Intro-Sulphur

Glycosides
1
• Definition:
Introduction
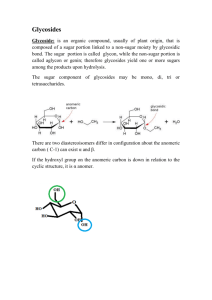
•Glycosides are non-reducing organic compounds that on hydrolysis with acids or enzymes yield:
–A sugar part (or glycone, formed of one or more sugar units).
–A non-sugar part (or aglycone, also called genin).
- The two parts are linked by glycosidic (acetal) linkage
- Their names frequently ends with -side
2
• Chemistry
• Glycosides are considered as acetals or sugar ethers where the –OH group of the anomeric carbon (i.e. at C-1 of the aldose sugar) is replaced by a moiety possessing a nucleophilic atom such as: O, S, N or C in the form of carbanion to give the corresponding O-, S-, N- or Cglycosides.
• Glycosides are non-reducing compounds (do not reduce
Fehling’s solution) unless the aglycone portion contains a reducing group (e.g. K-strophanthoside).
CHO
CH
2
OH
CH
2
OH
H
H OH
O
H H
H OH
OH
OH
HO H OH OH
OH O
H OH OH
OH
CH
2
OH or
-
Glucose Hemiacetal form
OH
CH
2
OH
O
OH
OH
H
OH
Hemiacetal form
H nucleophile
OH
CH
2
OH
O
OH
OH
H
OR
Acetal
(Glycoside)
3
• The common linkage between the sugar part and the aglycone is an oxygen linkage connecting the reducing group of a sugar and an alcoholic or a phenolic hydroxyl group of the aglycone to give O-glycosides.
• Since the sugars exist in isomeric - and -forms, both
- and -glycosides are theoretically possible, but most of the naturally occurring glycosides are of the -type.
• The most common sugar in glycosides is -D-glucose, but other sugars (like pentoses and deoxy-hexoses), or sugar derivatives (e.g. uronic acids such as glucuronic acid in glycyrrhizin) are also found.
The role of the sugar moiety in glycosides is :
To help in stabilization and solubilization of the molecule;
To carry the aglycone to the site of action.
4
Classification
They are classified according to:
1) Atom from the aglycone involved in the glycosidic linkage:
• Aglycone- O- Sugar O-glycosides
• Aglycone- C- Sugar C-glycosides
• Aglycone- S- Sugar S-glycosides
• Aglycone- N- Sugar N-glycosides
2) Number of sugars:
• One sugar monosides
• Two sugar Biosides
• Three sugars Triosides e.g. Salicin.
e.g. Diosmin.
e.g. Digoxin.
3) Nature of the glycoside:
• Primary glycosides: Originally present in the plant e.g.
Purpurea A
• Secondary glycosides: Resulted from removal of one sugar from the primary glycosides e.g. Digitoxin
5
4) Type of the glycosidic linkage:
- glycosides
- glycosides
5) Botanical source:
• Digitalis glycosides
• Senna glycosides.
6) Therapeutic use:
• Analgesic glycosides.
• Purgative glycosides.
• Cardiac glycosides.
7) Chemical nature of the aglycone:
• Flavonoidal glycosides.
• Steroidal glycosides.
• Anthraquinone glycosides.
• Cyanogenic glycoside
6
Physical Characters
:
Solids either amorphous or crystalline.
Non volatile.
Usually bitter in taste.
Soluble in water and polar organic solvents.
Reduce Fehling’s solutions only after hydrolysis.
7
Stability of Glycosides (reactions):
1- Effect of acid hydrolysis:
• Acids split sugars from the aglycones.
• The acetal linkage is more readily cleaved than the linkage between the individual sugars of the sugar chain.
• C-glycosides are resistant to acid hydrolysis.
8
2- Effect of alkaline hydrolysis:
• Hydrolysis of ester groups e.g. Lanatoside A to Purpurea A
• Opening of lactone rings e.g. Cardiac glycosides.
• Racemization of sugars
9
3- Enzymatic hydrolysis:
– Split the sugars stepwise starting from the terminal sugars.
– All plants producing glycosides have enzyme that can hydrolyze these glycosides.
– Enzymes are specific for the type of glycosidic linkages:
• Emulsin can hydrolyze - glycosides
• Invertase can hydrolyze - glycosides
• Myrosinase can hydrolyze S -glycosides.
10
Extraction and Isolation
o Because of the wide range of physical and chemical properties of glycosides and other constituents associated with them, no common general method for their isolation is recommended. o Water, methanol, water-ethanol and ethanol are the most common solvents for extraction of glycosides.
o Precautions before extraction
1) Deactivation of enzymes: o Drying for 15-30 min at 100 o C followed by slow drying at a low temperature.
o Dipping the fresh material into boiling water or boiling
alcohol for 10-20 min.
o Carrying out the extraction at very low temp.
o Freeze-drying of the plant material before extraction
(lyophilization).
o Carrying the extraction in the presence of (NH
4
)
2
SO
4
.
