口試投影片
advertisement

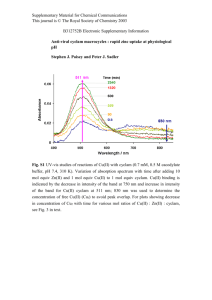
Synthesis and Characterization of Aminodipyridylphosphine Oxide Iron(II) Complexes. Catalytic Application on Microwave-Assisted Amidation of Aldehydes 學 生: 蔡俊偉 指導教授: 于淑君 博士 2013 / 07 /25 1 Green Chemistry 能源的綠色化 提升能源的效率 如微波 原料的綠色化 無毒無害的原料 可再生資源 化學反應的綠色化 原子經濟反應 觸媒的綠色化 無毒無害的觸媒 產品的綠色化 環境友好產品 溶劑的綠色化 無毒無害的溶劑 Chahbane, N.; Popescue, D., L.; Mitchell, D., A.; Chanda, A.; Lenoir, D.; Ryabov, A.D.; Schramm, K., W. and Collins, T., J. Green Chem. 2007, 9, 49–57. 2 Green Catalysts • Solid acid catalysts – Ex, Nafion-H, SO42-/ZrO2、SO42-/TiO2, … • HPA catalysts – H3PMo12O40, H4PW11VO40 • Zeolite catalysts – ZSM-5, X-type, Y-type • Metal catalysts – heterogeneous catalysis, homogeneous catalysis • Enzyme Sheldon, R., A.; Arends, I., W.,C., E.; and Hanefeld, U. (2007) Green Chemistry and Catalysis, 3 Wiley-VCH Verlag GmbH, Weinheim Application of Green Chemistry Epoxidation 物質 動物, 途徑 Lethal Dose, 50% (LD50) 1872 mg/ kg FeCl3.6H2O 大鼠, 口服 FeCl2.4H2O 大鼠,口服 1678 mg/ kg RuCl 3 Microwave assisted 210 mg/kg Anilkumar, G.; Bhor, S.; et. al. Tetrahedron Asymm., 2005 , 16, 3536–3561 Sonogashira coupling Conventional 大鼠, 口服 time yield (%) 18 h 80 25 min 97 Hasan, K.; Browne, N. and Kozak, C.,M. Green Chem., 2011, 13, 1230. 4 Phosphine Ligand Phosphines are electronically and sterically tunable. P P(Bu)3 O P O O P(OiPr)3 P P(Me)3 P P(o-tolyl)3 Chemical waste - water bloom Air/water sensitive and thermally unstable. Metal leaching. . Expensive. Kinzel, E. J. Chem. Soc. Chem. Commun. 1986 1098. 5 The Importance of Fe(II) • Iron is one of the most abundant metals on earth. (5.6% of earth’s crust. 4th most abundant element after oxygen, silicon, and aluminum. • Iron is environmentally friendly metal • Low toxic • In body play a important role to transport oxygen (woman 2.5g , man 4g) 60 mg/kg to iron poisoning 6 The Catalytic Applications of Fe(II) Ring Opening Reactions Diels-Alder Reaction Kharasch Reaction Sulfide Oxidations Cross-Coupling Reactions Aminochlorination Mukaiyama-aldol reaction Allylic Aminations Cycloadditions [2+1]-Cycloadditions [2+2]-Cycloadditions Baeyer-Villiger Reactions Amidation Reaction Acetalization 7 Amide Bond C. A. G. N. Montalbetti, V. Falque Tetrahedron , 2005, 61, 10827–10852 8 Application of Acyl Sulfonamides HCV NS5B polymerase allosteric inhibitors antitumor activity Navitoclax Hepatitis C Virus Non-structural protein 5B, NS5B 9 S. Jana, F. Hof. J. Org. Chem. 2011, 76, 3733–3741 Acyl Sulfonamides J. Chan,* K. D. Baucom, and J. A. Murry J. Am. Chem. Soc. 2007, , 129, 14106-14107 J. W. W. Chang and P. W. H. Chan*, et al. Angew. Chem. Int. Ed. 