EXPERIMENTAL STUDIES

EXPERIMENTAL STUDIES
FRAMEWORK:
Introduction
Types of experimental studies
Randomized control trial
– Types of randomized controlled trial (RCT)
– Phases of clinical trial
– Design and conduct of an RCT
– General plan and protocol of an RCT
– Study population
– Randomization
– Blinding
– Sample size
– Trial designs
– Expressing results of a randomized control trial
– Problems in conduct of an RCT
– Reporting and interpretation
Field trials
Community trials
Quasi-experimental studies
INTRODUCTION
The idea of epidemiology has its origins almost 2000 years back- Hippocrates – environmental factors can influence the occurrence of disease
Formal beginnings of epidemiology- until 19 th century- John
Snow- risk of cholera in London was associated with source of drinking water supply
“The study of distribution and determinants of health-related states or events in specified populations, and the application of this study to the control of health problems.” (John Last, 1995)
Epidemiologic studies -observational or experimental
Observational studies allow nature to take its course: the investigator measures but does not intervene
Experimental studies: investigator intervenes.
EPIDEMIOLOGIC STUDY DESIGNS
TYPE OF STUDY ALTERNATE
NAME
UNIT OF
STUDY
OBSERVATIONAL Descriptive
Analytical
Ecological
Cross-sectional
Case control
Cohort
EXPERIMENTAL Randomized controlled studies
Field trials
•Co relational
•Prevalence
•Case reference
•Follow-up
Intervention studies
Clinical trials
•Population
•Individuals
•Individuals
• Individuals
Patients
Healthy people
Community trials Community intervention studies
Communities
WHY THE NEED FOR EXPERIMENTAL STUDIES?
Because of variability
We wouldn’t need a science of experimental design if
– If all units (patients; communities) were identical
– If all units responded identically to treatments
We need experimental design to control variability so that treatment effects can be identified
The idea of controlling variability through design has a long history
In 1747 Sir James Lind’s studies of scurvy
“Their cases were as similar as I could have them. They all in general had putrid gums, spots and lassitude, with weakness of their knees. They lay together on one place … and had one diet common to all….
Lind then assigned six different treatments to groups of patients
In 1904, Karl Pearson suggested matching and alternation in typhoid trials
Randomization for experimental studies was established by R.A.
Fisher in 1923
In 1937, Sir Bradford Hill advocated alternation of patients in trials rather than randomization
The first modern randomized clinical trial in medicine is usually considered to be the trial of streptomycin for treating tuberculosis
It was conducted by the British Medical Research Council in
1946 and reported in 1948
RANDOMIZED CONTROLLED TRIAL
A randomized controlled trial is an epidemiological experiment to study a new preventive or therapeutic regimen.
Subjects in a population are randomly allocated to groups, usually called treatment and control groups
Results are assessed by comparing the outcome in the two or more groups.
The outcome of interest will vary but may be the development of new disease or recovery from the established disease.
For example, a trial conducted in Bangladesh (Molla et al, 1985) compared the use of rice-based and glucose based oral rehydration solution in 342 patients with acute watery diarrhoea during an epidemic of cholera in Bangladesh.
The study showed that the glucose component of the solution could be replaced by rice powder with improved results, as indicated by decrease in mean stool output and intake of solution
TYPES OF RANDOMIZED CLINICAL TRIAL
THERAPEUTIC TRIAL : In this type therapeutic agent or procedure is given an attempt to cure the disease, relieve the symptom or prolong the survival of those with the disease.
INTERVENTION TRIAL : In this type investigator intervenes before a disease has developed in individuals with characteristics that increase their risk of developing the disease.
PREVENTIVE TRIAL : In this type, an attempt is made to determine the efficacy of a preventive agent or procedure among those without the disease. These trials are also referred to as prophylactic trials.
SR.
NO.
1.
2.