11
2) Maintenance of neutral conditions: o Neutral pH should be maintained before and during extraction because:
-Acidity may result in hydrolysis. This is overcome by addition of little CaCO
3
(cautiously).
- Mild alkalinity may sometimes produce racemization.
3) Defatting of fat-rich organs before extraction (e.g. seeds as in linseed) : o High amounts of lipids hinder glycoside extraction.
Defatting is usually carried out with petroleum ether (nhexane).
12
Alcoholic and Phenolic Glycosides
1- Salicin
Source:
Salix species (Willow bark).
Nature:
Primary alcoholic and Phenolic glycoside (monoside).
Uses:
Analgesic- Antipyretic- Antiinflammatory.
CH
2
OH
O
Saliretin
HOH
2
C
+
Glucose
Acid
CH
2
OH
Enzyme
O-glc
CH
2
OH
OH
Saligenin
(Salicyl alch.)
+
Glucose
13
2- Arbutin
Source: Uva Ursi (Bearberry leaves).
Nature: Primary Phenolic glycoside (monoside).
Uses: Diuretic- Bactericidal, UT infections, and skin
lightening preparations (topically, inhibition of melanin synthesis).
OH
OH
Hydrolysis
OH
Hydroquinone
+ Glucose
O-glc
Arbutin
14
Aldehydic Glycosides
1- Glucovanillin
Source:
Vanilla pods.
Uses:
Flavouring agent, and Spray reagent.
Glucovanillin Vanillin
CHO CHO
Enzymatic Hydrolysis
+ Glucose
OCH
3
OCH
3
O-glc OH
Green vanilla pods
Bitter in taste
Odourless
Brown vanilla pods
Sweet in taste
Vanilla odour
In green pods in fermented pods (brown, dry) 15
Commercial Preparation of Vanillin
Eugenol
(clove oil)
CH
2
-CH=CH
2
OCH
3
OH
KOH
Iso-eugenol
CH=CH-CH
3
OH
OCH
3
Oxidation
Vanillin
Guaiacol
(from wood, expec)
OCH
OH
CH=CH-CH
2
OH
3
Coniferin
(lignin of confers)
CHCl
3
+ NaOH
Reflux
Vanillin
H
2
SO
4
/K
2
Cr
2
O
7
Vanillin
OCH
3
O-glc
16
Most vanillin is now prepared from phenol or lignin (cheap sources)
Cyanogenic Glycosides
Cyanogenic glycosides (Cyanogentic or
Cyanophore Glycosides) are O-glycosides yielding
HCN gas on hydrolysis .
They are condensation products of HCN to a carbonyl compounds (Cyanohydrin).
R
R
C O
HCN
R
CN
Glycosylation
C
R
Unstable
OH
R
CN
C
R
Stable
O-Sug
17
1- Amygdalin
Source: bitter almond.
Structures: It is a bioside of mandelonitrile.
HCN +
Benzaldehyde
CHO
O H
CH
CN
Mandilonitril
(1-6)
linkage
O glc glc
CH
CN
Amygdalin
Amygdalase
O glc
CH
CN
Prunasin
Prunase
18
2- Linamarin
Source: Linseed (Linum usitatissimum,
Linaceae).
Structures:
It is the glycosidic derivative of the cyanohydrin of acetone.
H
3
C O glc
C
H
3
C CN
Uses of cyanophoric glycosides:
Linamarin has a molluscicidal activity.
Amygdalin is used for the preparation of
Benzaldehyde.
Cyanogenic glycosides have role in cancer treatment
(probable HCN toxicity).
19
Test for Cyanogenic Glycosides:
• Reduce plant material to small pieces and moisten with water.
• Incubate at temp. less than 45 0 C for 60 min. with the neck of the flask is stoppered and have suspended sodium picrate paper.
• The paper will turns brick red due to the release of HCN gas.
20
Thioglycoside
Glucosinolates- Sulphur Glycosides
• They are S-glycosides widely distributed in family
Cruciferae.
• Sinigrin: In seeds of Brassica nigra (black mustard).
• Sinalbin: In Seeds of Brassica alba (white mustard).
S-Glc
H
2
C C
H
C
H
2
C
N-O-SO
3
K
Allylisothiocyanate
(pungent, volatile)
Sinigrin hydrolysis myrosinase
H
2
C C
H
H
2
C N C S + glucose + KHSO
4
• Uses: Rubefacients, Counter irritants, and condiment.
Garlic
• It consists of the bulb of Allium sativum Fam. Liliaceae.
• The intact cells of garlic contain an odorless, sulfur- containing amino acid derivative (+)-S-allyl-L-cysteine sulfoxide , commonly known as alliin .
• Alliin is hydrolyzed by the effect of alliinase enzyme present in different cells after crushing into allicin (diallyl thiosulfinate).
• Allicin is responsible for the characteristic odor and flavor of garlic.
• Allicin is a potent antibacterial, antihyperlipidemic, and it inhibits platelet aggregation and enhances the blood fibrinolytic activity.
Alliin
O
S
COOH
H
NH
2
Alliinase
O
S
Allicin
S
+ H
2
O
22