Chapter 1
advertisement

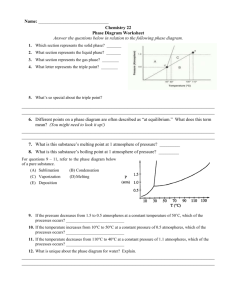
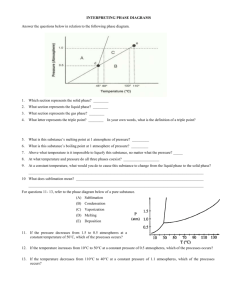
Chapter 8 Atom- the smallest particle of matter that has the properties of that matter boiling point- temperature where a liquid changes rapidly to a gas Not all of them are the same. Some boiling points (in degrees Fahrenheit) are: water: 212 tar: 572 propane: -45 ammonia: -28.1 jet fuel: 325 mercury: 675.1 olive oil: 570 chemical property- ability of a material to change its chemical makeup Some chemical properties are: Reactivity against other chemical substances Heat of combustion Enthalpy of formation Toxicity Chemical stability in a given environment Flammability Preferred oxidation state(s) Coordination number The chemical property of materials helps us identify them. We know how things behave when we make them go through a change, such as burning. How they react when we change them is their chemical property. A chemical property of ammonia is that when you mix it with bleach, toxic fumes are made. condensation- a change of state from a gas to a liquid as energy is removed Of course it’s a part of the water cycle, but did you remember that it’s what makes the water on the outside of drinks? conductivity- the ability to carry energy Copper, silver, and aluminum are commonly used as conductors of electricity. Did you know gold was a good conductor, too? That’s why it’s not a good idea for this person to smile in an electrical storm. density- a measure of the mass per unit volume of a substance Density is not the same as mass or volume. These two liter bottles have the same volume. Now fill one with soda and one with gold. The gold one would weigh way more because gold is more dense than the soda. The soda would weigh about five pounds. The gold one would weigh about 100 pounds. It means there are more gold atoms packed tighter together than the soda atoms. Density is a comparison of how much it weighs to how much space it takes up. deposition- the change of state from a gas to a solid Frost forming on plants and windows is a good example. It goes from water vapor to ice without going to liquid. evaporation- gradual vaporization that takes place at the surface of a liquid (going slowly from liquid to gas) Transpiration is the process of evaporation in plants. matter- anything that has mass and takes up space (volume) On the left are examples of things made of matter. We have to remember that those things are made up of these elements, though. All are examples of matter. melting- change of state from a solid to a liquid as energy is added melting point- temperature at which a substance changes from solid to liquid The melting points of different solids vary greatly. Carbon’s melting point is about 6381 degrees Fahrenheit. Iron’s point is about 2800. Tin’s point is about 450. (Butter, a sort of mixture, has a point of about 90.) molecule- two or more atoms joined by chemical bonds Here are two different views of a water molecule. physical property- a characteristic of matter that can be observed without changing matter into something new Here is a list of physical properties. You’re right; I don’t know what many of these are. •absorption •albedo •area •brittleness •boiling point •capacitance •color •concentration •density •dielectric •ductility •distribution •efficacy •electric charge •electrical conductivity •electrical impedance •electric field •electric potential •emission •flexibility •flow rate •fluidity •frequency •inductance •Intrinsic impedance •intensity •irradiance •length •location •luminance •luster •malleability •magnetic field •magnetic flux •mass •melting point •moment •momentum •permeability •permittivity •pressure •radiance •solubility •specific heat •resistivity •reflectivity •spin •strength •temperature •tension •thermal conductivity •velocity •viscosity •volume •wave impedance solubility- measure of how much of one substance can dissolve in another Bob found out the hard way that the solubility of a Twinkie is too low to make that Twinkiejuice drink he wanted so desperately to make. After a few days it dissolved- sort of. Either way, he should stick to Kool-Aid. It’s solubility is much better. states of matter- a physical form that matter takes; solid, liquid, and gas are the most common So which state is: ice? solid So which state is: soda? liquid So which state is: the helium in a balloon? So which state is: Jello? gas Trick question! It’s two: a solid inside a liquid! sublimation- the change of state from a solid to a gas Matter usually goes from solid to liquid to gas when energy is applied to it. Some solids can go