1023-L03-070112
advertisement

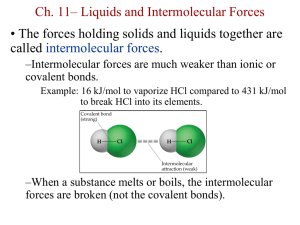
Intermolecular Forces and Liquids and Solids Chapter 12 1. See Table 12.3 in Chang. Note that glycerol has the highest viscosity of the liquids shown. Look up the structure and provide a reason why glycerol is more viscous than water. • Viscosity increases with the strength of intermolecular forces. • Glycerol and water are both polar compounds: dispersion forces and dipole-dipole forces. • Both are capable of hydrogen bonding, a special kind of dipoledipole force. • Difference in viscosity must be explained in terms of the number of hydrogen bonds that can be formed. 12.13 Arrange the following compounds in order of increasing boiling point: RbF, CO2, CH3OH, CH3Br. Explain your arrangement. Are any of the compounds ionic? If so, they will exhibit the stronger intermolecular forces. RuF Are any of the compounds nonpolar? If so, they exhibit the weakest intermolecular forces. CO 2 Are any compounds polar? If so, they will exhibit intermediate molecular forces. CH3OH, CH3Br Are any of the polar compounds capable of hydrogen bonding? If so, they will exhibit the stronger intermediate forces. CH OH 3 weaker stronger 12.19 These nonpolar molecules have the same number and type of atoms. Which one would you expect to have a higher boiling point? (Hint: Molecules that can be stacked together more easily have greater intermolecular attraction.) linear branched The molecule with the larger surface area has stronger van der Waals forces; for two molecules with the same molecular formula but different connectivities (structural isomers), the less compact molecule has the larger surface area. A linear isomer is less compact than a branched isomer. 12.15 Which member of each of these pairs of substances would you expect to have the highest boiling point: (a) O2 or N2, (b) SO2 or CO2, (c) HF or HI? 12.17 Explain in terms of intermolecular forces why (a) NH3 has a higher boiling point than CH4 and (b) KCl has a higher melting point than I2. Solids Crystalline—high order (regular repeating pattern) Amorphous—low order Attractions in Ionic Crystals In ionic crystals, ions pack themselves so as to maximize the attractions and minimize repulsions between the ions. Crystalline Solids basic repeating structural unit of a crystalline solid— unit cell spheres— lattice points At lattice points: • Atoms • Molecules • Ions 12.4 12.4 Shared by 8 unit cells Shared by 2 unit cells 12.4 12.4 1 atom/unit cell 2 atoms/unit cell 4 atoms/unit cell (8 x 1/8 = 1) (8 x 1/8 + 1 = 2) (8 x 1/8 + 6 x 1/2 = 4) 12.4 We can determine the empirical formula of an ionic solid by determining how many ions of each element fall within the unit cell. What are the empirical formulas for these ionic solids? (a) Green: chlorine; Gray: cesium (b) Yellow: sulfur; Gray: zinc (c) Green: calcium; Gray: fluorine (a) (b) CsCl ZnS (c) CaF2 Know diagonal of cube (c) in terms of r Know diagonal of one face (b) in terms of a If we can relate (c) to a then, we know (c) in terms of r 12.4 When silver crystallizes, it forms face-centered cubic cells. The unit cell edge length is 409 pm. Calculate the density of silver. d= m V V = a3 = (409 pm)3 = 6.83 x 10-23 cm3 4 atoms/unit cell in a face-centered cubic cell m = 4 Ag atoms x 1 mole Ag 107.9 g = 7.17 x 10-22 g x 23 6.022 x 10 atoms mole Ag 7.17 x 10-22 g m 3 = = 10.5 g/cm d= V 6.83 x 10-23 cm3 12.4