VSEPR Theory: Molecular Shapes & Polarity
advertisement

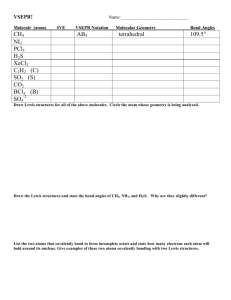
Valence Shell Electron Pair Repulsion Planar triangular Tetrahedral Trigonal pyramidal Bent VSEPR Theory Based on Electron Dot (Lewis structures) Theory predicts shapes of compounds abbreviated VSEPR VSEPR (pronounced “vesper”) stands for: Valence Shell Electron Pair Repulsion VSEPR predicts shapes based on electron pairs repelling (in bonds or by themselves) Electrons around central nucleus repel each other. So, structures have atoms maximally spread out Molecular Shape VSEPR theory assumes that the shape of a molecule is determined by the repulsion of electron pairs. Vocabulary: “domain” = any electron pair, or any double or triple bond is considered one domain. “lone pair” = “non-bonding pair” = “unshared pair” = any electron pair that is not involved in bonding “bonding pair” = “shared pair” = any electron pair that is involved in bonding Need help: VSEPR introduction video VSEPR practice problems How to determine shapes of molecules 1. Determine the central atom (usually the atom with the lowest subscript and/or the atom capable of forming the most bonds). 2. Draw the electron dot structure and bar diagram 3. Determine the molecular geometry using ALL electron pairs AND atoms around the central atom 4. Modify the geometry to determine the molecular shape if nonbonding electron pairs exist by ignoring them, BUT LEAVE THE ATOMS OF BONDED PAIRS WHERE THEY ARE. This is done because even if the electrons have no atom attached, these unbonded electron pairs still affect the shape of the structure NOTE- when counting the total number of electron pairs around the central atom, include both the bonding pairs and lone pairs. Double and triple bonds are counted as a bonding pair. 6. Use the VSEPR geometry table to predict the geometry of the molecule (it can only be one of 5 shapes!!). VSEPR THEORY: Example: BeH2 1) Central Atom? Be (only 1 atom) VSEPR THEORY: Example: BeH2 2) Electron Dot? H Be H 2) Bar Diagram? H—Be—H Note that Be violates the octet rule—this is an exception! VSEPR THEORY: Example: BeH2 3) Geometry? Hint: What is the furthest apart you can spread two atoms attached to a central atom? H Be H VSEPR THEORY: Example: BeH2 4) Shape? Ignore any unbonded pairs of electrons — not necessary in this case. LINEAR H Be H LINEAR They push each other to opposite sides of center (180 apart). BeH2 VSEPR THEORY: Example: BF3 1) Central Atom? B (only 1 atom) VSEPR THEORY: Example: BF3 2) Electron Dot? F B F F 2) Bar Diagram? F—B—F F Note that B violates the octet rule— this is an exception! VSEPR THEORY: Example: BF3 3) Geometry? Hint: What is the furthest apart you can spread three atoms attached to a central atom? F F B F VSEPR THEORY: Example: BF3 4) Shape? Ignore any unbonded pairs of electrons — not necessary in this case. trigonal planar F F B F TRIGONAL PLANAR They push each other apart equally at 120 degrees. BF3 VSEPR THEORY: Example: CH4 1) Central Atom? C (only 1 atom) VSEPR THEORY: Example: CH4 2) Electron Dot? H H C H H 2) Bar Diagram? H H—C—H H VSEPR THEORY: Example: CH4 3) Geometry? Hint: What is the furthest apart you can spread four atoms attached to a central atom? Think in 3D! H C H H H VSEPR THEORY: Example: CH4 4) Shape? Ignore any unbonded pairs of electrons —not necessary in this case. H tetrahedral C H H H TETRAHEDRAL Each repels the other equally - 109.5 - not the expected 90. Think in 3D. CH4 VSEPR THEORY: Example: NH3 1) Central Atom? N (only 1 atom) VSEPR THEORY: Example: NH3 2) Electron Dot? 2) Bar Diagram? H N H H H—N—H H VSEPR THEORY: Example: NH3 3) Geometry? Hint: What is the furthest apart you can spread three atoms plus one unbonded pair of electrons attached to a central atom? Think in 3D! ~109.5o N H H H VSEPR THEORY: Example: NH3 4) Shape? Ignore any unbonded pairs of electrons —it IS necessary in this case. trigonal pyramidal ~109.5o N H H H TRIGONAL PYRAMIDAL The thicker, lone pair forces the others a little bit closer together (~107.3) NH3 VSEPR THEORY: Example: H2O 1) Central Atom? O (only 1 atom) VSEPR THEORY: Example: H2O 2) Electron Dot? O H H 2) Bar Diagram? O—H H VSEPR THEORY: Example: H2O 3) Geometry? Hint: What is the furthest apart you can spread two atoms plus