2008, 47, 1138-1140 J. W. W. Chang and P. W. H. Chan*,et.al. J. Org. Chem. 2011, 76, 4894-4904 10 Motivation Well-defined structure Iron is environmentally friendly metal Iron is less expensive than other transition metals. - Rh2(esp)2 $ 30172 USD/mol ReagentPlus® (Aldrich) - Ru(TTP)CO $ 20740 USD/mol reagent grade (Sigma-Aldrich) - FeCl2.4H2O $ 148 USD/mol reagent grade (Sigma-Aldrich) Using bipyridine ligand to replace phosphine ligand in organomatallic catalysis. Microwave to replace thermal energy 11 Synthesis of 4C-Ppy2 and (4C-Ppy2)2FeCl2 80 % 80 % 80 % IR (KBr) : py C C, C N Ring stretching = 1590(s), 1426(s) cm-1 12 IR Spectra of 4C-Ppy2 and (4C-Ppy2)2FeCl2 Wavenumber (cm-1) pyridine ring vibration ν(C = N) νC-C (Py ring) νC-N (Py ring) wavernumber a. b. Δν Ln- FeCl2a 1650 → 1668 18 PdCl2(2-pmOpe)2b 1594 → 1609 15 11C-Ppy2-Cu(OTf) 1570 → 1592 22 11C-Ppy2-NiBr2 1577 → 1592 15 11C-Ppy2-Pd(OAc)2 1574 → 1586 12 1574 → 1586 12 11C-Ppy2-MnCl2 b. Hahn, F. E.; Langehahn, V.; Lügger, T.; Pape, T.; Le Van, D. Angew. Chem. Int. Ed. 2005, 44, 3759-3763. Zerong, L.; Zhongquan, L.; Ning, M. and Biao, W. Bull. Korean Chem. Soc. 2011, 32, 2537-3543 Iron Complexes –Catalyzed Amidation Reactions of Aldehyde with PhINTs entry 1 2 3 4 catalytic FeCl2.4H2O FeCl3.6H2O FeCl2 + 4C-Ppy2 FeCl3 + 4C-Ppy2 Yield(%)a 20 15 90 76 a Yields were determined by H-NMR. Reaction conditions: aldehyde (1 equiv.), PhINTs (1.5 equiv.), catalyst (0.05 equiv.), solvent = 0.67 mL, 50 oC, 3 hr 14 AA Spectrum of [CH3(CH2)3N(H)P(O)(2-py)2]2FeCl2 Calculated base on chemical formula 2.48710-6 mol/mg Fe 1.46710-6 mol/mg Fe 0.7 0.6 y = 0.0345x + 0.0558 R² = 0.9966 0.5 0.4 ppm 吸收度 3 0.153 6 0.267 Series1 9 0.3 0.365 Linear (Series1) 0.2 12 0.483 15 0.562 experimental AA data mol/mg Fe 1 1.679 10-6 2 1.653 10-6 3 1.589 10-6 0.1 0 0 5 10 15 20 15 Colorimetry of [CH3(CH2)3N(H)P(O)(2-py)2]2FeCl2 Fe2+ + 3phen (phen)3Fe(II) N N 1,10-Phenanthroline (4C-Ppy2)2-FeCl2 (3) FeCl2·4H2O mol/mg mol/mg 理論值a 1.48 × 10-6 5.031 × 10-6 AA 實驗值 1.58 × 10-6 4.671 × 10-6 傳統比色分析 1.42× 10-6 3.783 × 10-6 80 % 81 % 二價鐵純度b max = 510nm a. Calculated base on chemical formula b. (傳統比色分析/ AA 實驗值) × 100% Harvey, J.; Aubrey, E.; John, A. Smart, Analytical Chemistry 1955, 27, 26-29. Visible spectrum of (phen)3Fe(II) 16 ESI-MS Spectrum of [CH3(CH2)3N(H)P(O)(2-py)2]2FeCl2 1020805_130705183651 #1236 RT: 2.52 AV: 1 NL: 1.48E5 T: ITMS + c ESI Full ms [150.00-2000.00] 641.52 100 95 [CH3(CH2)3N(H)P(O)(2-py)2]2FeCl2+ = 641 (m/z) [CH3(CH2)3N(H)P(O)(2-py)2]FeCl2+ = 366 (m/z) 90 Simulated MS Data 85 100.0 80 80.0 366.00 60.0 75 100.0 Simulated MS Data 40.0 368.04 70 20.0 364.04 65 