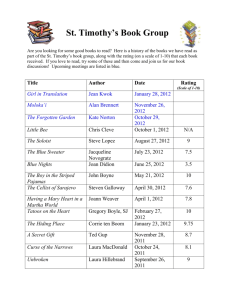
MENA3100
advertisement

MENA3100
1st lecture
General information, what to learn and
some repetition of crystallography
19/1-10
MENA3100
Student contact information
Name
19/1-10
E-mail
Phone
MENA3100
Who is involved?
•
Anette E. Gunnæs: eleonora(at)fys.uio.no, 91514080 (General, TEM, ED)
•
•
•
•
•
•
•
•
•
•
Johan Taftø: johan.tafto(at)fys.uio.no (waves optics, TEM, EELS)
Ole Bjørn Karlsen: obkarlsen(at)fys.uio.no (OM, XRD)
Sissel Jørgensen: sissel.jorgensen(at)kjemi.uio.no (EDS, XPS)
Spyros Diplas: spyros.diplas(at)smn.uio.no (XPS)
Klaus Magnus Johansen:k.m.h.johansen(at)smn.uio.no (SIMS)
Terje Finnstad: terje.finnstad(at)fys.uio.no (SPM)
Oddvar Dyrlie: oddvar.dyrlie(at)kjemi.uio.no (SPM)
Magnus Sørby: magnus.sorby(at)IFE.no (ND)
Geir Helgesen: geir.helgesen(at)IFE.no (ND)
Truls Norby: (SEM)
19/1-10
MENA3100
General information
•
Lectures
– Based on D. Brandon and W. D. Kaplan "Microstructural characterization of
materials". Second edition, published by Wiley, 2008.
– The curriculum is made up of the whole book, except these chapters: 3.4.3,
3.4.4, 3.4.6, 7.3 and 9.
– Some parts of the Brandon and Kaplan book will be regarded as self study
material
– Lecture notes that are made available on the course webpage are also
considered to be part of the curriculum.
•
Project work
–
–
–
–
•
Projects will be announced by the end of January/beginning of February
Three students will work together, rank projects with 1st-3rd priority
Written report, oral presentation and individual examination
Counts 40 % of final grade
Laboratories
– Four groups: A, B, C, D (4-5 students in one group)
– Individual reports
– All reports have to be evaluated and found ok before final written exam
19/1-10
MENA3100
Laboratory groups
A
C
(Tue. 14.15- )
B
(Tue. 16.15- )
(Wed. 10.15- )
D
(Wed. 12.15- )
A trip to IFE, Kjeller has been scheduled to
Wednesday 17th of February!
19/1-10
MENA3100
What to learn about
•
Imaging/microscopy
•
– Optical
– Electron
Spectroscopy
– EDS
• X-rays
– EELS
• SEM
• STEM
• TEM
• Electrons
– XPS, AES
– Scanning probe
• Electrons (surface)
• AFM
• STM
– SIMS
• Ions
Different imaging modes.
•
Diffraction
– X-rays
– Electrons
Mapping of elements or
chemical states of elements.
•
–
–
–
–
• ED in TEM and EBSD in SEM
– Neutrons
The same basic theory for all
waves.
19/1-10
Sample preparation
MENA3100
Mechanical grinding/polishing
Chemical polishing/etching
Ion bombardment
Crunching etc……
Probes used
•
•
Visible light
– Optical microscopy (OM)
•
X-ray
– X-ray diffraction (XD)
– X-ray photo electron spectroscopy
(XPS)
•
Neutron
– Neutron diffraction (ND)
•
Ion
– Secondary ion mass spectrometry
(SIMS)
– Cleaning, cutting and thinning
samples
19/1-10
MENA3100
Electron
– Scanning electron microscopy
(SEM)
– Transmission electron microscopy
(TEM)
– Electron holography (EH)
– Electron diffraction (ED)
– Electron energy loss spectroscopy
(EELS)
– Energy dispersive x-ray
spectroscopy (EDS)
– Auger electron spectroscopy
(AES)
Basic principles, electron probe
Electron
Auger electron or
x-ray
Valence
M
M
3d6
3p4
3d4
2p2
Electron
shell
L
3s2
2
2p4
3p
2s2
K
L
1s2
K
Secondary electron
19/1-10
Characteristic x-ray emitted or Auger
electron ejected after relaxation of inner
state.
Low energy photons (cathodoluminescence)
when relaxation of outer stat.
MENA3100
Basic principles, x-ray probe
X-ray
Auger electron
Valence
Secondary x-rays
M
M
Electron
shell
L
K
L
K
Photo electron
19/1-10
Characteristic x-ray emitted or Auger
electron ejected after relaxation of inner
state.
Low energy photons (cathodoluminescence)
when relaxation of outer stat.
MENA3100
Basic principles
X-rays
Electrons
(SEM)
BSE
X-rays (EDS)
SE
AE
(XPS)
Ions
(XD)
X-rays
Ions (SIMS)
AE PE
(Also used for
cleaning/thinning/cutting
samples)
SE
E=Eo
E<Eo
(EELS)
(TEM and ED)
19/1-10
You will learn about:
- the equipment
-imaging
-diffraction
-the probability for different events to happen
-energy related effects
-element related effects
-etc., etc., etc……..
