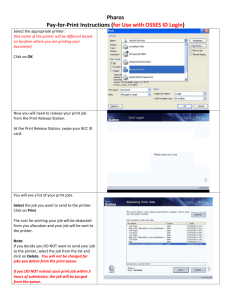
File
advertisement

Understanding Study Design & Statistics Dr Malachy O. Columb FRCA, FFICM University Hospital of South Manchester NWRAG Workshop, Bolton, May 2015 COIs: Interesting Confllicts! Editorial Board Roles: European Journal of Anaesthesiology British Journal of Anaesthesia International Journal of Obstetric Anesthesia Manuscript Types (7) (6) (5) (4) (3) (2) (1) Meta-analysis & systematic reviews Original research – PDBRCT Original research – other RCT Original research – observational Original research – retrospective Narrative reviews – including editorials Case reports, abstracts & letters Manuscript Types (7) (6) (5) (4) (3) (2) (1) Original research – PDBRCT Original research – other RCT Original research – observational Original research – retrospective Meta-analysis & systematic reviews Narrative reviews – including editorials Case reports, abstracts & letters Statistics: Definition …the discipline concerned with the treatment of numerical data derived from groups of individuals… Data …are always plural… ‘Datum’ is the singular… Types of Data Numerical – continuous & discrete Categorical – binary, nominal, ordinal Hypotheses Null hypothesis (HO) Alternative hypothesis (HA) P value and 95% confidence interval Two-sided by convention One-sided are rarely appropriate Equivalence, Non-inferiority, Superiority (Margins) Inequality is the usual HA Potencies and probabilities: One-sided P values suggest a one-sided story! Columb MO, Polley LS. Anesthesia & Analgesia 2001;92:278-9 Controlling Bias - Design Prospective > Retrospective Double Blind > Single Blind > Unblinded Randomised Controlled Trial > Unrandomised PDBRCT > Propensity Score Matching! PROBE (Single Blind) Sample Size Power analysis and sample size calculations. Columb MO, Stevens A. Current Anaesthesia & Critical Care 2008; 19: 12-4. Sample size Minimum difference that is (clinically) important Defines primary outcome! Multiple comparisons! Power analysis and sample size calculations. Columb MO, Stevens A. Current Anaesthesia & Critical Care 2008; 19: 12-4. Estimate of SD Published research Pilot data Empirical approach 1/5th – ‘one fifth’ of the range Power analysis and sample size calculations. Columb MO, Stevens A. Current Anaesthesia & Critical Care 2008; 19: 12-4. One-Fifth Range 4 SD = 95.4% of values 6 SD = 99.7% of values Take 1/5th range to approximate SD 20% of the range Power analysis and sample size calculations. Columb MO, Stevens A. Current Anaesthesia & Critical Care 2008; 19: 12-4. Standardised Difference Difference / SD Power analysis and sample size calculations. Columb MO, Stevens A. Current Anaesthesia & Critical Care 2008; 19: 12-4. Standardised Difference = 1.0 Power and sample size estimation Armitage & Berry pp 195-202 Sample size estimate for comparison of 2 equal groups 1 minimum difference to be significant 1 SD 0.05 type 1 error rate, P value 0.8 power 1.959964 Z 2 alpha 2 sided 0.841621 Z 2 beta 1 sided 18 = minimum n for each group 20 = nonparametric Nonparametric Adjustment Add 16% more subjects per group! Power analysis and sample size calculations. Columb MO, Stevens A. Current Anaesthesia & Critical Care 2008; 19: 12-4. Sample Size - Proportions Power analysis and sample size calculations. Columb MO, Stevens A. Current Anaesthesia & Critical Care 2008; 19: 12-4. Standardised Difference = 1.0 Sample size estimate for comparison of 2 proportions 0.75 proportion 1 0.25 proportion 2 0.05 type 1 error rate, P value 0.8 power 1.959964 Z 2 alpha 2 sided 0.841621 Z 2 beta 1 sided 0.5 pooled proportion 14 = minimum n per group for uncorrected Chi square test 18 = Fleiss continuity correction, minimum n per group Descriptive Statistics Sample Mean (SD) – 68% of data Median [interquartiles, range] Count/frequency Inferential Statistics - Precision Population estimates; precision Differences in means, medians, proportions Mean or mean difference Sampling theory! Population (variable X) x µ Randomization Population of means (variable ) Sample 1 x1,x2.....xn 1 Sample 2 x1,x2.....xn Sample 3 x1,x2.....xn 2 3 Distribution of sample means (variable ) µ ......... Sample j x1,x2.....xn j ..... 