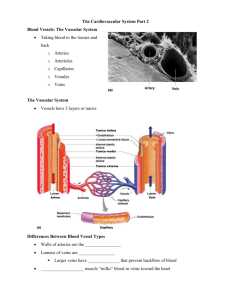
Fetal circulation
advertisement

Regulation of the heart and vessel activities Romana Šlamberová, MD PhD Department of Normal, Pathological and Clinical Physiology Distribution of blood circulation Total volume of blood in all vessels (intravascular volume): man: 5.4 l (77 ml / kg) woman: 4.5 l (65 ml / kg) Distribution: Heart 7% Pulmonary circulation 9% Systemic circulation 84% from that veins 75% large arteries 15% small arteries 3% capilaries: 7% Resistance of blood circulation Total peripheral resistance: of all paralel restistance in the body Actual resistance is given based on the lumen of vessels and viscosity of blood Percentage portion of resistance in different types of vessels: large arteries 19% small arteries 47% capillaries 27% veins 7% Resistance depends not only on type of vessel, but also on the actual situation of blood need in organs Regulation of blood circulation Mechanisms of regulation: Local Humoral (chemical) – O2, CO2, H+ Nervous Enzymatic and hormonal General Fast = short-term (regulate blood pressure) Slow = long-term (regulate blood volume) – several days Local chemical regulatory mechanisms The most obvious in the heart and the brain Goal: autonomic regulation of resistance by organ based on its metabolic needs Principal: accumulation of products of metabolism (CO2, H+, lactacid ) or consumption of substances necessary for proper function (O2) directly affects smooth muscles of vessels and induce vasodilatation Local nervous regulatory mechanisms The most obvious in the skin and mucous Goal: central regulation of blood distribution Principal: Autonomic nervous system Sympaticus Vasoconstriction – activation of α receptors in vesselsnoradrenalin (glands, GIT, skin, mucous, kidneys, other inner organs) Vasodilatation – activation of β receptors in vessels – adrenalin (heart, brain, skeletal muscles) Parasympaticus - Acetylcholin Vasoconstriction – heart Vasodilatation – salivatory glands, GIT, external genitals Local enzymatic and hormonal regulatory mechanisms Kinin ↑ = vasodilatation Cells of GIT glands contain kallikrein – changes kininogen to kinin → kallidin → bradykinin (vasodilatation) Kinins are any of various structurally related polypeptides, such as bradykinin and kallikrein, that act locally to induce vasodilation and contraction of smooth muscle. A role in inflammation, blood pressure control, coagulation and pain. Hormones of adrenal medula: adrenalin (vasodilatation), noradrenalin (vasoconstriction) General fast (short-term) regulatory mechanisms (1) Nervous autonomic reflexes Baroreflex glomus caroticum, glomus aorticum Afferentation: IX and X spinal nerve Centre: medulla oblongata, nucleus tracti solitarii Efferentation: X spinal nerve, sympatetic fibres Effector: heart (atriums), vessels Effect: After acute increase of blood pressure – activation of receptors – decrease of blood pressure (vasodilatation, decrease of effect of sympaticus) Baroreceptor sensitivity The sensitivity of baroreceptor reflex corresponds with good prognosis of life length (lower probability of heart attack) Depends on the tonus of n. vagus People with low vagotonus have higher incidence of unexpected death Association between baroreceptor sensitivity and hypercholesterolemia Persons with higher level of LDL cholesterol have lower baroreceptor sensitivity Koskinen et al. 1995 General fast (short-term) regulatory mechanisms (2) Receptors in the heart Reflex of atrial receptors – mechano- and volumoreceptors – activated by increased blood flow through the heart A receptors – sensitive to ↑ of wall tension after systole of atriums B receptors – sensitive to ↑ of wall tension after systole of ventricles Ventricular receptors – mechano- and chemical receptors activated in pathological cases Hypoxia of myocardium → decrease of heart rate (Bezold-Jarisch reflex) → protection of myocardium of larger damage General fast (short-term) regulatory mechanisms (3) Humoral mechanisms Adrenalin – β receptors → vasodilatation → ↓ peripheral resistance → blood from skin and GIT to skeletal muscles, heart and brain → ↑ minute heart volume Noradrenalin – α receptors → vasoconstriction → ↑ blood pressure Renin-angiotensin – activated by ↓ pressure in vas afferens General slow (long-term) regulatory mechanisms Regulatory mechanisms of water and electrolytes exchanges Regulation of total blood volume by kidneys Increase of ADH (vasopressin) When ↑ blood pressure → ↑ of filtration pressure in glomeruli → ↑ production of urine → ↓ volume of circulating blood → ↓ blood pressure ↑ ADH → ↑ of the permeability of collecting ductus for the water → water is reabsorbed → ↑ volume of circulating blood → ↑ blood pressure Increase of Aldosterone ↑ aldosterone → ↑ reabsorbtion Na+ and water → ↓ volume of urine → ↑ volume of circulating blood → ↑ blood pressure Intracardial regulatory mechanisms (1) Frank-Starling’s