Periodic table - St Bernard's College, Year 10 Core Science
advertisement

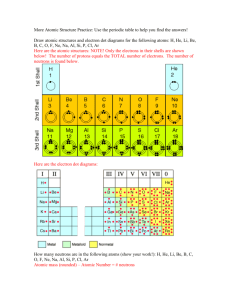
Structure and bonding Title • Atomic structure Aim • to draw and label the sub atomic particles of an atom. This dotted line is called The Staircase – it divides metals (left) from non-metals (right) 12 A column is called a 345678 Group. There are 8 A row is called a PERIOD VIDEO Quick quiz 1. What is found in the periodic table? 2. What are the rows called? 3. What are the columns called? 4. What element is found in: a) Group 8 period 2- Ne (neon) b) Group 6 period 2- O (oxygen) c) Group 1 period 4- Na (sodium) This dotted line is called The Staircase – it divides metals (left) from non-metals (right) Atoms ELEMENTS are made up of ATOMs. ATOMs are all made up of protons, neutrons and electrons. The Nucleus – this contains neutrons and protons Electrons – these orbit around the nucleus Different elements have different types of atoms. What the difference? Protons =2 Protons =4 Helium Beryllium Atomic Number • The atoms of same elements always contains the same number of protons. E.g. – Hydrogen atoms always contain 1 proton – Carbon atoms always contain 6 protons – Magnesium atoms always contain 12 protons • The number of protons is the atomic number. • It is the smaller of two number 12 C 6 There are 3 parts of an atom Type of sub atomic particle Relative charge Proton +1 (positive) Neutron 0 (neutral) Electron -1 (negative) 1/2000 of the size of a proton Activity How Many Protons? • Here is some data about elements taken from the Periodic Table. • What is the atomic number of the elements? 23 56 119 19 27 Na Fe Sn F Al 11 26 50 9 13 11 26 50 9 13 What element is it? • • • • • • • Atomic number. 12-Magnesium (Mg) 8- Oxygen (O) 6- Carbon (C) 20-Calcium (Ca) 3- Lithium (Li) 2- Helium (He) Atomic number • Atomic number = number of protons. • Atomic mass is the number of protons and neutrons added together 12 C 6 Atoms • In atoms number of protons = number of electrons So if the atomic number is 8 it will have, 8 protons and 8 electrons Why do atoms have charge? NO Activity • Complete as a class Atom Helium Nitrogen Oxygen Magnesium Sodium Symbol Protons Electrons + - Charge What is an isotope? • Different forms of the same element. • Same atomic number BUT a different number of neutrons and thus a different mass number. •99.76% of oxygen atoms have this configuration. 16 • Eg 8 O •0.04% have the mass number of 17 •0.20% have the mass number of 18 *Drawing Atoms* Aim: • To accurate diagrams of the electron structure of the first 20 elements. How Are Electrons Arranged? • Electrons are not evenly spread. • The exist in layers known as shells. • The arrangement of electrons in these shells is often called the electron configuration. 1st Shell 2nd Shell 3rd Shell 4th Shell Electron configuration • Shows how electrons are organised at an atomic level. Shell 1st 2nd Max numbers of electrons 2 8 3rd 4th 8 2 Which Shells Do Electrons go into? • Opposites attract. • Protons are positive (+) and electrons are negative (–) charged. • Electrons will occupy the shells nearest the nucleus unless these shells are already full. 1st Shell: Fills this first 2nd Shell: Fill this next 3rd Shell: And so on The Electrons in Carbon The Electrons in Silicon The Electrons in Neon Ions and Electron Structures 1. Ions are atoms that have either extra electrons added or electrons removed. e.g. Atoms Protons Electrons Lose 1 electron 1+ ion Gain 1 electron Protons Electrons So in ions the number of electrons no longer equals the number of protons 1- ion Protons Electrons The Electrons in a Sodium Ion In the sodium atom Atomic number = number of protons Number of electrons = 11 Na 23 = 11 Na 11 Na+ Bye! Electron lost Electron arrangement: 2.8.1 (Incomplete Shell) Electron arrangement: 2.8 (Full Shells) The Electrons in a Magnesium Ion In the magnesium atom Atomic number = number of protons Number of electrons = 12 24 = 12 Mg 12 Bye! Mg2+ Mg Bye! Electron arrangement = 2.8.2 (Incomplete shell) 2 electrons lost Electron arrangement 2.8 (Full Shells) Activity The Lithium Ion •How many electrons? 3 •How many electrons in the first shell? 2 •How many electrons in the second shell? 1 What electron arrangement? 7 Li 3 2.1 1st Shell = 2: full 2nd Shell = 1: not full How many electrons to lose? 1 Li Li+ New electron arrangement? Include a diagram 2.(0) Bye! Activity The Boron Ion •How many electrons? 5 •How many electrons in the first shell? 2 •How many electrons in the second shell? 3 What electron arrangement? 