The National Institutes of Health
advertisement

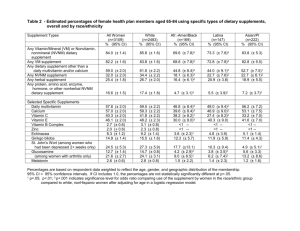
Natural Health Products in a Global Marketplace: Scientific and Regulatory Perspectives Joseph M. Betz, Ph.D. Office of Dietary Supplements U.S. National Institutes of Health http://dietary-supplements.info.nih.gov Overview Dietary Supplement Overview Introduction to Regulations Introduction to National Institutes of Health Overview of ODS Programs Dietary Supplements in 2001: $17.7 Billion Minerals Vitamins $1.2 $5.8 Herbs / Botanicals $4.1 Other Supplements $3.8 Sports Nutrition $1.5 Source: NBJ, derived from a variety of sources Top 10 Dietary Supplements for 2005 Multivitamins/minerals Calcium B vitamins Vitamin C Glucosamine/Chondroitin Vitamin E Fish oils Coenzyme Q10 Noni juice Probiotics Nutrition Business Journal Ten Most Common Reasons for Taking Supplements (The Slone Survey) • • • • • • • • • • • General health/good for you (16%) Arthritis (7%) Memory improvement (6%) Energy (5%) Immune booster (5%) Joints (4%) Supplement diet (4%) Sleep aid (3%) Prostate (3%) No reason (2%) All other reasons (45%) JAMA, 2002 Traditional (Herbal) Medicine in the U.S. Practice of medicine and the licensing of physicians are regulated by the 50 individual states National laws deal mostly with products • U.S. Food and Drug Administration (FDA) FD&C Act Products Foods, dietary supplements, or drugs based on “intended use” • e.g Psillyium husk can be a food, dietary supplement or drug • Applicable regulations for the category apply Intended use is ascertained by accompanying labeling claims, advertising materials, or oral or written statements Botanical Drugs Intended use to cure, treat, mitigate, etc. Subject to current drug regulations – Pre-market approval for GRASE Unique qualities recognized by the FDA – inherently more complex than synthetics – more likely to be components in mixtures – active constituents and activity not always well defined DSHEA (Dietary Supplement Health and Education Act - 1994) • Amended the FD & C Act • Defined dietary supplements • Established regulatory framework • Food and Drug Administration (FDA) • As foods, not as drugs • Established rules for what a label should contain • Gave FDA authority to write GMP • Called for creation of the Office of Dietary Supplements – Placed it in the Office of the Director at the National Institutes of Health Dietary Supplement • “...any product (other than tobacco) that contains a vitamin, mineral, herb, or other botanical or amino acid and is intended as a supplement to the diet.” • A concentrate, metabolite, constituent, or combination of the above – Prior to DSHEA, most botanicals treated as food additives or as drugs • Approved, unapproved Dietary Supplement Foods, therefore safe by definition • No premarket safety approval except “new dietary ingredients” (75 day notification) Statements of Nutritional Support (“Structure/Function”) claims permitted • No premarket approval for efficacy required (30 day notification) Disease claims prohibited Intended for ingestion NOT REPRESENTED AS CONVENTIONAL FOOD Guide to Dietary Supplements http://vm.cfsan.fda.gov/~dms/dietsupp.html New Dietary Ingredients • Not marketed in U.S. prior to 10/15/94 • Not a new ingredient: – If ingredient was “in the food supply… in a form... not chemically altered” • No list of “FDA approved” ingredients • Mfr. Determines whether “new” – notifies FDA 75 d pre-market • Exempt from food additive provisions Dietary Supplements • 21 CFR Part 111 • Current Good Manufacturing Practice in Manufacturing, Packaging, Labeling, or Holding Operations for Dietary Supplements; Final Rule • Published June 25, 2007 • http://www.cfsan.fda.gov/~lrd/fr07625a.html Dietary Supplements • 21 CFR Part 111 – Requires manufacturers to set specifications – Requires manufacturers to test to see if specifications are met using “scientifically valid” methods General Provisions • CGMP applies to activities associated with – – – – manufacturing packaging holding distributing • Manufacturer needs to comply with requirements applicable to operations performed – contractor needs to comply with applicable requirements