MENA3100
Introduction to crystallography
We divide materials into two categories:
– Amorphous materials
• The atoms are ”randomly” distributed in space
• Not quite true, there is short range order
• Examples: glass, polystyrene (isopor)
– Crystalline materials
• The atoms are perfectly ordered
• Short range and long range order
• Deviations from the perfect order are important
19/1-10
MENA3100
Introduction to crystallography
19/1-10
MENA3100
Basic aspects of crystallography
• Crystallography describes and characterise the structure of crystals
• Basic concept is symmetry
• Translational symmetry: if you are standing at one point in a crystal,
and move a distance (vector) a the crystal will look exactly the same
as where you started.
2D
b
a
a
a
a
1D
a
19/1-10
a
a
MENA3100
a
The lattice
•
described as a set of mathematical points in space
– each of these points represents one or a group of atoms, basis
Basis
a
a
+ Lattice
a
= crystal structure
a
19/1-10
a
a
MENA3100
Axial systems
The point lattices can be described by
7 axial systems (coordinate systems)
z
c
β
α
γ
a
x
19/1-10
b
y
Axial system
Axes
Angles
Triclinic
a≠b≠c
α≠β≠γ≠90o
Monoclinic
a≠b≠c
α=γ=90o ≠ β
Orthorombic
a≠b≠c
α= β=γ=90o
Tetragonal
a=b≠c
α= β=γ=90o
Cubic
a=b=c
α= β=γ=90o
Hexagonal
a1=a2=a3≠c
α= β=90o
γ=120o
Rhombohedral
a=b=c
α= β=γ ≠ 90o
MENA3100
Bravais lattice
The point lattices can be described
by 14 different Bravais lattices
Hermann and Mauguin symboler:
P
(primitiv)
F
(face centred)
I
(body centred)
A, B, C (bace or end centred)
R
(rhombohedral)
19/1-10
MENA3100
Unit cell
•
The crystal structure is described by specifying a repeating element
and its translational periodicity
– The repeating element (usually consisting of many atoms) is replaced by a
lattice point and all lattice points have the same atomic environments.
– The unit cells are the smallest building blocks.
– A primitive unit cell has only one lattice point in the unit cell.
Repeating element, basis
c
α
β
γ
b
Lattice point
a
19/1-10
MENA3100
Exaples of materials with a face
centered cubic lattice
Copper
19/1-10
MENA3100
Exaples of materials with a face
centered cubic lattice
Silicon
19/1-10
MENA3100
Exaples of materials with a face
centered cubic lattice
ZnS
19/1-10
MENA3100
What about other symmetry elements?
• We have discussed translational symmetry, but there are also other
important symmetry operations:
–
–
–
–
–
Mirror planes
Rotation axes
Inversion
Screw axes
Glide planes
• The combination of these symmetry operations with the Bravais
lattices give the 230 space groups
19/1-10
MENA3100
Space groups
•
Crystals can be classified according to
230 space groups.
•
Details about crystal description can
be found in International Tables for
Crystallography.
– Criteria for filling Bravais point
lattice with atoms.
•
•
Structural data for known crystalline
phases are available in books like
“Pearson’s handbook of
crystallographic data….” but also
electronically in databases like “Find
it”.
•
Pearson symbol like cF4 indicate the
axial system (cubic), centering of the
lattice (face) and number of atoms in
the unit cell of a phase (like Cu).
A space group can be referred to by a
number or the space group symbol
(ex. Fm-3m is nr. 225)
19/1-10
MENA3100
Lattice planes
•
Miller indexing system
–
–
–
z
Crystals are described in the axial
system of their unit cell
c/l
Miller indices (hkl) of a plane is found
from the interception of the plane with
the unit cell axis (a/h, b/k, c/l).
a/h
0
b/k
y
x
Z
The reciprocal of the interceptions are
rationalized if necessary to avoid
fraction numbers of (h k l) and 1/∞ = 0
(110)
Y
–
Planes are often described by their
normal
Z
Z
(001)
X
–
–
(111)
(010)
(hkl) one single set of parallel planes
{hkl} equivalent planes
Y
Y
(100)
X
19/1-10
MENA3100
X
Directions
•
•
wc
The indices of directions (u, v and w)
can be found from the components of
the vector in the axial system a, b, c.
z
[uvw]
c
The indices are scaled so that all are
integers and as small as possible
ua
a
b
vb
y
x
•
Notation
–
–
•
[uvw] one single direction or zone axis
<uvw> geometrical equivalent
directions
[hkl] is normal to the (hkl) plane in
cubic axial systems
19/1-10
Zone axis [uvw]
(hkl)
uh+vk+wl= 0
MENA3100
Reciprocal vectors, planar distances
–The normal of a plane is given by
the vector:
•
g hkl h a * k b* l c *
The reciprocal lattice is defined by
the vectors :
a * (b c ) / V
b * (c a ) / V
–Planar distance between the planes
{hkl} is given by:
d hkl 1 / g hkl
c* (a b) / V
a*=(bcsinα)/V
b*=(casinβ)/V
–Planar distance (d-value) between
planes {hkl} in a cubic crystal with
lattice parameter a:
d hkl
19/1-10
a
h2 k 2 l 2
MENA3100
c*=(absinγ)/V