law = initial length of the fibers is determined by the degree of diastolic filling of the heart, and the pressure developed in the ventricle is proportionate to the total tension developed. The developed tension increases as the diastolic volume increases until it reaches a maximum, then tends to decrease. Ganong: Review of Medical Physiology Intracardial regulatory mechanisms (2) Inotrophy = ability of muscle contraction and its dependency on other factors, e.g. initial tension of muscle fiber. Ionotropic effect of heart rhythm ↑ heart frequency → ↑ amount of Ca2+ that goes into heart cells → ↑ Ca2+ available for tubules of sarkoplasmatic reticulum → ↑ Ca2+ that is freed by each contraction → ↑ strength of contraction Extracardial regulatory mechanisms Cardiomotoric centers Inhibition – ncl. Ambiguus (beginning of n. vagus in medulla oblongata) Excitation - Th1-3 beginning of sympathetic fibres Vasomotoric centers In brain stem (medulla oblongata, Pons Varoli) In the hypothalamus (controls activity of vasomotoric centers in brain stem) Brain cortex – control both the hypothalamus and the brain stem Midbrain regions of CV control Rostral ventrolateral medulla Cardiac accelerator center Vasoconstrictor center Area postrema Nucleus tractus solitarius Nucleus ambiguous Cardiac decelerator center Caudal ventrolateral Medulla Fibers from this neurons project to the vasoconstrictor area and inhibit it Ackermann Cerebral chemoreceptors Chemoreceptors in the medulla are most sensitive to pCO2 and pH and less sensitive to pO2 Reflex during decreased cerebral blood flow: increase in pCO2 and decrease in pH activates chemoreceptors Increase in both sympathetic and parasympathetic outflow Increased contractility, increased total physical response, but decreased heart rate Intense arteriolar vasoconstriction redirects blood flow to the brain Sympathetic nerve activity and arterial pressure •Decreasing blood pressure is followed with increasing sympathetic nerve activity •Vasoconstriction increases blood pressure Respiration arytmia Heard frequency = 72 pulses/min, = pulse interval 0.83 s During relaxation the frequency changes based of the respiration (RESPIRATION ARYTMIA) inspiration - increased frequency expiration – decreased frequency Bradycardia = fysiological = deep long-term inspiration, deep forward bend and knee band = reflex changes of vagal tonus. Tachycardia = fysiological = swallow (decrease of vagal tonus), change of position from lying or sitting to standing (ORTHOSTATIC REACTION). Orthostatic reaction Changes in posture from supine position to standing Mechanisms Blood pools in the veins of lower extremities Venous return to the heart decreases, cardiac output decreases (Frank-Starling law) Mean arterial pressure decreases Decreased activation of baroreceptors Increased sympathetic outflow to the heart and blood vessels and decreased parasympathetic outflow Specific circulatory systems Pulmonary and systemic circulation differs in their pressure and resistence. Pressure in pulmonary circulation is about 4 – 5 times lower than in systemic one. Different organs have Differences in vascular resistance Differences in metabolic demands Local control (intrinsic) Hormonal control (extrinsic) Cerebral circulation 15 % of cardiac output Is controlled by local metabolites pCO2 (H+) is the most important vasodilator CO2 diffuses to vascular cells, forms H2CO3 (H+) Intracellular H+causes vasodilatation Increase in blood flow, removal of excess CO2 Decrease in pO2 increases cerebral blood flow Many vasoactive substances do not affect cerebral circulation, do not cross the blood-brain barrier Coronary circulation 5 % of cardiac output Local metabolic factors Hypoxia: increase in myocardial contractility – increased O2 consumption – local hypoxia Hypoxia causes vasodilatation of the coronary arterioles – compensatory increase in blood flow and O2 delivery Adenosine (from ATP) causes vasodilatation Mechanical compression of the blood vessels during systole in the cardiac cycle – brief period of occlusion and reduction of blood flow Pulmonary circulation 100% of cardiac output Lower pressure and low resistance Controlled by local metabolites, primarily by pO2 (bellow 70 mm Hg) Opposite effect than in other tissue – hypoxia causes vasoconstriction Mechanism – inhibition of NO production in endothelial cells of blood vessel walls Redistribution of blood from poorly ventilated areas to wellventilated areas Renal circulation 25 % of cardiac output Renal blood flow is autoregulated Constant blood flow even when renal perfusion pressure changes (80-200 mmHg) Renal autoregulation is independent of sympathetic innervation (transplanted kidney) Angiotensin II – vasoconstrictor for both afferent and efferent arterioles, but efferent arteriole is more sensitive Prostaglandins (E2, I2 – produced locally) – vasodilatation of both arterioles Skeletal muscle