Final Exam 2 Review Summer
advertisement

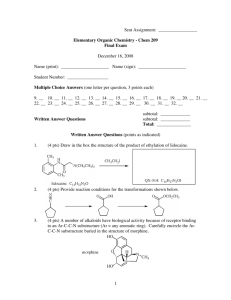
1) Molecular ion peak of 122 and 124 (3:1) 122-35 = 87/12 = 7 carbons 87-84 = 3 hydrogens C7H3Cl (2(7) + 2 – 3 – 1)/2 = 6 DOUS C6H15Cl 2(6) + 2 – 15 – 1 = -2/2 = -1 C5H11OCl (2(5) + 2 – 11 – 1)/2 = 0 C4H7O2Cl (2(4) + 2 – 7 – 1)/2 = 1 You have a carbonyl peak, so it’s the last formula, C4H7O2Cl And the peaks at 1000 and 1200 show a C-O so ester 1.310 triplet 3H 4.252 quartet 2H 4.062 singlet 2H The singlet shows that you have 1 carbon attached to the ester and the 2H shows this is where the Cl is And the triplet and the quartet represent the ethyl group attached to the carbonyl. The carbon between 160-180 is your carbonyl, so 3 other types of carbon. O Cl O 2) Molecular ion peak at 150 and 152 (1:1) 150 – 79 = 71/12 = 5 carbons 71 – 60 = 11 hydrogens C5H11Br 2(5) + 2 – 11 – 1 = 0 Nothing but C-H stretches so C5H11Br 3.417 triplet 2H 0.92 doublet 6H 1.86 sextet 2H 1.64 multiplet 1H 3.417 triplet 2H 0.92 doublet 6H 1.86 sextet 2H 1.64 multiplet 1H With 0 degrees of unsaturation we know this is a chain, so now what does it look like? We have a multiplet with an integration of 1H and a doublet with an integration of 6 so that tells us its an isopropyl group. R So that is 3 carbons and 7 hydrogens, leaving 2 carbons, 4 hydrogens and a bromine. 3.417 triplet 2H 0.92 doublet 6H 1.86 sextet 2H 1.64 multiplet 1H R So the sextet must be the carbon adjacent to the isopropyl group, because (1+1)(2+1)= 6 R So that leaves the triplet that integrates to 2 hydrogens, the triplet tells you it is next to a CH2 group and the 2 H integration tells you the bromine is attached b/c it should have 3 H otherwise. Br Br The carbon matches this structure b/c it tell syou there are 4 types of carbon. 3) Molecular ion peak of 73. 73 – 14 = 59/12 = 4 carbons 59 – 48 = 11 hydrogens so C4H11N 2(4) + 2 – 11 + 1 = 0 C3H7NO 2(3) + 2 – 7 + 1 = 2/2 = 1 Carbonyl peak tells you tha tit is C3H7NO also there are no N-H stretch so it’s a tertiary amide. 8.019 singlet 1H 2.970 singlet 3H 2.883 singlet 3H Singlet at 8.019 tells you it is attached to the carbonyl carbon. O So we have the following structure thus far: R H 8.019 singlet 1H R 2.970 singlet 3H 2.883 singlet 3H So two more singlets each representing 3 H so methyl groups. O H N N 4) Name the following compound: O Cl 2,2’-dimethylpropanoyl chloride 5) Name the following compound: N O N,N-dibenzylformamide H 6) Name the following compound: O O Isopropyl formate H Isopropyl methanoate 7) Name the following compound: O Benzoic propanoic anhydride O O 8) Draw 3-methylhexanoyl chlordie. O Cl 9) Draw isopropyl 3-ethyl-4-methylpentanoate O O 10) Draw N-tert-butyl-N-ethyl-2-isopropy-3,4dimethylpentanamide O N O O Cl Cl2 H2O, HCl H2NC(CH3)3 I2 excess O NaOH NHC(CH3)3 O O O CHI3 H3 O+ OH O O LDA, THF CH3CH2CH2Br LiAlH4 Br2 O H2O CH3COOH Br LiBr DMF Li2CO3 O OH O O O Br NaOCH3 Br2 CH3COOH LDA THF CH2CHCH2BR O O LDA THF CH3Br OH LiAl4 H2O OCH3 O CH3OH O O H+ HO DIBAL-H H2O CH3Li OH PCC O H2 O O CH3CH2Br O O Li CuI O Br Li2CO3 LiBr DMF (CH3CH2)2CuLi H2 O O O LDA, THF Br BrCH2CH2CH2CH2Br NaOCH2CH3 O O O O LDA, THF NaOCH2CH3 CH3CH2Br CH3CH2Br Br2 CH3COOH O O Li2CO3 LiBr DMF Br Br Br Br2 CH3COCl O FeBr3 AlCl3 OH HO O O Mg O TsOH MgBr O Br O O O MgBr O O H 2O H2 O O OH O PCC O O H3O+ O