two unbonded pairs of electrons attached to a central atom? Think in 3D! ~109.5o O H H VSEPR THEORY: Example: H2O 4) Shape? Ignore any unbonded pairs of electrons —it IS necessary in this case. bent ~109.5o O H H BENT The 2 bonding pairs are pushed apart by 3rd and 4th pair (not seen) H20 NOTE: The geometry around the central atom of a water molecule is tetrahedral The molecular shape is bent. H20 What do you mean by the molecular shape vs the geometry??? The geometry takes into account ALL the bonds and unbonded electrons The molecular shape “ignores” the unbonded pairs of electrons Comparing the 2 “bents”… Both bent molecules are affected by unshared pairs – 1 pair on the left, 2 on the right. Tetrahedral vs. Trigonal pyramidal Tetrahedral geometry Tetrahedral geometry around the central atom around the central atom Tetrahedral Molecular Shape Trigonal Pyramidal Molecular Shape Tetrahedral vs. Trigonal pyramidal On the right, the 4th lone pair, is not seen as part of the actual molecule, yet affects shape. If another one of the bonding pairs on “trigonal pyramidal” were a lone pair, what is the result? VSEPR overview Each shape has a name (you will have to know these) Names of Shapes: tetrahedral trigonal pyramidal Bent Linear trigonal planar Models Tetrahedral Triangular Planar Trigonal pyramidal Bent or V Linear H Another way to draw structures in “3D” C H H H N H H H H O H Molecule CH4 Lewis Structure Number of electron pairs 4 Tetrahedral NH3 Trigonal Pyramidal 4 (3 shared 1 lone pair) SHAPE Molecule Lewis Structure Number of electron pairs Bent H2O 4 (2 shared 2 lone pairs) Linear CO2 2 MOLECULAR SHAPE Molecule BeCl2 BF3 Lewis Structure Number of electron pairs Linear 2 Trigonal Planar 3 SHAPE Back to Electronegativy You must also figure our if there are polar or nonpolar bonds between the atoms AND if the overall molecule is polar or nonpolar What???????? What is the difference between a polar bond and a polar molecule? A polar bond is a charged bond. There is a positive and negative end that do not cancel each other out A polar molecule contains polar bonds that do not cancel each other out. Determine if OF2 is polar or non polar The electronegativities of oxygen and fluorine, 3.44 and 3.98, respectively, produce a 0.54 difference that leads us to predict that the O-F bonds are polar. The molecular geometry of OF2 is bent. Such an asymmetrical distribution of polar bonds would produce a polar molecule. Determine if BF3 is polar or non polar B-F bonds are polar but they are symmetrically arranged around the central fluorine atom. No side of the molecule has more negative or positive charge than another side, and so the molecule is nonpolar. Is CCl4, a polar molecule? Or nonpolar? In CCl4, there are 4 bonds arranged symmetrically around the central carbon atom. Each bond is polar, you can tell due to the electronegativity differences between carbon and chlorine. Because the 4 atoms of chlorine are arranged symmetrically, the polarity of the bonds cancel each other out, making a non-polar molecule. Predict whether the following molecules are polar or nonpolar: (a) BrCl; (b) SO2; SOLUTION (a) Chlorine is more electronegative than bromine. Consequently, BrCl will be polar with chlorine carrying the partial negative charge: (b) Because oxygen is more electronegative than sulfur, the molecule has polar bonds. For each of these, the VSEPR model predicts a bent geometry. Because the molecule is bent, the bond dipoles do not cancel and the molecule is polar Determine if OF2 is polar or non polar The electronegativities of oxygen and fluorine, 3.44 and 3.98, respectively, produce a 0.54 difference that leads us to predict that the O-F bonds are polar. The molecular geometry of OF2 is bent. Such an asymmetrical distribution of polar bonds would produce a polar molecule. QUIZ Which of the following molecules contains polar bonds but is nonpolar? a) H2O b) NH3 c) F2 d) CCl4 e) CH2Cl2 Answer In CCl4, there are 4 bonds arranged symmetrically around the central carbon atom. Each bond is polar, you can tell due to the electronegativity differences between carbon and chlorine. Because the 4 atoms of chlorine are arranged symmetrically, the polarity of the bonds cancel each other out, making a non-polar molecule.