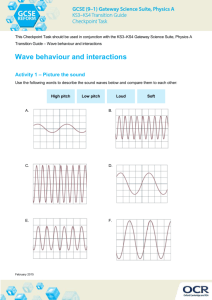
Sound, light, & color review answers
advertisement

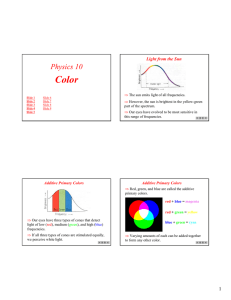
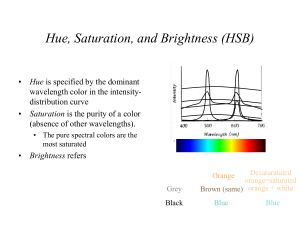
CP Physics Ms. Morrison Pitch: a person’s impression of a sound’s frequency Infrasonic: sounds below 20 Hz Ultrasonic: sounds above 20,000 Hz Compression: high pressure region of the sound wave, medium pushed together Rarefaction: low pressure region of the sound wave, medium spreads out Loudness: a person’s impression of sound intensity (proportional to wave’s amplitude) Forced vibration: vibration of an object caused by contact with another vibrating object Natural frequency: frequency at which minimum energy needed to make the material of the object vibrate and requires the least amount of energy to continue the vibrations Resonance: when the forced vibration of an object matches its natural frequency and causes a dramatic increase in the amplitude Beat: difference between the frequencies of two sounds heard together Longitudinal mechanical It must vibrate 20 – 20,000 Hz Speed of sound at 0 oC = 331 m/s Speed of sound at 20 oC = 343 m/s It increases – this is because the particles move faster and faster and transfer the wave energy more quickly. Solids Liquids Gases Decibel (dB) – logarithmic scale Swinging on a swing Musical instruments Opera singer shattering a glass Bridge vibrations A reflected sound wave Bats –echolocation Sonar Ultrasound Seeing unborn babies Examine heart Uses Doppler shift a. b. c. d. Brass: musician’s lips Reed: reed String: string against a sounding board Other (flutes): air column within the instrument Constructive interference – in phase – causes sound to become louder Destructive interference – out of phase – causes sound to become softer The speed of light is one million times faster than the speed of sound. 343 m/s vs. 3 x 108 m/s Transparent: material transmits light through it (can see through it) Translucent: material scatters the light it transmits (can see light but not through it, frosted windows) Opaque: material does not transmit light, cannot see through it or see light through it Quantum theory: energy given off as bundles of light energy called photons, explains how light interacts with matter Dual theory: light moves through space as a wave and interacts with matter as a particle Luminous: object gives off light, ex. Sun, lights, fire flies Illuminated: object reflects light, ex. Moon, mirror Photon: particle of light energy Radio waves Microwaves Infrared Visible light Ultraviolet X-rays Gamma Radio waves: radio and TV (longest λ, lowest frequency, and least energy) Microwaves: heating food Infrared: heat waves, used in heat lamps, night vision goggles, remotes Visible light: smallest part of EM spectrum, colors of light Ultraviolet: causes skin damage – sunburns, can kill bacteria (on fruits and vegetables) X-rays: used to see teeth and bones for medical purposes, airport security Gamma rays: used for radiation therapy, given off in nuclear explosions (shortest λ, highest frequency, most energy) Red Orange Yellow Green Blue Violet Its frequency Higher frequency = more energy Lower frequency = less energy Illuminated: moon Luminous: sun a. b. c. d. reflect: light bounces of boundary of a new medium Refract: light bends as it moves from one medium into another medium (air into water) Diffract: light passing through small opening spreads out on other side Interfere: passing through 2 slits interferes and produces light bands (constructive) and dark bands (destructive) Photosynthesis Absorb light to create electric current (solar calculators) Absorb light energy then release as color of light Light can only pass through in one direction – parallel to material’s polarizing axis Use polarized filter and turn 90o to see if light is blocked out Red Green Blue Cyan Magenta Yellow Two colors that produce white light when they are added together (primary light color + secondary light color) Red + Cyan Green + Magenta Blue + Yellow White light is seen when all colors reflected Black is seen when all colors absorbed Magenta light: red + blue Yellow light: red + green Cyan light: blue + green Light colors = additive process Pigments = subtractive process Pigments absorb some colors of light and reflect the remaining colors of light R G G B Color seen is BLACK B R G Y R Color seen is RED B R G Y C Color seen is GREEN B R G C Color seen is BLUE B R G G M Color seen is BLACK B t = 6.4 s v = 335 m/s d=? v=d t 335 = d 6.4 d = (335)(6.4) d = 2144 ÷ 2 d = 1072 m t = 7.2 s v = 340 m/s d=? v=d t 340 = d 7.2 d = (340)(7.2) d = 2448 m λ=? f = 375 Hz v = 530 m/s v = λf 530 = λ (375) 375 375 λ = 1.41 m vs = 25 m/s f = 325 Hz vd = 0 m/s v = 343 m/s f’ = ? f’ = f (v + vd) (v – vs) f’ = 325 (343 + 0) (343 – 25) f’ = 325 (1.08) f’ = 351 Hz vs = 0 m/s f = 288 Hz vd = 32 m/s v = 343 m/s f’ = ? f’ = f (v + vd) (v – vs) f’ = 288 (343 + 32) (343 – 0) f’ = 288 (1.09) f’ = 313.92 Hz 528 – 520 Hz = 8 Hz 10.5 yrs (365d/y)(24h/d)(3600 s/h) = 3.31128 x 108 s = t v = 3.0 x 108 m/s d=? v = d/t d = vt d = (3 x 108)(3.31128 x 108) d = 9.93 x 1016 m t = 180 s v = 3 x 108 m/s d=? v = d/t d = vt d = (3 x 108)(180) d = 5.4 x 1010 m d = 1.081 x 1011 m v = 3 x 108 m t=? v = d/t t = d/v t = 1.081 x1011 3 x 108 t = 360.33 s λ=? f = 94.1 MHz = 94.1 x 106 Hz v = 3 x 108 m/s v = λf 3 x 108 = λ (94.1 x 106) 9.41 x 106 94.1 x 106 λ = 3.19 m λ = 620 nm = 620 x 10-9 m v = 3 x 108 m/s f=? v = λf 3 x 108 = 620 x 10-9 f 620 x 10-9 620 x 10-9 f = 4.84 x 1014 Hz λ = 4.90 x 10-7 m v = 3 x 108 m/s f=? v = λf 3 x 108 = 4.9 x 10-7 f 4.9 x 10-7 4.9 x 10-7 f = 6.12 x 1014 Hz