0.0 362.00 366.00 368.00 372.00 374.00 643.16 642.16 40.0 366.53 Experimental MS Data 95 90 85 55 370.00 NL: 3.56E5 100 20.0 80 75 65 50 45 644.16 639.16 70 Relative Abundance 640.16 0.0 638.00 60 55 50 640.00 642.00 40 367.48 1020805_13070518 365 1 #1236 RT: 2.52 T: ITMS + c ESI Full ms [150.00-2000.00] 25 AV: 1 648.00 650.00 NL: 1.23E5 633.52 100 20 95 15 90 364.49 10 0 369.48 85 5 35 646.00 35 30 40 644.00 Experimental MS Data 368.47 45 80 365.49 359.34 359 360.39 360 361.42 361 362.49 362 363.51 363 370.47 364 365 366 367 368 369 370 371.58 371 372 373.38 373 374.47 75 70 374 m/z 65 Relative Abundance Relative Abundance AV: 1 60.0 369.04 364.00 1020805_130705183651 #755 RT: 1.69 T: ITMS + c ESI Full ms [150.00-2000.00] 60 641.14 80.0 367.02 366.53 30 60 55 642.53 50 643.53 45 40 35 30 25 20 25 644.56 15 639.49 10 675.42 5 645.61 640.49 676.45 637 638 647.50 646.55 0 639 640 20 641 642 643 644 645 646 647 m/z 15 10 379.02 5 435.35 573.21 491.34 550.28 613.35 722.47 752.26 0 350 400 450 500 550 600 650 m/z 700 750 824.49 861.35 889.44 800 850 900 938.06 950 1000.32 1000 17 EPR Spectrum of [CH3(CH2)3N(H)P(O)(2-py)2]2FeCl2 g = 2.199 g = 2.04 (radical) 77 k, MeOH 300 k, MeOH 18 Li Zhong-Fang, et al. Chinese Journal of Inoranic chemistry 2003, 19.7, 691-698. Synthesis of 11C-Ppy2 93 % 80 % Lin, Y.-Y; Tsai, S.-C.; Yu, S. J. J. Org. Chem. 2008, 73, 4920-4928. 19 Synthesis of (11C-Ppy2)2-FeCl2 IR (KBr) : py C C, C N Ring stretching = 1588(s), 1425 (s) cm-1 20 IR Spectra of 11C-Ppy2 and (11C-Ppy2)2-FeCl2 L1 L1-FeCl2 2 180 160 140 T( B%) 120 1575cm-1 100 1424cm-1 80 60 νC-C (Py ring) 40 1588cm-1 20 0 3000 νC-N (Py ring) 1426cm-1 2500 2000 1500 1000 A Wavenumber 21 AA Spectrum of [HO(CH2)11N(H)P(O)(2-py)2]2FeCl2 Calculated base on chemical Formula 1.106 10-6 mol/mg Fe 1.937 10-6 mol/mg Fe 0.7 0.6 y = 0.0345x + 0.0602 R² = 0.9935 experimental AA data 0.5 mol/mg Fe 0.4 0.3 1 1.324 10-6 2 1.413 10-6 0.2 0.1 0 0 2 4 6 8 10 12 14 16 22 Colorimetry of [HO(CH2)11N(H)P(O)(2-py)2]2FeCl2 Fe2+ + 3phen (phen)3Fe(II) N N 1,10-Phenanthroline (11C-Ppy2)2-FeCl2 (6) FeCl2·4H2O mol/mg mol/mg 理論值a 1.11 × 10-6 5.031 × 10-6 AA 實驗值 1.32 × 10-6 4.671 × 10-6 傳統比色分析 1.03× 10-6 3.783 × 10-6 78 % 81 % 二價鐵純度b max = 510nm a. Calculated base on chemical formula b. (傳統比色分析/ AA 實驗值) × 100% Harvey, J.; Aubrey, E.; John, A. Smart, Analytical Chemistry 1955, 27, 26-29. 