11 B 5 2.3 1st Shell = 2: full 2nd Shell = 3: not full How many electrons to lose? New electron arrangement? Bye! Bye! 3 2.(0) BB3+ Bye! Open to valence table 5.1 • Draw a before and after using a Bohr diagram showing the following atoms becoming ions. • • • • • • Sodium Oxygen Chlorine Calcium Nitrogen Aluminium Ionic Compounds • Once an ion has been created it will attract ions of the opposite charge they form ionic compounds. • For example magnesium + oxide = magnesium oxide • To write this as a chemical formula you need to take into account the ions charges (valencies) Magnesium has a positive charge of 2 while oxide has a negative charge of 2. Ionic Formulas Step 1 - write the name and symbols (the positive goes first) Magnesium Oxide 2+ 2Mg O Write the valencies above the symbols Step 2 - crisscross the valencies to get correct subscripts and leave out the + & - signs Magnesium Oxide 2+ 2Mg2 O2 Step 3 - write the correct formula with subscripts Mg2O2 = MgO (you can simplify this to MgO as each has 2 they cancel each other out) Ionic formulas • What happens when you try the following • Potassium + oxide • • • • Your new formula looks like this potassium oxide 1+ 2K2 O1 • K2O • When the charge is one only you can leave this out Ionic Formulas • Try these in your book (write ALL STEPS) 1. Calcium + oxide 2. Barium + oxide 3. Silver + chloride 4. Sodium + bromide 5. Lead + permanganate • Ionic formulas When they get more complex- • Zinc • 2+ • Zn + nitrate 1(NO3) • Zn(NO3)2 • • • • Potassium + sulfate 1+ 2K (SO4) K2SO4 Creating an ionic compound • Aim: to make the ionic compound Magnesium oxide and explain its bonding. • What you know about Magnesium and Oxygen. Colour, solid liquid or gas, electron structure. Use pictures Equipment & Safety Equipment • Goggles • 2cm strip of Mg • Heat proof mat • Tongs • Bunsen burner Safety • While heating Mg DO NOT look directly at it. • Ensure all heated material stays on heat proof mat • Ties in Observations 1. What did you see hear and smell? 2. What was the product formed? 3. What did it look like? Explaining observations 1. Why did you see or hear something? 2. How can you explain why magnesium and oxygen have combined? 3. Use the electron configuration. 4. How many atoms form the compound? 5. Does the properties change of Mg and O? 6. Why does this happen? 7. Write and equation to show this chemical reaction. Negative Ions. • These are ions formed by the atom gaining one or more electrons. Gain 1 e • They are called anions. This is because during It is nearly Ions usually electrolysis they move towards the anode. always have non-metal OUTER The charge number of atoms that on the ions is equal to theelectron gain electrons that the atom has gained.shells that Gain 3 eelectrons 3are either completely In equations the charge is usually shown above 2- or and to the right of the symbol. (E.g.. Ofull ). else empty Gain 2 e • • - 2- The Electrons in a Sulphide Ion. In the sulphur atom Atomic number = number of protons Number of electrons = 16 32 = 16 S2- S 2 electrons gained Electron arrangement: 2.8.6 (incomplete shell) Electron arrangement 2.8.8 (Full shells) S 16 The Electrons in a Fluoride Ion. In the fluorine atom Atomic number = number of protons Number of electrons = 9 19 =9 F F2- F 1 electron gained Electron arrangement: 2.8.7 (incomplete shell) Electron arrangement 2.8.8 (Full shells) 9 Activity The Oxide Ion 8 •How many electrons? •How many electrons in the first shell? 2 •How many electrons in the second shell? 6 What electron arrangement? 16 O 8 2.6 1st Shell = 2: full 2nd Shell = 6: not full How many electrons to gain? 2 OO2- New electron arrangement? 2.8 Drag the words at the top to their correct places in the sentences. Word check 1. Which of the following is not a subatomic particle? A. B. C. D. Proton. Isotope. Neutron. Electron. 2. The element Cobalt has a relative atomic mass of 59 and an atomic number of 27. Which of these is a true statement about each neutral cobalt atom? A. B. C. D. It contains 59 neutrons. It contains 27 electrons. It contains 32 protons. It contains equal numbers of neutrons and electrons. 3. The Periodic Table displays iron as shown below. This indicates that Fe atoms: 56 A. B. C. D. Fe contain 56 neutrons. 26 contain 30 electrons. contain 26 protons. contains more protons than neutrons. 4. Bromine consists of a mixture of two isotopes: Bromine-79 and Bromine-81 • Which of the following is true: 80 Br A. B. C. D. 35 Both isotopes contain 35 protons. Bromine 79 contains 46 neutrons. Bromine 81 contains 44 neutrons. Bromine-81 is more reactive than bromine79.