TYPE EXAMPLES
THERAPEUTIC AZT treatment for AIDS
Simple mastectomy for breast cancer
Early photocoagulation for persons who have developed
Diabetic Retinopathy
(Early Treatment Diabetic Retinopathy Study Research
Group,1991)
Beta Blocker Heart Attack Trial, 1982
INTERVENTION AZT treatment of HIV positive patients without AIDS
3.
PREVENTIVE
Mammography to detect asymptomatic breast cancer
Cholesterol lowering drugs to reduce risk of MI e.g. Coronary Primary Prevention Trial, Oslo Heart
Study.
Hepatitis B vaccination to prevent hepatitis and HCC
Education on use of condoms to reduce risk of HIV transmission
PHASES OF A CLINICAL TRIAL
PHASE I
– The researchers test an experimental drug or treatment in a small group of people (20-80)
– It is tested for the first time in humans to evaluate its safety
– Determine a safe dosage range (Maximally Tolerated dose), and
– Identify any toxic or side effects
PHASE II
– The experimental study drug or treatment is given to a larger group of people (100 300) with the target disease
– To look for the pharmacokinetic and pharmacodynamic effects of the drug on these patients
– To see if it is effective and to further evaluate its safety.
PHASE III:
– The experimental study drug or treatment is given to large groups of people (1,000 3,000)
– To confirm its effectiveness
– Monitor side effects
– Compare it to commonly used treatments, and
– Collect information that will allow the experimental drug or treatment to be used safely.
PHASE IV:
– Also called as the “Post Marketing Surveillance”,
– Post marketing studies delineate additional information including the drug's risks, benefits, and optimal use.
STEPS IN CONDUCT OF A RANDOMIZED
CONTROLLED TRIAL
The protocol
Selecting reference and experimental populations
Randomization
Intervention
Follow up
Assessment
GENERAL PLAN AND PROTOCOL OF A CLINICAL TRIAL
The plan of a clinical trial is formally stated in a “ protocol ”
It contains the objectives and the specific procedures to be used in the trial.
It should be written before the start of the trial
Contain information as the methods for selecting and then allocating the study groups, details on performance of any laboratory tests or administration of drugs
Once the protocol has been written, the epidemiologist compiles the “ manual of operations ”.
GENERAL OUTLINE OF A PROTOCOL FOR CLINICAL TRIAL
Rationale and background of the study
Specific objectives for the study
Concise statement of the study design (masking, randomization schemes, types and duration of treatments, number of patients)
Criteria for including or excluding patients
Outline of treatment procedures
Definition of all clinical, laboratory and other methods
Methods of assuring integrity of the data
Major and minor outcomes
Provisions for observing and recording side-effects
Procedures for handling problem cases
Procedures for obtaining informed consent forms from the concerned subjects
Procedures for periodic review of the trial
Procedures for termination of the trial
Procedures for analysing results
Procedures for communicating results of trial to study subjects and other interested persons
Appendices: Forms
Population without condition
STUDY POPULATION AND RECRUITMENT OF
PARTICIPANTS
POPULATION AT LARGE
Definition of condition
POPULATION WITH CONDITION
Entry criteria
With condition but ineligible
STUDY POPULATION
Enrolment
Eligible but not enrolled
STUDY SAMPLE
RANDOMIZATION
Randomization is of central importance in clinical trials
Critical element of randomization- unpredictability of next assignment.
It prevents selection bias and ensures against accidental bias.
It produces comparable groups, and eliminates the source of bias in treatment assignments
Can be achieved through random number tables or computer generated.
DESIGN AND CONDUCT OF A RANDOMIZED
CONTROLLED TRIAL
BLINDING
To remove the sources of bias in the observation 3 procedures have been developed- single masking, double masking or triple masking- also called as blinding.
SUBJECT
OBSERVER
DATA ANALYST
TYPES OF BLINDING
SINGLE DOUBLE
X
-
-
X
X
-
TRIPLE
X
X
X
SAMPLE SIZE
Clinical trials need to be designed with adequate power to answer the question being posed.
