or less consciously, he asks, 'What is the utility to me of adding one more animal to my herd?'" The Tragedy of the Commons, Garrett Hardin, Science, 162(1968):1243-1248. There is a new system for borrowing chemicals: If you borrow a stock chemical (not mixed solutions), you must fill out a borrow card with the following information and place it on the shelf in place of the borrowed chemical: - Chemical Name -Your Name -Your Professor’s Name - Room in which the chemical will be used **Borrow cards are NOT necessary for solutions unless it is a stock bottle. THE STOCKROOM IS A RESTRICTED AREA. DO NOT OPEN THE DOOR TO ANYONE OTHER THAN UCA PERSONNEL AND APPROVED STUDENTS. IF YOU ARE UNSURE, CONTACT THE MAIN CHEMISTRY DEPARTMENTAL OFFICE BEFORE ALLOWING ACCESS. Waste containers must be labeled with the following: Name of waste (solute & solvent) Estimated amount of waste Estimated concentration of solute(s) Experiment in which the waste was generated (if applicable) Corrosives Examples: thionyl chloride, bromine, sulfuryl chloride, benzoyl chloride, triethyl amine, trihydrofluoride, benzene sulfuryl chloride Oxidizing Inorganic Acids Examples: sulfuric, nitric, chromic, and perchloric acids Organic acids & Non-oxidizing Inorganic Acids Examples: acetic, butyric, and formic acids; hydrochloric, hydrofluoric, hydrobromic, hydroiodic, and phosphoric acids Inorganic chemicals and solutions are organized alphabetically by chemical name, NOT chemical formula. Eg. NaCl would be filed with S, for sodium chloride, not N. There are areas in the stockroom set aside for specific types of waste (see next slide). To store waste containers until the following year, ensure that waste containers: Are labeled appropriately Are tightly sealed If the waste chemical should be kept cool, please store with the refrigerator waste. Stockroom Schematic Key: Fire extinguisher First aid kit Flammables Attic/Glassware Storage Bases Inorganic Waste Organic Resins glassware Air Conditioning Bulk Inorganic Organic Waste Metals Sink Base solns Empty bottles Temporary waste station Sink Acid solns Attic Organic Chemicals (A-S) Oxidizers Waste Household goods General Chemistry Solns Plasticware/ equipment Glassware/ equipment Fume Hoods/ Acids Stockroom Office Eye Wash/ Safety Shower Exit Exit Safety Glass Shower Tubing Sink Files Oxidizers Prepped Organic Organic Chemicals Labs (T-V) Sink Refrigerator Bulk Organic Indicators Inorganic Chemicals Chem demos Attic Dumbwaiter Incoming chemicals must be labeled with the following: Date received NFPA hazard diamond (if not labeled by company) These metal trash cans are for empty glass chemical containers. “Empty” is defined as less than 2.5 cm of residue or less than 3% of the total weight. Depending on the chemical, use water or an organic solvent to rinse the container before placing in a metal trash can. If the chemical is hazardous (toxic, corrosive, reactive, or highly flammable), collect the rinse and place in a labeled chemical waste container. Take care when transporting chemicals and use rubber carriers for glass, 4 L containers. If elemental mercury is spilled, notify your research advisor immediately for safe cleanup and disposal. Keep ferromagnetic objects away from strong magnetic fields, such as near our Nuclear Magnetic Resonance (NMR) instrument. Check the label on each chemical container before using it to make sure it contains what is needed for the experiment. Only take as much of the chemical as is needed for the experiment at hand. If a small fire is confined to a container, I will allow it to burn until the fuel has been exhausted or cover the container. If there is a small fire that is not in a container, use a fire extinguisher. If there is a large, open fire, leave the area immediately and notify others. If clothing is on fire, Stop, Drop, and Roll (not run!). New fire blanket instructions posted. Do not use an open flame in the vicinity of flammable organic solvents. Do not store oxidizers near flammables with an NFPA rating of 2 or above. Check for leaks in gas cylinder connections, make sure that gas cylinders are strapped to the wall, and used a cylinder cart for transporting gas cylinders (with caps). Recognize that at the temperature of liquid nitrogen (77 K), oxygen in the atmosphere condenses and might react with chemicals in the container or liquid nitrogen trap to form a potentially explosive mixture. Do not use liquid nitrogen in a small, confined space since the nitrogen can displace oxygen in the air and result in asphyxiation.