Chemical Nomenclature

Chemical
Nomenclature
Learning Targets
•Write name & write symbol of selected elements
•Write name & write formula of a variety of compounds.
Naming Inquiry
Formula
NaCl
KI
MgCl
2
Name
Sodium Chloride
Potassium Iodide
Magnesium Chloride
What is a binary compound?
Naming Inquiry
Formula
NaCl
Name
Sodium Chloride
KI Potassium Iodide
MgCl
2
Magnesium Chloride
What does the chemical formula of each indicate?
Naming Inquiry
Formula
NaCl
Name
Sodium Chloride
KI Potassium Iodide
MgCl
2
Magnesium chloride
Why are there two chlorines for one magnesium?
Naming Inquiry
Formula
NaCl
KI
Name
Sodium Chloride
Potassium Iodide
MgCl
2
Magnesium Chloride
What are some naming rules that can be discovered from these examples?
I.
A.
Elements (periodic table)
Symbols: come from the first letter, or first two letters of the element’s name.
1. First letter is always UPPERCASE, second letter always lower case.
a.
b.
Co, cobalt vs. CO, carbon monoxide
No, nobelium vs. NO, nitrogen oxide
2. Some use latin name for symbol, ex. Gold
(latin name = aurum or Au)
NOTES
II.
Formula: Represents composition of a compound
A.
Tells proportions of elements in a compound relative to each other.
1. Subscripts are used to indicate number of atoms of each element in a compound. ( ALWAYS Whole numbers!!)
2. The subscript follows the atom or atoms it refers to. If subscript follows “( )” then everything inside the “( )” is multiplied by the subscript.
Examples:
CO
2
One atom of C, 2 atoms of O
H
2
O
2 atoms of H, one atom of O
Fe
3
(PO
4
)
2
3 atoms of Fe, 2 atoms of P & 8atoms of O
Formulas, continued
B.
All atoms have oxidation numbers
(valence charge). This is the combining power of the atom.
ex : Everything in Group 1 has a
+1 charge. Group 17 has -1 charge
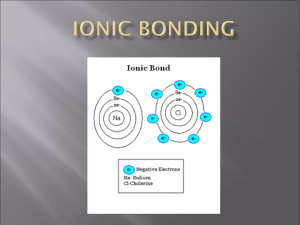
Na +1 Cl -1 combined = 0
** You want your valences to always = 0
Examples continued
What happens when it doesn’t = 0?
Mg +2 Cl -1 = 0
You add another Cl -1
-2 so… it now looks like this:
MgCl
2
Now the “combined power” is 0
You Try
Ca +2 N -3
K + P -3
Al +3 O -2
C.
Polyatomic ions or “radicals” are groups of atoms that behave as if they are single atoms. They also have oxidation numbers. Examples: SO
4
-2
(sulfate ion), NO
3
-1 (nitrate ion)
D.
We will use the concept of valence to write formulas
1.
Valence is either “+” or “-”
2.
In ALL compounds, the sum of the valences adds up to zero.
3.
Metals are always “+”
4.
Non-metals can be “+” or “-”
5.
Most radicals are “-”, only common “+” radical is NH
4
+1 (ammonium)
Application
1.
Example 1: Lithium Chloride …Li 1+ and Cl 1, so the formula must be
LiCl.
2.
Example 2: Calcium Oxide …Ca 2+ and O 2, so the formula must be CaO
3.
Example 3: What about Lithium
Oxide?
4.
Answer: you need two lithium atoms for every one oxygen to balance…Li
2
O
E.
Atoms that have a constant valence:
1. (+1) = first column and Ag,
2. (+2) = 2 nd column plus Zn and Cd
3. (+3) Al. (See columns already marked on your periodic table.)
(H can be -1 in some special cases)
III.
Naming Binary Salts & Writing their
Formulas
A.
Metal plus a non-metal (two elements).
B.
Named by adding the name of the first element (metal) to the second element
(non-metal) whose name is modified to end in “ide.”
1. Examples:
• oxygen becomes “oxide”
•
• chlorine becomes “chloride” sulfur becomes “sulfide”
• etc…
Now… The Rules
Rule #1: ends in “ide” = Binary salt
NaCl
Sodium chloride
Al
2
O
3
Aluminum oxide
KI
Potassium iodide
•
Rule #2: valences must = 0
C.
Most metals have a variable valence and this must be indicated in the name of the compound.
1.
The Stock System: The valence (+) of the metal is given as a Roman numeral.
a. Iron (Fe) can either be Fe +2 or Fe +3 therefore both forms exist in compounds.
1.
2.
3.
Iron (II) chloride = FeCl
2
Iron (III) chloride = FeCl
3
The Latin names ferrous (for Fe +2 ) and ferric (for Fe +3 ) may also be used.
How does the Stock System
Work?
For metals in Groups 3-15
Iron (II) chloride = FeCl
2
The (II) tells me the valence of Fe.
I know that each Cl has a -1 charge. I have 2 Cl’s for a total charge of -2, so the Fe has to be +2 to make the valences add up to zero
FeCl
3 what is the oxidation of Fe?
Iron (III) chloride, Fe +3
You Try -
Write the name or the formula using the
Stock System
CuCl
Copper (I) chloride
CrO
3
Chromium (VI) oxide
Tin (IV) oxide
SnO
2
Nickel (II) oxide
NiO
End notes 10/5 & 10/6