circulation 25 % of cardiac output Sympathetic innervation At rest: activation of a1 (noradrenaline) receptors causes vasoconstriction, increased resistance and decreased blood flow Activation of b2 (adrenaline) receptors causes vasodilatation Local metabolites During exercise: local vasodilator – lactate, adenosine, K+ Skin circulation 5 % of cardiac output Dense sympathetic innervation – regulates blood flow for regulation of body temperature Increase core body temperature – decrease sympathetic tone to the smooth muscle sphincters controlling A-V anastomoses - increase skin blood flow Arteriovenous anastomoses – permit bypass of the capillary vessels Fetal circulation (1) The circulatory system of a human fetus works differently from that of born humans, mainly because the lungs are not in use: the fetus obtains oxygen and nutrients from mother through the placenta and the umbilical cord. Blood from the placenta is carried by the umbilical vein. About half of this enters the ductus venosus and is carried to the inferior vena cava, while the other half enters the liver proper from the inferior border of the liver. Fetal circulation (2) The blood then moves to the right atrium of the heart. In the fetus, there is an opening between the right and left atrium (the foramen ovale), and most of the blood flows from the right into the left atrium, thus bypassing pulmonary circulation (which aren't being used for respiration at this point as the fetus is suspended in amniotic fluid). The majority of blood flow is into the left ventricle from where it is pumped through the aorta into the body. Some of the blood moves from the aorta through the internal iliac arteries to the umbilical arteries, and re-enters the placenta, where carbon dioxide and other waste products from the fetus are taken up and enter the mother's circulation. Some of the blood from the right atrium does not enter the left atrium, but enters the right ventricle and is pumped into the pulmonary artery. In the fetus, there is a special connection between the pulmonary artery and the aorta, called the ductus arteriosus, which directs most of this blood away from the lungs. Postnatal development of circulation With the first breath after birth, the pulmonary resistance is dramatically reduced. More blood moves from the right atrium to the right ventricle and into the pulmonary arteries, and less flows through the foramen ovale to the left atrium. The blood from the lungs travels through the pulmonary veins to the left atrium, increasing the pressure there. The decreased right atrial pressure and the increased left atrial pressure pushes the septum primum against the septum secundum, closing the foramen ovale, which now becomes the fosse ovalis. This completes the separation of the circulatory system into the left and the right. The ductus arteriosus normally closes off within one or two days of birth, leaving behind the ligamentum arteriosum. The umbilical vein and the ductus venosus closes off within two to five days after birth, leaving behind the ligamentum teres and the ligamentum venosus of the liver respectively. Differences between fetal and adult circulatory systems The fetal foramen ovale - the adult fosse ovalis. The fetal ductus arteriosus - the adult ligamentum arteriosum. The extra-hepatic portion of the fetal left umbilical vein - the adult ligamentum teres hepatis (the "round ligament of the liver"). The intra-hepatic portion of the fetal left umbilical vein (the ductus venosus) - the adult ligamentum venosum. The proximal portions of the fetal left and right umbilical arteries - the adult umbilical branches of the internal iliac arteries. The distal portions of the fetal left and right umbilical arteries the adult medial umbilical ligaments. Fetal hemoglobin differs from adult hemoglobin. Fetal hemoglobin (1) Fetal hemoglobin differs most from adult hemoglobin in that it is able to bind oxygen with greater affinity than the adult form, giving the developing fetus better access to oxygen from the mother's bloodstream. The P50 value for fetal hemoglobin (i.e., the partial pressure of oxygen at which the protein is 50% saturated; lower values indicate greater affinity) is roughly 19 mmHg, whereas adult hemoglobin has a value of approximately 26.8 mmHg. Fetal hemoglobin (2) At birth, fetal hemoglobin comprises 50-95% of the child's hemoglobin. These levels decline after six months as adult hemoglobin synthesis is activated, while fetal hemoglobin synthesis is deactivated. Soon after, adult hemoglobin (hemoglobin A) takes over as the predominant form of hemoglobin in normal children. Neonatal jaundice tends to develop because of two factors Decrease of the number of erythrocytes. The breakdown of fetal hemoglobin as it is replaced with adult hemoglobin The relatively immature hepatic metabolic pathways, which are unable to conjugate bilirubin as fast as an adult.