Chapter 12
advertisement

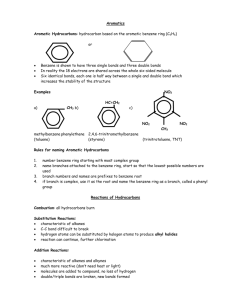
Chapter 12 - Reactions of Benzene - EAS 12.1 Introduction to benzene vs. alkenes 12.2 Mechanistic principles of Electrophilic Aromatic Subsitution 12.3 Nitration of benzene, reduction to aminobenzenes 12.4 Sulfonation of benzene 12.5 Halogenation of benzene 12.6 Friedel-Crafts alkylation of benzene 12.7 Friedel-Crafts alkylation of benzene 12.8 Alkylbenzenes via acylation then reduction 12.9 Rate and regioselectivity in EAS 12.10 Nitration of toluene - rate and regioselectivity 12.11 Nitration of CF3-benzene - rate and regioselectivity 12.12 Substituent effects in EAS: Activating Substituents 12.13 Substituent effects in EAS: Strongly Deactivating Substituents 12.14 Substituent effects in EAS: Halogens 12.15 Multiple substituent effects in EAS 12.16 Regioselective synthesis of disubstituted aromatic compounds 12.17 Substitution in Naphthalene 12.18 Substitution in heterocyclic aromatic compounds Chapter 12 - Reactions of Benzene - EAS YSU Introduction – Benzene vs. Alkenes Br Br Br no heat dark Br Br Br no heat dark no colour change (no reaction) Completely delocalized (6) pi system lends stability (aromatic) YSU 12.2 Mechanistic Principles of EAS Alkenes react by addition E Y E E Y Y Benzene reacts by substitution E Y H Y E -H Y E Resonance-stabilized cation YSU General Mechanism of EAS on Benzene Energy diagram for EAS in benzene Figure 12.1 YSU Electrophilic Aromatic Substitutions on Benzene H HNO3, H2SO4 NO2 H SO3, H2SO4 SO3H H Br2, Fe 12.3 Nitration 12.4 Sulfonation 12.5 Halogenation Br YSU 12.6 Friedel-Crafts Alkylation of Benzene H CH3CH2Cl CH2CH3 AlCl3 CH3 H C CH3 (CH3)3CCl CH3 AlCl3 H3 C H Cl CH3 CH3 C CH3 CH3 AlCl3 Problems: Alkyl groups may rearrange during reaction Products are more reactive than benzene Uses: Alkyl benzenes readily oxidized to benzoic acids using KMnO4 YSU 12.7 Friedel-Crafts Acylation of Benzene H O O RCCl C R AlCl3 O H H3 C O O O CCH3 CH3 AlCl3 O RCCl AlCl3 O RC R C O Products react more slowly than benzene - cleaner reaction No carbocation rearrangements YSU 12.8 Alkylbenzenes via Acylation/Reduction O Zn, HCl C (Clemmensen) H H C R R or NH2NH2, KOH heat (Wolff-Kischner) O Cl AlCl3 Zn, HCl O Make the acyl benzene first (clean, high yielding reaction) Reduce the ketone group down to the methylene (C=O to CH2) Avoids rearrangement problems, better yields YSU Aminobenzenes via Nitration/Reduction (not in text) H O N Sn, HCl N O H H H HNO3, H2SO4 NO2 Sn, HCl N H Make the nitro benzene first, clean high yielding reaction Reduce the nitro group down to the amine Difficult to introduce the amino group by other methods YSU 12.9 Activation and Deactivation by Substituents (Rates) X X HNO3, H2SO4 NO2 CF3 H 0.000025 1.0 CH3 25 Relative rates in nitration reaction now bringing in a second substituent YSU 12.9 Nitration of Toluene vs Nitration of (Trifluoromethyl)benzene CH3 CH3 HNO3, H2SO4 CH3 CH3 NO2 + + NO2 NO2 63% 3% 34% CH3 is said to be an ortho/para director (o/p director) - Regioselectivity CF3 CF3 HNO3, H2SO4 CF3 CF3 NO2 + + NO2 NO2 6% 91% 3% CF3 is said to be a meta director (m director) - Regioselectivity YSU 12.10 Rate and Regioselectivity in Nitration of Toluene CH3 CH3 HNO3, H2SO4 CH3 CH3 NO2 + + NO2 NO2 63% 3% 34% Fig. 12.5 – Energy diagrams for toluene nitration (vs. benzene) YSU 12.11 Rate and Regioselectivity in Nitration of CF3C6H5 CF3 CF3 CF3 HNO3, H2SO4 CF3 NO2 + + NO2 NO2 6% 91% Fig. 12.6 – Energy diagrams for CF3C6H5 nitration (vs. benzene) 3% YSU 12.12 Substituent Effects – Activating Substituents General : all activating groups are o/p directors halogens are slightly deactivating but are o/p directors strongly deactivating groups are m directors Activating Substituents O R alkyl OCH3 Example: RO HO hydroxyl HNO3, H2SO4 RCO alkoxy acyloxy OCH3 OCH3 NO2 + NO2 Alkyl groups stabilize carbocation by hyperconjugation Lone pairs on O (and others like N) stabilize by resonance YSU 12.13 Subst. Effects – Strongly Deactivating Substituents O O O HC R C HO C aldehyde ketone carboxylic acid Cl O O C RO C ester acyl chloride O HO S N C O2N O nitrile sulfonic acid NO2 nitro NO2 Br2, Fe Example: Br Second substituent goes meta by default – best carbocation YSU 12.14 Substituent Effects – Halogens X X X = F, Cl, Br X = F, Cl, Br X Reactivity (i.e. rate) is a balance between inductive effect (EW) and resonance effect (ED) – larger Cl, Br, I do not push lone pair into pi system as well as F, O, N, which are all first row (2p) Example: Cl Cl HNO3, H2SO4 Cl Cl NO2 + + NO2 NO2 30% 1% 69% Regioselectivity - second substituent goes o/p – better carbocations YSU 12.15 Multiple Substituent Effects CH3 O H3C O NHCH3 CH3 O O CH3 NHCH3 Br Br2, acetic acid CH3 AlCl3 Cl CH3 CH3 Cl 87% 99% Br Br2, Fe NO2 CH3 CH3 CH3 NO2 CH3 NO2 HNO3, H2SO4 C(CH3)3 86% C(CH3)3 88% Interplay between power of directing group and size of substituents YSU 12.16 Regioselective Synthesis of Disubstituted Derivs. O O CH3 O CH3 CH3 Br Br NO2 CO2H CH2CH3 Br NO2 Have to be careful about when to introduce each substituent Remember – isomers (e.g. o/p mixtures) may be separated YSU 12.17-12.18 Substitution in Other Aromatic Systems 12.17 Naphthalene O CH3 O O CH3CCl CH3 but not AlCl3 90% 12.18 Heterocycles SO3H SO3, H2SO4 N HgSO4, 230 oC O H3C O N O O BF3 CH3 CH3 O O YSU