from solid to gas without the liquid stage. A good example would be dry ice (carbon dioxide). thermal expansion- increase in size due to increase in temperature Thermal expansion is why we make sidewalks with little gaps between each piece. If there are no gaps, then the sidewalk expands when it gets really hot and cracks. vaporization- change of state from a liquid to a gas Evaporation is vaporization, but only on the surface of the liquid. Boiling is fast vaporization. The heat energy makes more than just the surface molecules vaporize. Much more of the liquid is turning to gas. Microscopic water droplets suspended in the air above a hot tea cup after that water vapor has sufficiently cooled and condensed. Water vapor behaves like a gas and is, thus, invisible, but the clouds of condensed water droplets refract and diffuse the sun light and so are visible. Quiz Time! atom, boiling point, chemical property, condensation, conductivity, density, deposition, evaporation, matter, melting Which word means the ability to carry energy? conductivity Quiz Time! atom, boiling point, chemical property, condensation, conductivity, density, deposition, evaporation, matter, melting Which word means the smallest particle of matter that has the properties of that matter? atom Quiz Time! atom, boiling point, chemical property, condensation, conductivity, density, deposition, evaporation, matter, melting Which word means anything that has mass and takes up space? matter Quiz Time! atom, boiling point, chemical property, condensation, conductivity, density, deposition, evaporation, matter, melting Which word means temperature at which a liquid rapidly changes to gas? boiling point Quiz Time! atom, boiling point, chemical property, condensation, conductivity, density, deposition, evaporation, matter, melting Which word means change of state from solid to liquid? melting Quiz Time! atom, boiling point, chemical property, condensation, conductivity, density, deposition, evaporation, matter, melting Which word means a characteristic of matter only observable when it is changed into something new? chemical property Quiz Time! atom, boiling point, chemical property, condensation, conductivity, density, deposition, evaporation, matter, melting Which word means a measure of the mass per volume of a substance (how heavy vs. how much space it takes up)? density Quiz Time! atom, boiling point, chemical property, condensation, conductivity, density, deposition, evaporation, matter, melting Which word means a change of state from gas to liquid? condensation Quiz Time! atom, boiling point, chemical property, condensation, conductivity, density, deposition, evaporation, matter, melting Which word means the change of state from a gas to a solid? deposition Quiz Time! atom, boiling point, chemical property, condensation, conductivity, density, deposition, evaporation, matter, melting Which word means slow vaporization on the surface of a liquid? evaporation Quiz Time! melting point, molecule, physical property, solubility, states of matter, sublimation, thermal expansion, vaporization Which word means two or more atoms joined by chemical bonds? molecule Quiz Time! melting point, molecule, physical property, solubility, states of matter, sublimation, thermal expansion, vaporization Which word means increase in size due to increase in temperature? thermal expansion Quiz Time! melting point, molecule, physical property, solubility, states of matter, sublimation, thermal expansion, vaporization Which word means the temperature where solid turns into liquid? melting point Quiz Time! melting point, molecule, physical property, solubility, states of matter, sublimation, thermal expansion, vaporization Which word means the change of state from a solid to a gas? sublimation Quiz Time! melting point, molecule, physical property, solubility, states of matter, sublimation, thermal expansion, vaporization Which word means a physical form that matter takes? states of matter Quiz Time! melting point, molecule, physical property, solubility, states of matter, sublimation, thermal expansion, vaporization Which word means it can be observed without changing the matter into something new? physical property Quiz Time! melting point, molecule, physical property, solubility, states of matter, sublimation, thermal expansion, vaporization Which word means a change of state from liquid to gas? vaporization Quiz Time! melting point, molecule, physical property, solubility, states of matter, sublimation, thermal expansion, vaporization Which word means how much of one substance can dissolve in another? solubility