B10, B11. C11, C12, C13 Key Notes

Ecosystems & the Environment
Key Notes
B10, B11, C11, C12, C13
ENERGY FLOW IN ECOSYSTEMS
Ecosystem
• An ecosystem is a unit containing all of the organisms and their environment, interacting together, in a given area.
Food Chain
• A chart showing the flow of energy
(food) from one organism to the next, beginning with a producer.
Quaternary consumers
Tertiary consumers
Secondary consumers
Primary consumers
A terrestrial food chain
Primary producers
A marine food chain
Grasslands Ecosystem Food Chain
• Grass grasshoppers toad hognose snake hawk
Trophic Levels
• Trophic Level: the position an organism has in its food chain or food web.
Trophic Level
T1
T2
T3
T4
T5 producer primary consumer secondary consumer tertiary consumer quaternary consumer
For a food chain to function…
• In which trophic level would you want the largest number of organisms?
(think, pair, share)
Food Web
• Food Webs are a network of interconnected food chains showing the energy flow through part of an ecosystem.
• Lines are drawn as in a food chain, except it reflects the diversity in diet of each organism.
Humans
Baleen whales
Crab-eater seals
Smaller toothed whales
Leopard seals
Elephant seals
Sperm whales
Birds Fishes Squids
Carnivorous plankton
Copepods
Euphausids
(krill)
Phytoplankton
Make your own food web
• Use the following website (link on Haiku also) to make 1 or 2 food webs:
• Online Build a Food Web http://coolclassroom.org/cool_windows/home.html
• Then you can play the food web game with your partner.
https://www.brainpop.com/games/foodfight/
• With your partner, make a food chain/web based on what one you ate for dinner last night.
Energy in Ecosystems
• The SUN is the principle source of energy input to biological systems.
In a food chain…
• Producers are organisms that make their own organic nutrients, usually using energy from sunlight, through photosynthesis.
C
C
C
C C
C
• When that chemical energy (carbs, lipids, or protein) is digested, absorbed, and assimilated, it passes from one trophic level to the next.
T2: Horse
Energy
Flow
T1: Grass
C
C
C
C C
C
• Consumers are organisms that that get their energy by feeding on other organisms.
– Herbivores are animals that get their energy by eating plants.
– Carnivores are animals that get their energy by eating other animals.
• Decomposers are organisms that get their energy from dead or waste organic matter.
Energy Flow
• Energy is lost at each trophic level.
…in the form of: heat loss during cell respiration, organic materials not consumed or digested
• Energy transformations are never 100% efficient.
– Often only
10%
of energy flows from one level to the next!
• Animals lose heat as they move around.
• Warm-blooded animals use a lot of energy to heat their bodies.
T4: Tertiary consumers
T3: Secondary consumers
T2: Primary consumers
T1: producers
Energy Pyramid
10 kJ
100 kJ
1,000 kJ
10,000 kJ
1,000,000 kJ of sunlight
Energy Pyramid
The Carbon Cycle
The Carbon Cycle
Carbon Dioxide
• CO
2 is formed:
– As a product of complete combustion of carbon containing substances
• such as fossil fuels: coal, oil, natural gas
• such as organic wastes: burning wood, plants, animals
– As a product of respiration
• Can you write the equation?
– As a product of the reaction between an acid and a carbonate
• Can you write the equation for the reaction between hydrochloric acid and calcium carbonate?
– As a product of thermal decomposition
Thermal Decomposition
• Thermal decomposition is when heating causes chemical compounds to break apart.
• Carbonates (except those of Na and K) decompose to the oxide and carbon dioxide.
• CuCO
3
(s) + heat CuO (s) + CO
2
(g)
• (See chemistry textbook pg. 189)
Human Impact
• How does deforestation (cutting down forests) affect
CO
2 and O
2 concentrations in the atmosphere?
• How does the combustion of fossil fuels affect CO
2 and O
2 concentrations in the atmosphere?
Greenhouse Gasses
• Some gasses, such as CO
2 and methane, can build up in the earth’s atmosphere, trapping some wavelengths of radiation from the sun.
• These gasses keep the earth warm, like a greenhouse.
Global Warming
• The enhanced greenhouse effect is known as
“global warming”
• Why this trend?
• Industrialization
Air “pollution”
• Air can be polluted by many things.
• Greenhouse gasses (like CO
2 and methane) pollute the air because they build up in the atmosphere, contributing to global warming.
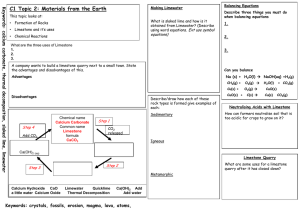
Making Lime from Limestone
• Let’s go through the steps of how lime is made from limestone.
• Take notes on your help sheet in your journals as we go.