23 Fe(II) Visible spectrum of (phen) 3 ESI-MS Spectrum of [HO(CH2)11N(H)P(O)(2-py)2]2FeCl2 1020704_130705183651 #494-617 RT: 0.84-1.11 AV:93 NL: 6.98E6 T: ITMS + c ESI Full ms [350.00-2000.00] 869.34 100 95 90 85 80 [HO3(CH2)11N(H)P(O)(2-py)2]2FeCl+ = 869 (m/z) 75 70 Simulated MS Data Relative Abundance 65 60 55 50 45 40 Experimental MS Data 35 30 25 20 15 390.36 417.31 10 444.27 5 372.41 0 350 400 450 577.20 480.28 515.20 547.56 500 550 600 778.51 630.10 664.33 700.38 728.42 650 700 m/z 750 800 833.38 850 914.03 965.38 942.37 900 950 1000 24 Optimization of Reaction Conditions entry 5 mol % 1 2 3 4 5 6 7 8 9b 10 PhINTs(equiv.) 2 2 2 0.5 1.5 1.5 1.5 1.5 1.5 1.5 b. no cat. temp. rt 40 40 40 40 40 40 40 40 rt hour 18 18 6 6 6 3 3 3 18 3 colvent CH2Cl2 CH2Cl2 CH2Cl2 CH2Cl2 CH2Cl2 CH2Cl2 CHCl3 CH3CN CHCl3 CHCl3 conv.% 87% 95% 85% 56% 88% 56% 89% 65% 30% 50% 25 Reaction Conditions Screening entry 1 2 3 4 5 6 7 8 9 solvent CHCl3 CH2Cl2 CH2Cl2 CH3CN CH2Cl2 : Toluene = 1 : 1 CHCl3 : Toluene = 2 : 1 MeOH DMSO [Bmim]Br time (hr) 3 3 6 3 3 3 3 3 3 Polarity (P*) yielda (%) 4.1 3.1 89 56 85 5.8 65 0.5 × 3.1 + 0.5 × 2.4 24 0.67 × 4.1 + 0.33 × 2.4 18 5.1 20 7.2 NA 13 a Yields were determined by H-NMR. Reaction conditions: aldehyde (1 equiv.), PhINTs (1.5 equiv.), catalyst (0.05 equiv.), solvent = 0.67 mL, 40 oC, 3 hr NA = not available P*AB=QAP*A+QBP*B [Bmim]PF6 dipole moment 14.89 D 26 (4C-Ppy2)2-Fe(II) Complex –Catalyzed Amidation Reactions of Aldehyde with PhINTs General reaction conditions: Aldehyde (1 equiv.), PhINTs (1.5 equiv.), Catalyst (0.05 equiv.) Solvent = 0.67 mL, 40 oC, 3 h. a Yields were determined by 1H-NMR. b. PhINTs = 2 equiv. c 18 h 27 Summary of Fe(II) catalytic Activity (4C-Ppy2)2FeCl2 Yield(%)a (11C-Ppy2)2FeCl2 Yield(%)a Entry (4C-Ppy2)2FeCl2 Yield(%)a (11C-Ppy2)2FeCl2 Yield(%)a 1 90 86 7 90 85 2 92 87 8 85 77 3 82 80 9 85 86 4 90 85 10 78 75 5 59 64b 57 11 67 84b 65 6 92 83 12 87 80 Entry Product Product General reaction conditions: Aldehyde (1 equiv.), PhINTs (1.5 equiv.), Catalyst (0.05 equiv.) Solvent = 0.67 mL, 40 oC, 3 h. a Yields were determined by 1H-NMR. b. PhINTs = 2.0 equiv. Proposed Mechanism of Amidation Reactions of Aldehyde with PhINTs byproduct J. W. W. Chang and P. W. H. Chan* J. Org. Chem. 2011, 76, 4894-4904 29 Microwave Assisted Amidation Reactions of Aldehyde with PhINTs entry time. ( s ) solvent Yield 1 2 M.W. power 300W 300W 5min 5min 29% 20% 3 4b 5 6 7 8 300W 300W 300W 600w 600w 600w 2min 5min 5min 7min 7min 7min 9c 10d 600w 600w 7min 7min CHCl3 CHCl3 + 3drop [Bmim][PF6] DMSO CHCl3 CHCl3 + 1 drop DMSO CHCl3 CHCl3 + 1 drop MeOH CHCl3 + 1 drop DI water CHCl3 CHCl3 a Yields No product 5% 11% 53%, 28% 22%, Dielectric loss 0.43 37.12 0.43+37.12 0.43+21.4 0.43+9.89 67% 87% were determined by H-NMR. Reaction conditions: aldehyde (1 equiv.), PhINTs (1.5 equiv.), catalyst (0.05 equiv.), solvent = 0.67 mL. b no cat. . c aldehyde = 0.4 mol PhINTs = 0.6 mol