Acids and Bases
advertisement

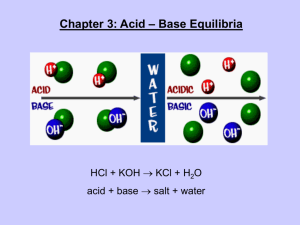
Acids and Bases PGCC CHM 101 Sinex General properties ACIDS • Taste sour • Turn litmus • React with active metals – Fe, Zn • • • • BASES Taste bitter Turn litmus Feel soapy or slippery (react with fats to make soap) React with acids • React with bases blue to red red to blue Definitions Arrehenius only in water • Acids – produce H+ • Bases - produce OH- • Acids – donate H+ Bronsted-Lowry • Bases – accept H+ any solvent Lewis • Acids – accept e- pair • Bases – donate e- pair used in organic chemistry, wider range of substances The hydrogen ion in aqueous solution H+ + H2O H3O+ (hydronium ion) Examples Arrhenius HCl Bronsted-Lowry HCl Lewis BF3 NaOH HCN :NH3 NH3 The Bronsted-Lowry Concept Conjugate pairs HCl Cl- NH4+ NH3 CH3COOH CH3COO- HNO3 NO3- How does a conjugate pair differ? H+ transfer Neutralization In general: Acid + Base Salt + Water All neutralization reactions are double displacement reactions. HCl + NaOH NaCl + HOH HCl + Mg(OH)2 H2SO4 + NaHCO3 Does pure water conduct electrical current? Water is a very, very, very weak electrolyte. H2O H+ + OH- How are (H+) and (OH-) related? (H+)(OH-) = 10-14 For pure water: (H+) = (OH-) = 10-7M This is neutrality and at 25oC is a pH = 7. water Let’s examine the behavior of an acid, HA, in aqueous solution. HA What happens to the HA molecules in solution? 100% dissociation of HA HA H+ Strong Acid AWould the solution be conductive? Partial dissociation of HA HA H+ Weak Acid AWould the solution be conductive? HA H+ + A- HA H+ A- Weak Acid At any one time, only a fraction of the molecules are dissociated. Strong and Weak Acids/Bases Strong acids/bases – 100% dissociation into ions HCl HNO3 H2SO4 NaOH KOH Weak acids/bases – partial dissociation, both ions and molecules CH3COOH NH3 acid rain (NOx, SOx) pH of 4.2 - 4.4 in Washington DC area pH 0-14 scale for the chemists 2 3 4 5 acidic (H+) > (OH-) normal rain (CO2) pH = 5.3 – 5.7 6 7 8 neutral @ 25oC (H+) = (OH-) distilled water fish populations drop off pH < 6 and to zero pH < 5 9 10 11 basic or alkaline (H+) < (OH-) natural waters pH = 6.5 - 8.5 12 pH of Rainwater across United States in 2001 You are here! Why is the eastern US more acidic? http://nadp.sws.uiuc.edu/isopleths What is acid rain? Dissolved carbon dioxide lowers the pH CO2 (g) + H2O H2CO3 H+ + HCO3- Atmospheric pollutants from combustion NO, NO2 + H2O … HNO3 SO2, SO3 + H2O … H2SO4 pH < 5.3 both strong acids Behavior of oxides in water– Group A basic 1A amphoteric acidic 3A 4A 5A 6A 7A 2A Group B 105 Db 107 Bh basic: Na2O + H2O 2NaOH (O-2 + H2O 2OH-) acidic: CO2 + H2O H2CO3 8A When life goes either way amphoteric (amphiprotic) substances Acting like a base + H+ H2CO3 accepts H+ HCO3- Acting like an acid - H+ CO3-2 donates H+ pH The biological view in the human body acidic 1 2 3 4 5 6 basic/alkaline 7 8 9 10 Tortora & Grabowski, Prin. of Anatomy & Physiology, 10th ed., Wiley (2003) 11 Does the pH influence the activity of an enzyme? Trypsin is a digestive enzyme. Where? Intestinal pH range 7.0-8.5 The amino acid glycine - amphoteric It’s an acid and a base! Gain of H+ Loss of H+ H3N+-CH2-COOH H2N-CH2-COOH H2N-CH2-COOChime structure The amino acid glycine - Zwitterion formation Transfer of H+ from carboxylic acid group to amine group. + A dipolar ion forms. H2N-CH2-COOH Chime structure H3N+-CH2-COOintramolecular acid-base reaction Show how water can be amphoteric. H2O + H+ - H+ Dilution water (solvent) solute moles of solute remain constant Vfinal diluted, Mfinal molesinitial = molesfinal Vinitial concentrated, Minitial adding water lowers the solute concentration Mfinal x Vfinal = Minitial x Vinitial Titration Calculation HCl + NaOH indicator NaCl + HOH A way to analyze solutions! at equivalence point: moleHCl = moleNaOH moles = M x VL Macid x Vinitial acid = Mbase x Vburet