100 random samples of size 4 100 random samples of size 20 100 random samples of size 50 100 random samples of size 250 Inferential Statistics - Precision SD of sampled means is the SE of mean SE mean = SD / n SEM = 68%CI, (precision) SEM x 1.96 = 95%CI (precision) Test statistic = difference / SE difference P value Significance P value – ‘probability of the observed difference or greater assuming the null hypothesis’ Type I or alpha error <0.05; false +ve Type II or beta error <0.20; false -ve Multiple comparisons - Bonferroni correction Corrections to 95% CI of difference Group Tests Groups Parametric 1 2 One sample t Unpaired t >2 ANOVA Parametric repeated One sample t Paired t Repeated measures ANOVA Nonparametric Wilcoxon signed rank MannWhitney U KruskalWallis Nonparametric repeated Wilcoxon signed rank Wilcoxon matched pairs Friedman’s Proportions Binomial Chi square or Fisher’s exact Chi square or expanded Fisher Statistical Analyses Correlation – Pearson, Spearman, intraclass Regression – linear, logistic, probit, survival Diagnostics – sensitivity, specificity, ROC curves Reference intervals – normal range Agreement – kappa, Bland-Altman plots Transformations Table. Some useful transformations Types of data Transformations Positive skew, increasing variance Logarithmic = ln x, log10 x Negative skew, decreasing variance Square power = x 2 Count; Poisson distribution Square root =x Time; survival data Inverse = 1/x Proportion; p, Binomial distribution Logit = ln (p/1-p) Arcsin = Arcsinp Probit = Cumulative Normal Deviate Time-to-Event: Log Transformation Analyses for RCT Per-Protocol (PP) Treatment-Received (TR) Received allocated treatment and completed protocol Largest estimate of effect size Selection bias for post-treatment withdrawals Received allocated treatment May not have completed the protocol Selection bias for pre-treatment withdrawals Intention-to-Treat (ITT) All randomised subjects – NO WITHDRAWALS May or may not have received the intervention Underestimates true effect size of treatment Most robust analysis MOCPASS – columbmo@msn.com Power and sample size estimation Armitage & Berry pp 195-202 Sample size estimate for comparison of 2 equal groups 20 minimum difference to be significant 20 SD 0.05 type 1 error rate, P value 0.8 power 14 = minimum n per group for uncorrected Chi square test 18 = Fleiss continuity correction, minimum n per group 20 = nonparametric Power estimation for comparison of 2 equal groups 20 minimum difference to be significant 20 SD 0.05 type 1 error rate, P value 20 sample size n in each group 1.959964 Z 2 alpha 2 sided 1.202314 Z 2 beta 1 sided 0.25 proportion 1 0.75 proportion 2 0.05 type 1 error rate, P value 0.8 power 1.959964 Z 2 alpha 2 sided 0.841621 Z 2 beta 1 sided 0.5 pooled proportion 1.959964 Z 2 alpha 2 sided 0.841621 Z 2 beta 1 sided 18 = minimum n for each group Sample size estimate for comparison of 2 proportions Power estimation for comparison of 2 equal groups 0.25 proportion 1 0.75 proportion 2 0.05 type 1 error rate, P value 20 n per group 1.959964 Z 2 alpha 2 sided 1.388312 Z 2 beta 1 sided 1.002815 Z 2 beta, corrected 0.5 pooled proportion 0.917479 = power for uncorrected Chi square test 0.89 = power of study 0.84 = power with Fleiss continuity correction