Fe cat. = 10 mol% solvent = 1 ml. d 30 solvent = 0.4 mL [Bmim][PF6] dipole moment 14.82 D Optimization of Reaction Conditions under Focused Microwave entry 1 2 3 4 5 6 7 8 9 10 11 12 13 14 M.W. power 50 W 100w 150W 150W 150W 150W 150W 150W 150W 150W 150W 200W 200W 250W 250W time. ( min ) 1 1 1 2 5 8 10 1 3 3 5 1 1 1 2 aldehyde ( M) 0.5 0.5 0.5 0.5 0.5 0.5 0.5 0.5M+1 drop BmimBr 0.5M+1 drop BmimBr 0.5M+2 drop BmimBr 0.5M+1 drop BmimBr 0.5 0.5 M+1 drop BmimBr 0.5 0.5 temp. (oC) 35 43 64 63 66 65 70 69 83 124 95 66 110 62 68 yield(%) 30% 40% 47% 52% 68% 77% 75% 76% 88% 67% 91% 57% 54% 63% 51% a Yields were determined by H-NMR. Reaction conditions: aldehyde = 0.2 mol (1 equiv.), PhINTs (1.5 equiv.), catalyst (0.05 equiv.), solvent 31 CHCl3 = 0.4 mL . (4C-Ppy2)2-Fe(II) Complex Catalyzed Amidation Reactions of Aldehyde with PhINTs a Yields were determined by H-NMR. Reaction conditions: aldehyde = 0.2 mol (1 equiv.), PhINTs (1.5 equiv.), catalyst (0.05 equiv.), solvent CHCl3 = 0.4 mL. b PhINTs (2.0 equiv) 32 Summary of Fe(II) Catalytic Activity under Focused Microwave (4C-Ppy2)2FeCl2 Yield(%)a (11C-Ppy2)2FeCl2 Yield(%)a Entry (4C-Ppy2)2FeCl2 Yield(%)a (11C-Ppy2)2FeCl2 Yield(%)a 1 90 80 7 82 73 2 93 83 8 86 72 3 76 64 9 71 63 4 86 78 10 75 75 5 54 50 11 80b 65b 6 90 80 12 84 68 Entry a Yields Product Product were determined by H-NMR. Reaction conditions: aldehyde = 0.2 mol (1 equiv.), PhINTs (1.5 equiv.), catalyst (0.05 equiv.), solvent CHCl3 = 0.4 mL. b PhINTs (2.0 equiv) 33 Synthesis of the RS-Au-L1-FeCl2 RS = CH3(CH2)7SH Au-RS RS-Au-L1 RS-Au-L1-FeCl2 34 IR Spectra of L1 , Au-L1 and Au-L1-FeCl2 Au-L1-FeCl2 Au-L1 L1 160 140 1585cm-1 120 100 1428cm-1 1575cm-1 1422cm-1 B T (%) 80 60 40 1575cm-1 20 0 1426cm-1 -20 3500 3000 2500 2000 A Wavenumber 1500 1000 35 TEM Image of RS-Au-L1-FeCl2 O (2-py)2P N H O (2-py)2P N H HO N P(2-py)2 SS S S Au S SSS O N P(2-py)2 H 9 RS-Au-L1 Fe Particle size distribution 2.68 ± 0.3 nm Particle size distribution 7.32 ± 1.2 nm Element Weight % Atomic % Fe 6.23 7.05 Cu 70.60 70.21 Au 19.67 6.31 36 RS-Au-L1-FeCl2 Complex –Catalyzed Amidation Reactions of Aldehyde with PhINTs 30 % Particle size distribution 6.87 ± 1.4 nm 37 Summary 1.We have successfully synthesized green catalysts [4C-Ppy2]2FeCl2、 [ 11C-Ppy2]2-FeCl2 .Their structures were studied by IR, ESI-MS, AA, EPR spectroscopies 2.We have successfully demonstrated the catalytic activity of the Fe(II) complexes for amidation reactions of aldehyde with PhINTs. 3. The Fe(II)-catalyzed amidation reactions of aldehyde with PhINTs can be further accelerated under microwave irradiation conditions. 