• CaCO
3
(s) + heat CaO (s) + CO
2
(g)
• Calcium carbonate calcium oxide + carbon dioxide
• Limestone lime + carbon dioxide
• CaO (s) + H
2
O (l) Ca(OH)
2
(s)
• Calcium oxide + water calcium hydroxide
• Lime + water slaked lime
Making Lime from Limestone
• The series of 3 video clips on Hai-goda will demonstrate how a lime kiln is used to turn limestone into lime.
• Read page 240-241 in your textbook and make a diagram of the lime kiln and add to your notes page.
Online
• Go through the Limestone- GCSE review link on Hai-goda to be sure you’ve got all of the details about making lime from limestone!
Lime Uses
• What is the formula for slaked lime? Is it an acid or a base?
• Lime is used to treat acidic soil and to neutralize industrial waste products.
• Cool fact! Limewater is a weak solution of calcium hydroxide (slaked lime) that is used as the test for CO
2 gas.
Neutralizing Vinegar Lab
• Read the background, preparation, and procedure on the Neutralizing Vinegar with slaked lime Lab.
• Create a data table as shown in #2 of the preparation section on the back of your lab handout.
• It may take 2 trials before you get good data for your lab.
Neutralizing Vinegar
• Record your observations carefully—you will be graphing your data as part of your analysis.
• When you finish, clean up and work on the
Analyzing your results and Questions.
• These will be turned in for a grade!
AIR & WATER
Rust Lab
• Let’s set up the rust lab.
• One group at your lab bench will be using galvanized nails, and the other group at your bench will be using regular nails.
What is air made of?
• Think about the air around you (you know, that stuff that you breathe in and out about
12 times a minute). What is the composition of the air in our atmosphere?
• Read and take notes on p. 210 – 211 about the composition of air.
Separation of Liquid Air
• Air is a mixture of gases.
• Because several gasses are useful to us, we must separate them from the rest of the gases in the air.
• Read Page 212 and create a flow chart that details the steps of the fractional distillation of liquid air.
– Each step should be in your own words!!
– List the uses of each component of liquid air
Air Pollution
• Today you will be taking on the role of a scientist/conservationist who is responsible for informing the citizens in our school about common pollutants! Here is your task…
Pollutant Research Assignment
• You must create a visual brochure after researching about the following pollutants. Your brochure can take any form (A3 size, A4 size, single-sided, folded, hand-drawn, computer done, etc.) but it must be a hard copy to be turned in at the start of class on day 6.
• The pollutants you must discuss are:
– Carbon Dioxide
– Carbon Monoxide
– Sulfur Dioxide
– Nitrogen Oxides
For each pollutant include…
• The name and chemical formula of your pollutant
• The source of your pollutant (Where does it come from?
Why does it form?)
• The adverse/harmful effects of your pollutant on health and man-made structures.
• What the public can do to help reduce the presence of this pollutant in the air.
• Chem textbook pg. 214 – 215 may be helpful
• You must keep a list of your sources/websites to turn in with your brochure. This does not need to be in MLA format. Just a link is ok.
Complete & Incomplete Combustion
• When a hydro-carbon fuel burns in the presence of oxygen, carbon dioxide and water are formed.
• Complete combustion produces CO
2
• Ex. C
3
H
8
+ 5O
2
3CO
2
+ 4 H
2
O
• Incomplete combustion when there is not enough air produces carbon monoxide (CO) which is deadly to animals!
• Ex. 2C
3
H
8
+ 7O
2
6CO + 8 H
2
O
Oxides of Nitrogen
• Reducing air pollution from car exhaust is hugely important.
• Catalytic converters are used (pg. 214 – 215)
• Video Clip – take notes!
• Catalysts like platinum, palladium, and rhodium SPEED up these reactions!
• In the first catalyst compartment:
2NO (g) N
2
(g) + O
2
(g)
• Then, in the second catalyst compartment:
2CO(g) + O
2
(g) 2CO
2
(g)
• The catalysts are coated on a ceramic honeycomb or beads… why?
Rusting Lab Review:
Input the results of your rusting lab into your data table.
What is required for rusting?
4Fe(s) + 3O
2
(g) + 4H
2
O(l) 2Fe
2
O
3
2H
2
O (s) iron + oxygen + water hydrated iron (III) oxide (rust)
How can rusting be prevented?
– Read pages 216 and 217 and compare your results / analysis questions to what the textbook explains.
– You should add summary notes about different methods for preventing rusting
% Oxygen in the Air Lab
• We are going to do a little experiment to help us determine the amount of oxygen in the air around us.
• Listen carefully to the instructions, then grab your supplies and carry out the experiment with your partner.
• When you finish your experiment, clean up and work on your calculations/analysis.
Lab Debrief
• Review your notes about the composition of air.
– How close was your experimental value for
% oxygen to the actual value?
– Why is oxygen such an important component of air? Hint: think about its uses!!