and – contracting firm responsible for contractor’s performance Proposed Requirement Highlights • • • • Personnel Physical plant internal environment Equipment and utensils Production and process controls – quality control unit – master manufacturing & batch production records Public Law 109–462 • Dietary Supplement and Nonprescription Drug Consumer Protection Act – http://www.fda.gov/opacom/laws/pl109462.html – 22 Dec 2006, took effect Dec 2007 • Mandatory Serious Adverse Event Reporting for OTC drugs and Dietary Supplements – Manufacturer must provide a mechanism for reporting (reports to manufacturer) – Manufacturer must report serious events to FDA Public Health Security and Bioterrorism Preparedness and Response Act of 2002 • Signed into law June 12, 2002 • Relevant Provision is Title 3: Protecting Safety and Security of Food and Drug Supply. • Title 3 contains 15 sections with relevant provisions to the DS industry. Homeland Security • SECTION 303: Administrative Detention – FDA may order the detention of foods when credible evidence exists that they present a threat of serious adverse health consequences or death to humans or animals • SECTION 305: Registration of Facilities – Requires that any facility engaged in manufacturing, processing, packaging or holding food for consumption in the U.S. register with the Secretary Homeland Security • SECTION 414: Maintenance/Inspection of Records – Companies and individuals involved in any aspect of food production must make and keep records – If FDA suspects dangerous adulteration, records must be made available • SECTION 307: Prior Notice of Imported Food Shipments – Require submission of prior notice of arrival providing each of the following: • Identity of: article, manufacturer and shipper, grower, country of origin, country from which article is shipped, port of entry – Articles failing to meet these requirements shall be refused admission to the U.S The National Institutes of Health DSHEA and the NIH • DSHEA called for creation of the Office of Dietary Supplements • Placed at the National Institutes of Health in the Office of the Director • Formally established in 1995 • Strategic planning process guided the first few years of its activities NIH is the Nation’s Medical Research Agency • 27 Institutes and Centers • Total NIH Budget for 2006: $28 billion • Grants, Contracts, Cooperative Agreements • Department of Health & Human Services www.nih.gov Research • Intramural- “in-house” at Institutes • Extramural – Grants, Cooperative Agreements, Contracts • http://grants.nih.gov/grants/funding/funding.htm – More than 80% of the NIH budget – Subject to competitive review • Investigator-initiated • Request for Application (RFA) The Primary Function of NIH is to Conduct and Support Medical Research • 65% Basic Research • 25% Translational • 15% Clinical NIH Funding for Dietary Supplement Research Has Increased Significantly $ Million 300 250 200 150 100 50 0 1999 2000 2001 2002 2003 2004 2005 THE OFFICE OF DIETARY SUPPLEMENTS ODS Mission is to Strengthen Knowledge and Understanding of Dietary Supplements – Evaluate Scientific Information – Stimulate and Support Research – Disseminate Research Results – Educate the Public to Foster an Enhanced Quality of Life and Health for U.S. Population Echinacea Echinacea purpurea Office of Dietary Supplements FY 2003 $23.0 M 2% 6% 29% 15% 5% 5% 12% 22% 4% Centers Program Grants Databases Administrative NHANES Evidence Program Analytical Methods Conferences Training ODS Has a Broad Array of Programs • Co-funding with NIH Institutes • Evidence-Based Reviews • Botanical Research Centers • Training St. John’s Wort Hypericum perforatum • Analytical Methods and Reference Materials • Communications and Information ODS Activities are Comprehensive and Global • Dietary Supplement Databases • Surveys of Supplement Use • International Collaborations • Workshops, Conferences, Seminars Ginkgo Ginkgo biloba RESEARCH AGENDA: Grants Co-funded with NIH ICs ODS Grant “Portfolio” Growth of ODS co-funded grant portfolio: • 1996: 6 grants initiated, $300,000 • 2002: 39 grants initiated, total of 59 (new and continuing), $9.3 million • 2003: total of 72 grants, $13.5 million Broad portfolio: basic, clinical, training Details available on the ODS website RESEARCH AGENDA: EvidenceBased Review Evidence-Based Review Program • Systematic review of the literature, with meta-analysis as appropriate, on DS efficacy and safety • In collaboration with the Agency for Healthcare Research and Quality (AHRQ) Evidence-Based Practice Center Network • Major reason for conducting these reviews is to assist NIH in the development of research agendas ODS Evidence-Based Review Program • Chromium and insulin sensitivity* • Ephedra for weight management and athletic performance enhancement* (with NCCAM) • Omega-3 fatty acids for cardiovascular disease prevention* (with NHLBI) and other health indications • Health effects of soy (with NCCAM) • Antioxidant phytochemicals/B vitamins and neurodegenerative diseases*(with NCCAM) • Multivitamins/multiminerals and chronic disease prevention (with OMAR) • Coagulation, diet, and dietary supplements • Health effects of vitamin D (with multiple partners) *Congressional mandate Evidence • • • • • • Pre-Clinical Ecologic Observational Cohort Intervention RCT Ephedra Efficacy and Safety • Systematic review: weight management and athletic performance enhancement • All languages, both published and unpublished • Conclusions: – Modest effect on weight in short term and no evidence of effect on athletic performance – Some side effects seen in trials, some serious adverse events filed with FDA and to a major ephedra manufacturer • Released by Secretary of DHHS, Feb 2003 – Shekelle P et al, JAMA 289:1537-1545, 2003 • ODS and NCCAM convened expert panel to recommend research agenda based on review Evidence-Based Review of Vitamin D in Relation to Bone Health Conducted by University of Ottawa Evidence-Based Practice Centre Funded by NIH Office of Dietary Supplements & Agency for Health Care Research and Quality Available at http://www.ahrq.gov/clinic/tp/vitadtp.htm & the ODS website Conference Objectives Evaluate available evidence on the efficacy and safety of vitamin D, using the AHRQ evidence-based review titled, Effectiveness and Safety of Vitamin D in Relation to Bone Health as the framework. Identify gaps in knowledge on the efficacy and safety of vitamin D in general and across the life cycle. . Findings from the Evidence Based Review • Strong evidence that Vitamin D supplementation reduces falls, fractures and bone loss in men and women 60+ y • Not possible to separate the impact of vitamin D from Ca supplementation— typical amounts used were 700-800 IU Vitamin D/d and 500-1,200 mg Ca/d • Sparse data on other subgroups • Difficult to identify a specific blood level of 25(OH) D indicative of optimal bone health in all population subgroups 2003 Conference: Vitamin D and Health in the 21st Century; Bone and Beyond (NICHD & ODS) Evidence-based Review (AHRQ & ODS) Standard Reference Materials Vitamin D Status 25-OH D2 and D3 Serum 25-OH D Measures in NHANES (NIST & ODS) (NCHS, FDA, ODS) Analytical Methods Development D2 & D3 in Foods & Dietary Supplements (USDA & ODS) Health Effects of Omega-3 Fatty Acids • Asthma • Cancer • • • • • • • • • Cardiovascular Disease Cardiovascular Disease Risk Factors Arrhythmogenic Mechanisms Child and Maternal Health Cognitive Function Eye Health Type II Diabetes, Rheumatoid Arthritis, and Other Diseases Mental Health Organ Transplantation Omega-3 Fatty Acids • 11 reports; 3 EPCs – 9 focused on human studies – 2 focused on animal and in vitro models – Collaborated on methodological elements • Multiple NIH Institutes and Centers • Multiple diseases and risk factors Omega-3 Fatty Acids Or Photo of Menhaden - Courtesy of National Oceanic and Atmospheric Administration (NOAA) Omega-3 and Cancer • RAND EPC (MacLean CH, JAMA 295:403, 2006) – Tumor incidence: prospective cohort studies – Clinical outcomes: RCTs – Tumor behavior: animal, cell culture studies Omega-3 and Tumor Incidence • 19 prospective cohorts, 11 cancers – Breast: 2 significant (1 positive, 1 negative), 5 not significant – Lung: 2 significant (1 positive, 1 negative), 4 not significant • Supported by another systematic review (Hooper L, BMJ 332:752, 2006) Health Effects of Omega-3 Fatty Acids • Secondary CVD Prevention – Reduces all cause mortality and other CVD outcomes (sudden death, cardiac death, MI) • Primary CVD Prevention – Large, consistent beneficial effect found for serum triglyceride levels – Little or no effect found for other CV risk factors and markers of CVD Omega-3 Reports • Heterogeneous data; challenging to evaluate – Methods of estimating fish or omega-3 fatty acid intake – Background diets – Methods of reporting results • Adverse events from clinical studies appear to be minor – However, AERs not reported in a systematic way From Concept to Research: Chromium Conference (1999) Evidence-based Review (2000) Initiative (2001) Funding (2001-2004) • Chromium and Insulin Action • Chromium Enhancement of Insulin Signaling • Chromium Effects in Impaired Glucose Tolerance • Chromium Picolinate in the Metabolic Syndrome • Elucidating the Biochemistry of Chromium III • Novel Method for Chromium Analysis in Biological Fluids • Adjuvant Therapy for Vascular Inflammation in Diabetes Systematic Reviews for Nutrition Topics • Applications – – – – Research Agenda (NIH) Clinical Practice (ADA) Nutrient Recommendations (IOM) Public Health (Gov’ts) • Challenges – Heterogeneity – Background Diet – Misgivings Systematic Reviews for Nutrition Topics • Questions Paramount • Why Do an EBR? – Vitamin E and Mortality – Antioxidants and Mortality • EBRs Inform, but Do Not Dictate • Keep Them Current RESEARCH AGENDA: Analytical Methods and Reference Materials Saldahna, LG, Betz, JM, Coates, PM (2004) JAOAC INT 87:162-165 Quality Correct plant Slifman et al., NEJM 339, 806-811, 1998 Correct plant part Plant collected at proper time of year Pathogen free Not filthy or decomposed, not moldy Aflatoxin, pesticide, toxic elements within acceptable range No extraneous material-prescription drugs, etc. “High quality” raw materials/finished product • Standardization? Marker Compounds? • Basic research into bioactive compounds needed Product consistency “The Marketplace” LA Times August 31, 1998 “Remedy's U.S. Sales Zoom, but Quality Control Lags St. John's wort: Regulatory vacuum leaves doubt about potency, effects of herb used for depression.” By TERENCE MONMANEY, Times Medical Writer ConsumerLab.com December 20, 2002 “1500% Variation Found in Strength of Garlic Supplements: Many labels not helpful, misleading” Can J Clin Pharmacol (2003) 10(3):114-118 “Only two products (of 54) were observed to have a total naphthodianthrone concentrations within 10% of their label claim” Draves AH, Walker SE Analytical Methods and Reference Materials • Part of original ODS Strategic Plan • U.S. Senate language for FY 2002, 03 Called for ODS to allocate sufficient funds to speed up an ongoing collaborative effort to develop and disseminate validated analytical methods and reference materials for the most commonly used botanicals and other dietary supplements. Program Goals • Designed to provide opportunities for public/private partnership – – • • • Establish priorities (botanicals and others) Identify potential research partners Develop, validate, share analytical methods Produce, share reference materials Applications – – Research: characterization of test substances Industry: implementation of GMPs/product formulation Recommendations • Pay attention to basic quality issues – Identity, contaminants • Accept role of existing frameworks for methods validation • Accept recommendations of AOAC DS task group for prioritization of ingredients Recommendations • • • • • Consensus process: experts, stakeholders Proposed a steering committee Timely output Program should reward innovation Define appropriate phytochemical markers (basic research) • Emphasis on methods for raw materials Needs • New methods • Validation of methods for reliability, accuracy, precision • Calibration standards (reference chemicals) for methods • Matrix reference materials for evaluation of method performance • All must be publicly available Strategic Overview 1. Short-term: build the infrastructure to support development of validated methods and reference materials; 2. Medium-term: fund