If N denotes the required sample size in each arm, then it is given by the formula:
2N= 2[ Zα√{2p(1-p)} + Zβ√{p c
(1-p c
)+ p i
(1-p i
)}] 2
(p c
-p i
) 2
Where, p c
= prevalence or anticipated event rate in control arm, p i
= prevalence or anticipated event rate in intervention arm, and p= (p c
+ p i
)/2
TRIAL DESIGNS
PARALLEL DESIGN:
– Participants are allocated to the intervention and control groups and stay in that group until the end of the study
CROSS-OVER DESIGN :
– Each participant serves as his or her own control.
– In the simplest case, half of the participants would receive intervention followed by control, and the other half the reverse.
– Advantage -smaller sample size may be required.
– Disadvantage -
Outcome in this design needs to be reversible
Secondly there is assumption of no carry-over effect from one period to the next .
Planned cross-over
Unplanned cross-over
FIGURE: DESIGN OF A PLANNED CROSS-OVER TRIAL
UNPLANNED CROSS-OVER
FIGURE: UNPLANNED CROSS-OVER
FACTORIAL DESIGN
If there is an interest in studying more than one intervention at a time, a factorial design may be more effective than a parallel design
EXAMPLE OF FACTORIAL DESIGN (PHYSICIANS’ HEALTH STUDY)
Randomized
22,071
Aspirin
11,037
Placebo,
11,034
Beta carotene
5,517
Placebo
5,520
Beta carotene
5,520
Placebo
5,514
ASPIRIN
+
-
BETACAROTENE
+ -
Betacarotene
+Aspirin
Aspirin only
Betacarotene only Neither Aspirin, nor betacarotene
EXPRESSING RESULTS IN AN RCT
Relative risk (RR):
– ID(exposed)/ID(unexposed)
Relative risk ratio(RRR):
– (1-RR)
Absolute risk reduction(ARR):
– ID(unexposed)-ID(exposed)
Efficacy=
(Rate in those who receive placebo)- (Rate in those who receive vaccine)
Rate in those who received the placebo
NUMBER NEEDED TO TREAT (NNT ): The number of patients who would need to be treated (NNT) to prevent one adverse outcome such as death. This can be calculated by:
NNT= __________________1__________________
(Rate in untreated group) - (Rate in treated group) or,
1/ Absolute risk reduction(ARR)
NUMBER NEEDED TO HARM (NNH) - Risk of side-effects by calculating the number needed to harm to cause one additional person to be harmed.
INTENT TO TREAT ANALYSIS:
Also called as Randomized or Method Effectiveness Analysis.
It compares the outcome according to the randomized group
(Gold Standard) and adherence to intervention is not necessary
Advantages:
Randomization is maintained:
Treatment assignment is based on chance alone.
Randomization provides Theoretical foundation for Statistical test of significance.
Disadvantages:
Does not take into account - protocol violation.
Groups do not remain comparable at the end
Analysis may underestimate the adverse effect.
PER PROTOCOL ANALYSIS
:
In this type analysis is done for only those who fully complied to the protocol
It does not include cross-over in final analysis
Provides a fair idea of efficacy for treatment
However, it may be biased, as randomization is compromised
AS TREATED ANALYSIS
Subject analyzed according to treatment taken or not.
(no relation with randomization).
Non compliant from treatment and vice versa analyzed accordingly.
AT is shown if ITT shows no effect ( why trial done).
REPORTING AND INTERPRETATION OF RCT
Background and rationale
Specification of the primary question and response variables used to assess it.
Pre-specified secondary questions
Nature of the study population including eligibility criteria and informed consent.
Sample size calculations and assumptions used for that calculation.
Basic study design and allocation procedures
Data collection procedures including efforts to minimize biases.
Presentation of key baseline characteristics, by group.
Process measures such as adherence, concomitant therapy use, participants lost to follow.
Results for the primary outcome, secondary outcomes and adverse events.
Adverse effects
Special analyses, such as subgroups, covariate adjustment, and data derived hypothesis.
Interpretation, implications and conclusion in context of the study and information external to the trial
A structured abstract that accurately reflects the body of the paper.