38 39 4C-Ppy2-FeCl2 (3) 11C-Ppy2-FeCl2 (6) FeCl2.6H2O AA 理論值 1.48 × 10-6 mol/mg 1.11 × 10-6 mol/mg 5.031 × 10-6 mol/mg AA 實驗值 1.58 × 10-6 mol/mg 1.32 × 10-6 mol/mg 4.671 × 10-6 mol/mg 傳統比色分析 1.42× 10-6 mol/mg 1.03× 10-6 mol/mg 3.783 × 10-6 mol/mg 80 % 78 % 81 % 二價鐵純度a a. (傳統比色分析/ AA 實驗值) × 100% 40 瓦數 時間 焦耳 600 W 7 min 252000 5.5 倍 150 W 5 min 45000 31倍 130 W 180*60 1404000 傳統加熱 620 W 300 oC 6格 呈線性 1.3格 130W 40 oC 41 (11C-Ppy2)2-Fe(II) Complex Catalyzed Amidation Reactions of Aldehyde with PhINTs a Yields were determined by H-NMR. Reaction conditions: aldehyde = 0.2 mol (1 equiv.), PhINTs (1.5 equiv.), catalyst (0.05 equiv.), solvent CHCl3 = 0.4 mL. b PhINTs (2.0 equiv) 42 43 表3.12 用 [CH3(CH2)3N(H)P(O)(2-py)2]2FeCl2 (3) 鐵催化劑 對於PhINTs 競爭反應進行amidation 反應的催化結果 entry 1 2 3 4 5b 6c solvent thermal thermal microwave microwave thermal thermal CHCl3 CHCl3 + 1.1 mol DI water CHCl3 CHCl3 + 1.1 mol DI water CHCl3 + 1.1 mol DI water CHCl3+ 1.1 mol DI water yield (%)a 90 26 90 10 NA NA byproduct yield (%)a 32 82 33 76 45 80 1 a Yields were determined by H-NMR. Reaction conditions: aldehyde = 0.2 mol (1 equiv.), PhINTs (1.5 equiv.), catalyst (0.05 equiv.), solvent CHCl3 = 0.67 mL (thermal) 0.4 mL (MW).b no Fe cat. C no aldehyde 44 Reproposed Mechanism of Amidation Reactions of Aldehyde with PhINTs J. W. W. Chang and P. W. H. Chan* J. Org. Chem. 2011, 76, 4894-4904 45 non classed Staudinger reaction mechanism 46 Staudinger reaction mechanism 47 P P(Bu)3 25 mL 211.5 USD O P O O P(OiPr)3 100 mL 31.9 USD P P P(Me)3 P(o-tolyl)3 25 G 396 USD 10G 135.5USD 48 理論值 = 0.0002 mol*0.05 = 0.00001mol 0.00001*55.845/1 5.584 × 10-4g (in 1ml ) CHCl3 (ml) 4C-0.2M Aldehyde (mol) 1 Temp./Time AA (mg/L) 取0.1 ml 0.2 40oC/3h 4C-0.3M 1 0.3 4C-0.2M 1 4C-0.3M total Fe g 4.614 4.614 × 10-4g 40oC/3h 6.786 6.786 × 10-4g 0.2 Rt. / 10 min 4.005 4.005 × 10-4g 1 0.3 Rt. / 10 min 3.779 3.779 × 10-4g 11C-0.2M 1 0.2 40oC/3h 3.560 3.560 × 10-4g 11C-0.3M 1 0.3 40oC/3h 3.615 3.615 × 10-4g 49 Synthesis of Spacer-Linker L1 Lin, Y.-Y; Tsai, S.-C.; Yu, S. J. J. Org. Chem. 2008, 73, 4920-4928. 50 1 H NMR Spectra of Au NPs L1 and L1-Metal N-H O (2-py)2P N H O (2-py)2P N H HO N P(2-py)2 SS S S Au S SSS O N P(2-py)2 H 9 RS-Au-L1 Py N-H RS-Au-L1-FeCl2 d4-MeOH * 51 水 甲醇 大鼠, 口服 大鼠, 口服 Lethal Dose, 50% (LD50) >90,000mg/kg 5,628 mg/kg 氯化鈉 大鼠, 口服 3,000 mg/kg 維他命A 二氯化亞鐵 三氯化鐵 大鼠, 口服 大鼠, 口服 大鼠, 口服 2,000 mg/kg 1678mg /kg 1872mg/ kg 物質 動物, 途徑 釕鹽 52 (11C-Ppy2)2 -FeCl2 Complex –Catalyzed Amidation Reactions of Aldehyde with PhINTs General reaction conditions: Aldehyde (1 equiv.), PhINTs (1.5 equiv.), Catalyst (0.05 equiv.) Solvent = 0.67 mL, 40 oC, 3 h. a Yields were determined by 1H-NMR. b. PhINTs = 2.0 equiv. 53