Titration & Oxidation #'s
advertisement

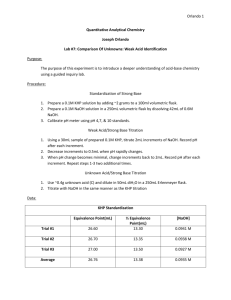
Chapter 4 Titration and oxidation numbers Part VI Titration Acid base reactions are often called neutralization reactions. One type of neutralization reaction is a titration. Titration: using a measured volume of a solution of known concentration to find the concentration of an unknown solution. Vocabulary Titration is a type of volumetric analysis. Titrant: aka standard solution. Analyte: solution being analyzed. Equivalence point: where [H+] = [OH-] The equivalence point is marked with a color change by an indicator. (as close as possible) This color changed is called the endpoint. Requirements 1. 2. 3. There are 3 requirements for a successful titration. The exact reaction between titrant and analyte must be known (and rapid). The equivalence point must be accurately marked. The volume of the titrant used to reach equivalence point must be known accurately. Titration Not all titrations are acid-base neutralization reactions. The next lab is a neutralization titration known as Standardization of a Solution. Example: Standardization A 1.3009 g sample of KHP potassium hydrogen phtalate (KHC8H4O4) with molar mass of 204.22 g/mol is weighed out. It has one acidic hydrogen. It is dissolved in water and phenolpthalein, an indicator is added. The student titrates the unknown NaOH to the pink endpoint. The difference in the buret reading is 41.20 mL. Calculate the concentration of NaOH. Answer: Reaction: OH- (aq)+ HC8H4O4- (aq) H2O (l)+ C8H4O42(aq) Ratio is 1:1 1.3009g KHP x 1mol/204.22g=0.0063701 mol KHP 0.0063701 mol KHP = 0.006371 mol NaOH M NaOH = 0.0063701 mol NaOH/0.04120 liters 0.1546 M NaOH A tough one: You have a mixture of carbon tetrachloride, (CCl4) and benzoic acid (HC7H5O2) a weak acid with one acidic hydrogen. A sample of this mixture weighing 0.3518 g was mixed with water and the resulting solution required 10.59 mL of 0.1546 M NaOH for neutralization. What was the mass percent of benzoic acid in this solution? Answer: You have both CCl4 and HC7H5O2 in solution but only the acid will react with NaOH, and in a 1:1 ratio. HC7H5O2(aq) + OH-(aq) H2O(l) +C7H5O2- (aq) How many moles of NaOH used? 10.59ml x (1L/1000mL) x (0.1546 mol/1 L)= 0.001637 mol OH- = 0.001637 mol HC7H5O20.001637 mol HC7H5O2 x 122.12g/1 mol = 0.1999 g HC7H5O2 Is that the answer? % mass (0.1999 g of acid/ 0.3518 total mass)*100 =56.82% Reread the question!! Oxidation Rules In many reactions there is an exchange of electrons. This transfer is called an oxidation-reduction reaction of redox for short. LEO says GRE Oxidation states or Oxidation numbers 1. 2. 3. A method for tracking electrons: 6 simple rules An atom in an element is zero Na(s) O2(g) A monatonic ion is the same as its charge. H+ =+1, O2- = -2 Fluorine is -1 in its compounds Oxidation states or Oxidation numbers cont. Oxygen is usually -2 in its compounds. The exceptions are peroxides O2-2 O=-1 5. Hydrogen is +1 in its compounds 4. 6. Also Important: the sum of oxidation numbers in a neutral compound = zero. The sum of oxidation numbers in an ion equal its charge. Try CO2 SF6 NO3C=+4, O=-2 S=+6, F=-1 N=+5 , O=-2