development of validated methods and reference materials using standard NIH mechanisms; 3. Long-term: make methods and reference materials readily accessible to the user community. Saldahna, Betz, Coates (2004) JOURNAL OF AOAC INTERNATIONAL 87:162-165 Tactical Approach • Program with 4 major components – – – – Methods development Methods validation Reference materials development Outreach/dissemination • Includes education and training Accomplishments • 8 supplement methods have been approved as first action Official Methods-7 more expected by March 2008 • Published collaborative studies: – – – – – Ephedrine alkaloids in botanicals/dietary supplements Ephedrine alkaloids in plasma/urine Glucosamine in raw materials and finished products β-carotene in raw materials and finished products2 Flavonol glycosides in Ginkgo biloba raw materials and finished products – Campesterol, stigmasterol, and beta-sitosterol in Saw Palmetto raw materials and finished products Accomplishments • 9 Published Single Laboratory Validation Studies • 7 single-laboratory validation studies in progress • 38 additional ingredients in various stages of study Reference Materials • Reference Materials being developed by the ODS program: – Calibration Standards-Single chemical entities • For construction of calibration curves for quantitative analysis • For confirming analyte identity – Matrix Reference Materials-Systematically characterized supplement raw materials & finished products • Evaluation of analytical methods performance in individual laboratory settings – No identity reference materials (for now) Calibration Standards • Mix of research and procurement – National Research Council (NRC)-Panax ginsenoside RM – Calibration standard production for methods development • Echimidine, lycopsamine, n-methyltyramine – Calibration standards for AOACI validation work • Hypericin, hyperforin, ginkgolides, etc. – USP Reference Materials • Berberine, hydrastine, β-sitosterol, 6-gingerol, shoagol, eleutherosides B and E, actein, 23-epi-26-deoxyactein, secoisolariciresinol diglucoside Matrix Reference Materials National Institute of Standards & Technology (NIST) • 5-year project to produce SRMs for 6-8 botanicals-Interagency Agreement (IAG) – NIST, CFSAN, CDER, ODS collaboration – Obtain authentic Botanical Reference Materials (BRM) – Develop and validate analytical methods for actives and/or markers – Assign values using the NIST certification approachMeasure with 2 or more methods/labs – Assign values for heavy metals/pesticides – Issue NIST Certificate of Analysis National Institute of Standards & Technology (NIST) • Completed • • – • • • Ephedra Ω-3’s in cod liver oil RM Ginkgo biloba β-carotene (carrot oil) Bitter orange Multivitamin/mineral • 2007 • • • • • • • Saw palmetto α-tocopherols Green tea Various Vaccinium berries Ω-3’s in seed oils Vitamin D in serum Vitamins B6, B12 in serum • Acquisitions in progress (no date available) • St. John’s wort • Black cohosh Future Directions • Based in part on reviewer recommendations: – Accelerate methods development/validation by: • Supporting mechanism for selection, prioritization, review of methods • Devoting more resources to direct funding of laboratory work • De-emphasizing Official Methods of Analysis • Concentrating on optimization/Single Laboratory Validation studies • Engaging ODS Botanical Centers Program with AMRM Future Directions • Provide more resources for development, optimization, and validation of methods and materials for nutrient determination in DS – Systematic review of the state of nutrient methods Future Directions • Provide increased resources for tools that allow laboratories to demonstrate proficiency – Emphasis on evaluation tools for SLV methods – Expand NIST SRM program – NIST pilot laboratory proficiency program has begun and is recruiting participants • Provide increased resources for tools that allow stakeholders to use methods – Calibration standards Conclusions • Past 5 years have been a time to identify stakeholders and build an analytical community – Researchers, QA personnel, Contract Laboratories – Progress is accelerating • Supplement Methods