c9 vespr
advertisement

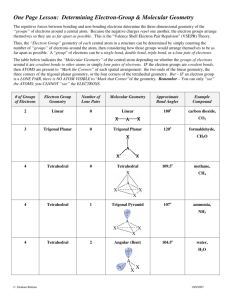
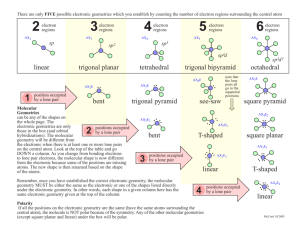
MOLECULAR GEOMETRY 1 MOLECULAR GEOMETRY VSEPR • Valence Shell Electron Pair Repulsion theory. • Most important factor in determining geometry is relative repulsion between electron pairs. Molecule adopts the shape that minimizes the electron pair repulsions. 2 3 Electron Pair Geometries Figure 9.12 4 No. of e- Pairs Around Central Atom 2 Example F—Be—F Geometry linear 180Þ F 3 F planar trigonal B F 120Þ H 4 C H 109Þ tetrahedral H H No. of e- Pairs Around Central Atom 2 5 Example F—Be—F Geometry linear 180Þ F 3 F planar trigonal B F 120Þ H 4 C H 109Þ tetrahedral H H No. of e- Pairs Around Central Atom 2 6 Example F—Be—F Geometry linear 180Þ F 3 F planar trigonal B F 120Þ H 4 C H 109Þ tetrahedral H H 7 Electron Pair Geometries Figure 9.12 Structure Determination by VSEPR Ammonia, NH3 1. Draw electron dot structure 2. Count BP’s and LP’s = 4 H H •• H N H H lone pair of electrons in tetrahedral position N H 8 3. The 4 electron pairs are at the corners of a tetrahedron. Structure Determination by VSEPR Ammonia, NH3 There are 4 electron pairs at the corners of a tetrahedron. lone pair of electrons in tetrahedral position N H H H The ELECTRON PAIR GEOMETRY is tetrahedral. 9 The MOLECULAR GEOMETRY — the positions of the atoms — is PYRAMIDAL. Ammonia, NH3 The electron pair geometry is tetrahedral. lone pair of electrons in tetrahedral position N H H H 10 11 The electron pair geometry is TETRAHEDRAL. Water, H2O 1. Draw electron dot structure 2. Count BP’s and LP’s = 4 3. The 4 electron pairs are at the corners of a tetrahedron. 12 Water, H2O The electron pair geometry is TETRAHEDRAL The molecular geometry is BENT. Geometries for Four e-Pairs Figure 9.13 Electron-Pair repulsion DECREASE angle 13 14 Formaldehyde, CH2O 1. Draw electron dot structure 2. Count BP’s and LP’s at C • • H O C • • H 3. There are 3 electron “lumps” around C at the corners of a planar triangle. • • O • • The electron pair geometry is PLANAR TRIGONAL with 120o bond angles. C H H 15 Formaldehyde, CH2O • • O The electron pair geometry is PLANAR TRIGONAL • • C H H The molecular geometry is also planar trigonal. 16 Structures where Central Atom do NOT contain 4 Electron Pairs Often occurs with Group 3A elements and with those of 3rd period and higher. 17 Boron Compounds •• • • • • Consider boron trifluoride, BF3 •• • • F •• The B atom is surrounded by only 3 electron pairs. Bond angles are 120o Geometry described as planar trigonal F B • • • • F •• Compounds with 5 Pairs Around the Central Atom 90Þ F F P Trigonal bipyramid F 120Þ F F 5 electron pairs 18 19 Sulfur Tetrafluoride, SF4 • Number of valence electrons = 34 • Central atom = S 90Þ •• F F F •• S •• F •• •• •F• • • •• F S •• •• F •• •• • •F •• Electron pair geometry 120Þ --> trigonal bipyramid (because there are 5 pairs around the S) •• •• •• F •• • •• •F •• •• S F •• •• Sulfur Tetrafluoride, SF4 •F• • • •• Lone pair is in the equator because e’s require more room. 90Þ F •• S F F F 120Þ 20 Molecular Geometries for Five Electron Pairs Figure 9.14 21 22 Compounds with 6 Pairs Around the Central Atom 6 electron pairs 90Þ F F S F Octahedron F F F 90Þ Molecular Geometries for Six Electron Pairs Figure 9.14 23 Electronic Geometries Molecular Geometries 24