Water
• What is water used for? (pg. 218)
– At home…
– On farms…
– In industry…
– In power stations…
Chemical Test for Water
• What are chemical tests for the presence of water?
– If a liquid contains water, it will…
• Turn white anhydrous copper (II) sulfate blue
• Turn blue cobalt chloride paper pink
• What is a test for water purity?
– If a liquid is pure water, it will boil at 100 o C AND freeze at 0 o C.
The Water Cycle
Water Pollution
• Describe how can water get polluted, including by:
– Sewage
– Chemical waste
– Acid rain
• How is Acid Rain formed?
Acid Rain
Acid Rain
• What are the causes of acid rain?
• What are the effects on the environment of acid rain?
• How can acid rain be reduced?
Water
• What are the steps taken to purify the water you drink?
– Read pages 218 and 219.
• As you read, create an outline of the steps necessary to purify water in your science journal
– You must be able to describe each step of the process, so take the time to learn each one and describe the process in your own words!
• Draw a labeled diagram of a water purification plant.
Sulfur
• Sulfur can be converted into sulfuric acid.
• Sulfuric acid is used in making:
– Fertilizers like ammonium sulfate
– Paints
– Plastics
– Soaps & detergents
– Car batteries
The Contact Process
• Use your notes and pages 232-233 in your textbook to complete the diagram of the manufacturing of sulfuric acid in your journals.
• Make sure your notes have all relevant chemical equations!
Cool facts!
• The reaction in the converter is reversible, so the mixture is passed over four layers of catalysts to give the reactants more opportunities to react.
• The reaction in the converter is exothermic, but the catalyst will work best at temperatures around 450 o C, so cold water is piped around the converter to cool it off. The water becomes hot and the steam is used generate electricity!
• In the absorber, the sulfur trioxide is dissolved in the 98% H
2
SO
4 because if more water were used, a thick dangerous mist of acid would be created.
Making H
2
SO
4
• As you watch the video clip about making sulfuric acid through the Contact Process, add to your notes.
SOIL & FERTILIZERS
Manufacturing Ammonia
• Let’s watch a video!
• Read about the Haber Process on pages 226-
227.
– As you read, make notes in your science journal about this process. Be sure to include chemical equations for the reactions involved as you encounter them.
– After you finish reading, use your laptops to follow the two web-links on Haiku to help your understanding.
Improving Ammonia Yield
X
Y
Haber Process
• The Haber Process occurs at 450 o C and 200 atm.
• 450 o C gives a faster rate than 350 o C
• 200 atm is safer and costs less than 400 atm
• Although the yield is not high immediately, ammonia is constantly removed so more will formed and the unreacted gasses get recycled.
• http://www.bbc.co.uk/schools/gcsebitesize/sc ience/add_gateway_pre_2011/chemical/amm oniarev3.shtml
Making Ammonia in the Lab
• In addition to the Haber process, ammonia can be produced on a small scale in the lab.
– Heat ANY ammonia compound with a strong alkali
(base) and the base will displace the ammonia from its compound!
– Write and balance the following example: ammonium phosphate + sodium hydroxide sodium phosphate + water + ammonia
Why is Ammonia important?
• Ammonia is a vital component in fertilizers!
• Why do we need fertilizers that contain nitrogen, phosphorus, and potassium?
– See Chem textbook pages 228-229.
• Write a letter from the perspective of one plant to another about why you love N, P, and K. (Or write the letter to NPK themselves).
The Downside of Fertilizers
• Read “It’s not all good news” on page 229.
– How can the environment be impacted by fertilizer usage?
• Let’s watch a video!
The process of eutrophication
CONSERVATION
Conservation
• Need for conservation of species & habitats
• Impact of Deforestation
• Need for conservation of natural resources
(water and non-renewable materials including fossil fuels)
Deforestation
• Use this website http://wwf.panda.org/about_our_earth/deforest ation/ and become experts in the way deforestation effects these areas:
• Group 1: Extinction of plants & animals
• Group 2: Loss of soil
• Group 3: Flooding & landslides
• Group 4: CO
2 buildup
• Is the impact of deforestation severe enough to demand immediate attention? Why or why not?
• What actions could you take to help reduce deforestation?
• Choose 2 actions that you will take, and explain how you will do them.
Conservation Day 2
• Watch planet earth video
Conservation Day 3
• Write an editorial to your local newspaper, persuading readers to take an active role in conserving EITHER:
– Species
– Habitats of species
– Forests
– Water
– Non-renewable materials like fossil fuels
• Research and add 3 specific points of research to your answer.
Conservation HW
• Day 1:
– Pg. 271 Q’s 1 – 2
– Read & take notes on pg. 279 – 283
• Day 2:
– Read & take notes on pg. 284 – 287
– Q’s 1 – 6 on pg. 286
• Day 3:
– Pg. 289 Q’s 1 & 2