Chapter Thirteen PPT
advertisement

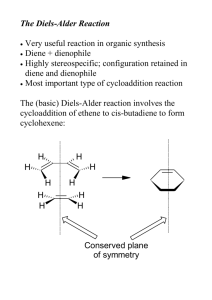
Chapter 13: Conjugated p-Systems • Allylic Substitution—Allyl Radicals (Section 13.2) • Allyl Radical Stability (Section 13.3) • Allyl Cation/Anion (Section 13.4) • Resonance Structures Revisited (Section 13.5) • Alkadienes, Polyunsaturated Hydrocarbons (Section 13.6) • 1,3 Butadiene (Section 13.7 – 13.8) • UV-Vis Spectroscopy (Section 13.9) • Electrophilic Attack: 1,4 Addition (Section 13.10) • Diels-Alder Reactions (Section 13.11) Allylic Substitution Br Br2, CCl4 Br 0 oC high temperature Br or dilute X2 + HBr • First Reaction Addition of Br2 to Alkene • Second Reaction Allylic Substitution • Illustrates Reaction’s Dependence Upon Conditions Allylic Chlorination Cl2, 400 oC Gas Phase • Allyl Choride Synthesis Known as “Shell Process” • Radical Substitution Mechanism (Multi-Step) Initiation Propagation Termination Cl Allylic Chlorination: Mechanism Cl Cl H Cl 2 Cl Cl Cl + HCl Cl + • Allylic C—H Bonds Relatively Ease to Dissociate • Termination Arises from Any Combination of Radicals Cl Allylic Bromination: NBS O O + NBr light or ROOR Br + NH CCl4 O • NBS: N-Bromosuccinimide (Low Br2 Concentration) • Nonpolar Solvent, Dilute Conditions • Primarily Get Allylic Substitution Product O Allylic Radical: MO Description p3 p2 p1 • Three p Orbitals Combine to Form 3 p Molecular Orbitals • One Unpaired Electron (Radical) p Molecular Orbitals: General Rules • p Molecular Orbitals are Symmetric • Nodes Are Through Atoms or Bonds • Nodes Represent an Orbital Phase Change (+/-) • In Allyl Radical, Unpaired Electron on C1 and C3 (NOT C2) • p Molecular Orbitals Explain Resonance in Allyl Radical • Same Orbital Picture for Same Carbon Scaffold (Orbital Occupancy Changes) Allylic Radical: MO Description Allyl Cation Allyl Anion p3 p3 p2 p2 p1 p1 • Same Orbitals as Allyl Radical (Different Occupancies) Resonance: The Carbonate Ion 2- 2- O C O O O 2- O O C C O O O • Double headed arrows indicate resonance forms • Red “Curved Arrows” show electron movement • Curved Arrow notation used to show electron flow in resonance structures as well as in chemical reactions: we will use this electron bookkeeping notation throughout the course Rules for Drawing Resonance Structures 2- 2- O C O O O 2- O O C C O O O 1. Hypothetical Structures; “Sum” Makes Real Hybrid Structure 2. Must be Proper Lewis Structures 3. Can Only Generate by Moving Electrons (NO Moving Atoms) 4. Resonance Forms are Stabilizing 5. Equivalent Resonance Structures Contribute Equally to Hybrid Rules for Drawing Resonance Structures 6. More Stable Resonance Forms Contribute More to Hybrid Factors Affecting Stability 1. Covalent Bonds 2. Atoms with Noble Gas (Octet) Configurations H2C O CH3 vs. H2C O CH3 3. Charge Separation Reduces Stability 4. Negative Charge on More Electronegative Atoms Alkadienes (Polyunsaturated HCs) H2 C CH2 1,2-Propadiene 1,3-Butadiene CH2 (3Z)-Penta-1,3-diene CH2 (3E)-Penta-1,3-diene CH2 (2Z,4E)-Hexa-2,4-diene Pent-1-en-4-yne • Follow the General IUPAC Rules We’ve Used This Semester Alkadienes: 1,3-Butadiene 1.47 Å 1.34 Å 1.34 Å 1,3-Butadiene s-cis Conformation s-trans Conformation • Conformations Not True cis/trans (Single Bond Rotomers) • Conformations Will be Important for Diels-Alder Reactions Alkadienes: 1,3-Butadiene MOs ANTI-BONDING Orbitals LUMO HOMO BONDING Orbitals What Would Butadiene Cation/Anion Occupancies Look Like? UV-Vis Spectroscopy • Measures Absorbance at Wavlengths Spanning UV/Vis Regions • UV: Ultraviolet Vis: Visible • Typically Record Solvent Spectrum First, Subtract From Sample • Intensity (y-axis) is the Molar Absorptivity (Extiction Coefficient) • Conjugated Dienes Have Absorptions Detectable by UV-Vis • Absorbances of Conjugated Dienes Typically > 200nm • More Conjugation (# of p Bonds) Greater Wavelength • Smaller HOMO/LUMO Gap Greater Wavelength (E=hc/l) UV-Vis Absorption Spectrum 1000 800 700 600 550 500 360 340 320 300 N N 6000 Hy2Na –1 –1 400 380 (nm) t-Bu / M cm 450 4000 lmax 2000 0 10 15 20 25 3 h /10 cm 30 35 –1 Representative UV Spectrum: Top Axis is Nanometers, Bottom cm-1 1,4 Addition in Conjugated Dienes Cl HCl + 25 oC 78% Cl 22% Cl 1,2 addition 1,4 addition H H Cl • 1,4 Addition Due to Stability and Delocalization in Allyl Cation • Look at the Intermediate (Carbocation) Observed in Reaction 1,4 Addition in Conjugated Dienes H+ • Resonance Forms (Hybrid) Explain Possible Addition Products • 1,4 Product is Thermodynamic Product: Lower Energy • 1,2 Product is Kinetic Product: Reaction Occurs Faster Elevated Temperatures Favor Thermodynamic Addition Products Diels-Alder Reactions: 1,4 Cycloadditions 1,4 Cycloaddition Diene Dienophile (s-Cis) Diels-Alder Adduct • Diels-Alder Reactions are 1,4 Cycloadditions • Diene (4 p e¯) and Dienophile (2 p e¯) Form Cyclic Structure • Usually Requires Elevated Temperature Conditions • Usually Energetically Favored (2 s Bonds Stronger than 2 p) Diels-Alder Reactions: 1,4 Cycloadditions O O 1,4 Cycloaddition Diene Dienophile (s-Cis) Diels-Alder Adduct O AlCl3 Et2O 25 oC + OCH3 O O OCH3 Representative Diels-Alder Reactions O Diels-Alder Reactions: 1,4 Cycloadditions GENERAL NOTES ON DIELS-ALDER REACTIONS: • Stereospecific: Syn Additions, Retain Dienophile Configuration • Diene Must React in s-Cis Conformation (Strain in New Ring) • Under Kinetic Conditions, Endo Products are Favored R H H R Endo Exo …That’s All Folks! (For Slides, Anyway)