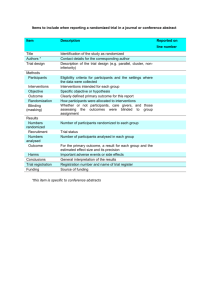
CONSORT STATEMENT
The CONSORT (Consolidated Standards for Reporting Trials) statement is used worldwide to improve the reporting of randomized controlled trials.
The CONSORT 2010 Statement is this paper including the 25 item checklist in the table and the flow diagram.
It provides guidance for reporting all randomised controlled trials, but focuses on the most common design type — individually randomised, two group, parallel trials.
Flow diagram of the progress through the phases of a parallel randomised trial of two groups (that is, enrolment, intervention allocation, follow-up, and data analysis)
PROBLEMS ENCOUNTERED WHILE CONDUCT OF AN RCT
Volunteers and non participants
Non-compliance and refusal to continue in the trial
Lost to follow up
Co-Interventions
FIELD TRIALS
Involve people who are disease free but presumed to be at risk;
Data collection takes place in the field, usually among noninstitutionalized people in the general population.
Field trials are often huge undertakings involving major logistic and financial consideration.
COMMUNITY TRIALS
Experiments -involve communities
Unit of study – groups in community
For higher prevalence of risk factors for a certain disease in a population
In a community trial the epidemiologist selects two communities that are similar in many respects as possible.
Community assent for participation in the trial is obtained from various political and other community leaders following which the trial is started.
CONDUCT OF A COMMUNITY TRIAL
Community trials have six stages. Each stage depends upon successful completion of the previous stage:
Development of a protocol
Community selection and recruitment
Establishment of a baseline and community surveillance
Intervention selection and assignment
Oversight and data monitoring
Evaluation
GENERIC DESIGN OF COMMUNITY TRIAL
QUASI-EXPERIMENTAL STUDY
Ideally, an experimental design should have three essential elements of “randomization”; “controls” and “blinding”.
Sometimes one may not be able to randomly allocate subjects into two groups or at times the placebo part of the comparison group may not be possible.
Such circumstances, when an intervention has been given to one group of subjects and not given to another group but the procedure of random allocation or placebo control has not been enforced for various reasons of impracticability or ethics, are called as “Quasi-experimental” studies.
ETHICAL CONSIDERATIONS IN A RANDOMIZED CLINICAL
TRIAL (Hill,1977)
REGISTRATION OF CLINICAL TRIALS IN INDIA
The Clinical Trials Registry –India is an online, primary register of the WHO’s International Clinical Trials Registry Platform.
It was launched on 20 July at the National Institute of Medical
Statistics, New Delhi.
Registration is voluntary and free
The register is searchable free of charge.
Public disclosure of all 20 items in the WHO Trial Registration
Data Set is mandatory for a valid registration number to be allocated.
REFERENCES:
Detels R, Beaglehole R, Lansang MA, Gulliford M. Oxford textbook of public health. Volume 2 . Methods of public health. 5 th edition, Oxford University Press,
2009.
Beaglehole R, Bonita R, Kjellstorm T. Basic epidemiology. WHO, Geneva, 2002.
Gordis L. Epidemiology. Third edition, Elsevier Saunders. 2004
Lillienfield DE, Stolley PD. Foundations of epidemiology. Third edition. Oxford
University Press, 1994.
MacMohan B, Pugh TF. Epidemiology Principles and methods. Fifth edition,
Little, Brown and Company, Boston, 1970.
Fletcher RH, Fletcher SW. Clinical epidemiology The essentials. 4 th
Lippincot Williams and Wilkins, 2005.
edition,
Textbook of public health. AFMC, Pune in collaboration with WHO India Country
Office.
Kenneth F Schulz, Douglas G Altman, David Moher, for the CONSORT Group.
CONSORT 2010 Statement: updated guidelines for reporting parallel group randomised trials; BMJ; 2010; 340:698-702.
Donner Allan. Approaches to sample size estimation in the design of clinical trials-a review. Statistics in medicine, 1984; 3; 199-214.