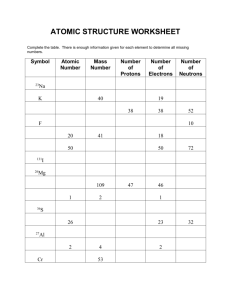
different # neutrons
advertisement

Topic: Iosotopes and Avg. Atomic Mass Do Now: 1) 2) 3) 4) 5) Dalton 1766 Atoms (tiny) Atoms of same element are identical * Indestructible * Combine in whole # ratio (H2O) In chem rxn. are combined, separated, rearranged *turned out to not be entirely be true Remember #2…well Dalton was correct… These three hydrogens are found in nature All H’s have 1 proton H-1: mass # = 1 1-1 = 0 neutrons H-2: mass # = 2 2-1 = 1neutron H-3: mass # = 3 3-1 = 2 neutrons Isotopes atoms of same element with different # neutrons (mass # is different) # protons stays same! (atomic # is same) Consider U-234, U-235, & U-238 the atomic number of U? 92 How many protons in U? 92 How many neutrons in U-234? 234 – 92 = 142 How many neutrons in U-235? 235 – 92 = 143 How many neutrons in U-238? 238 – 92 = 146 How many electrons in U? 92 What’s Average Atomic Mass Average Atomic Mass The atomic masses reported in the periodic table represent the weighted average of the masses of the naturally occurring isotopes of that element Unit for Avg. Atomic Mass = Amu (or regents says just u) – atomic mass unit Define as 1/12 the mass of a Carbon-12 atom Avg. Atomic Mass of Chlorine (Cl has 2 isotopes) % Abundance Cl-35 Cl-37 75.770% 24.230% Mass(percent) + mass(percent) +…= avg. atomic mass 35(.75770) + 37(.24230) = 35.453 amu NOW YOU TRY Avg. Atomic Mass of Si 92.21% of Si has a mass of 27.97693 amu 4.70% of Si has a mass of 28.97649 amu 3.09% of Si has a mass of 29.97376 amu Avg. Atomic Mass of Si 0.9221 X 27.97693 25.7975 amu 0.0470 X 28.97649 1.3619 amu + 0.0309 X 29.97376 0.9262 amu 28.0856 amu