published in J AOAC Int: – 2000-2001-Zero – 2002-2006-approximately 100 – Challenges remain • Estimated number of unique products projected by 2010 = 40,000 RESEARCH AGENDA: Dietary Supplement Ingredient Database Database of DS Ingredients • ODS held workshops with Federal and academic researchers, and the private sector to explore the feasibility of developing databases for dietary supplements • Considerable research funded by NIH and other agencies on dietary supplement use – Depends on accurate measures of intake • Emerging opportunities for public-private partnership in meeting research needs • Published in J Nutrition 133: 573S – 634S, 2003 • Congressional language (’04) Progress Toward Analytically Supported Dietary Supplement Database NHANES Label-Based Dietary Supplement Database DSID Dietary Supplement ingredient Database at ARS Goal: Analytically Supported Dietary Supplement Database RESEARCH AGENDA: Botanical Research Centers Congressional Mandate Botanicals Initiative 1999 “Establish a botanical research initiative with major research institutions in the United States.” Office of Dietary Supplements National Institutes of Health National Center for Complementary and Alternative Medicine National Institutes of Health What is a Botanical? • Whole plants or plant parts (e.g., bark, leaves, stems, roots, flowers, fruits, seeds, berries, extracts) • Medicinal Plants and Foods Ginger Zingiber officinale • Algae and macroscopic fungi Cranberry Vaccinium macrocarpon Flaxseed Linum usitatissimum Green Tea Camellia sinensis St. John’s Wort Hypericum perforatum BRC Program Objectives • Build Collaborative Research Teams to Advance Basic Science to Inform Clinical Studies – Characterize Chemical Composition and Study Their Biological Effects – Develop and Improve Preclinical Model Systems • Cultivate the Use of Contemporary Technologies and Innovative Approaches • Conduct Clinical Studies (optional) Echinacea Echinacea purpurea Botanical Research Centers 2005 - 2010 • Multidisciplinary Teams • Emphasis on Quality Assurance/Quality Control • Studies of Basic Mechanisms • Identify Active Constituent(s) and Explore Mechanism(s) of Action • Clinical Evaluation (optional) • Thematic Focus With High Public Health Impact EAC Research Projects Administrative Core Pilot Studies Training P1 P2 P3 Resource Cores Botany Plant Sciences “Chemistry” Clinical Studies Biostatistics Each NIH Botanical Research Center has its Own Individual Focus • University of Illinois at Chicago – Botanicals for Women’s Health • Purdue University/University of Alabama Birmingham/ Rutgers – Botanicals for Age-Related Diseases • Iowa State University/University of Iowa – Botanicals and Immune Function Each NIH Botanical Research Center has its Own Individual Focus • Pennington Biomedical Research Center/Rutgers – Botanicals and Metabolic Syndrome • Memorial Sloan Kettering/Cornell/Chinese University Hong Kong – Botanical Immunomodulators and Cancer • Wake Forest/Brigham and Women’s Hospital – Botanical Lipids and Inflammatory Diseases Sample Research Questions • Do Soy Isoflavones Have Beneficial Skeletal Effects in Postmenopausal Women? • Can Black Cohosh Alleviate Menopausal Hot Flashes? • Do Anthocyanins Improve Insulin Sensitivity? • Does Echinacea have antiviral activity? Two EB Articles • Botanicals for age-related diseases: from field to practice. CM Weaver, S Barnes, JM Wyss, H Kim, DM Morre, DJ Morre, JE Simon, M Lila, EM Janie, and MG Ferruzzi • Technologies and experimental approaches at the National Institutes of Health Botanical Research Centers. S. Barnes, DF Birt, BR Cassileth, WT Cefalu, FH Chilton, NR Farnsworth, I Raskin, RB van Breemen, and CM Weaver Multidisciplinary Research • Collaborative Effort • Make Use of Unique Areas of Expertise • Integrate Activities • Develop Collegial Environment • Recognize Individual Contributions www.nih.gov RESEARCH TRANSLATION: Information Resources Information Resources • Conferences/Workshops • Research Findings • Fact Sheets – Vitamins, minerals, botanicals • Bibliographic Database – World literature – Consumer version "The secret of being tiresome is to tell everything." -Voltaire