CH 4
advertisement

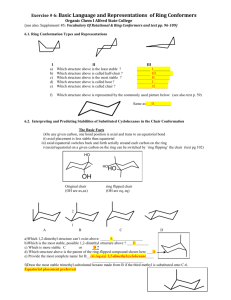
CH 4: Organic Compounds: Cycloalkanes and their Stereochemistry Renee Y. Becker CHM 2210 Valencia Community College 1 Cycloalkanes • Rings of carbon atoms (CH2 groups) • Formula: CnH2n • Nonpolar, insoluble in water • Compact shape • Melting and boiling points similar to branched alkanes with same number of carbons 2 Naming Cycloalkanes • Cycloalkane usually base compound – May be cycloalkyl attachment to chain • It is off of a chain that has a longer carbon chain • Number carbons in ring if >1 substituent. • Number so that sub. have lowest numbers – Give first in alphabet lowest number if possible CH2CH3 CH2CH3 CH3 3 Naming Cycloalkanes • Find the parent. # of carbons in the ring. • Number the substituents 4 Example 1 Give IUPAC names CH2CHCH2CH3 CH3 H3C CH3 CH3 5 Example 2 Draw the structure a) propylcyclohexane b) cyclopropylcyclopentane c) 3-ethyl-1,1-dimethylcyclohexane 6 Stereoisomerism • Compounds which have their atoms connected in the same order but differ in 3-D orientation 7 Cis-Trans Isomerism • Cis: like groups on same face of ring • Trans: like groups on opposite face of ring • Sub. Do not have to be on adjacent carbons of ring 8 9 Cycloalkane Stability • 5- and 6-membered rings most stable • Bond angle closest to 109.5 • Angle (Baeyer) strain • Measured by heats of combustion per -CH2 – The more strain, the higher the heat of combustion, per CH2 group – The energy released as heat when one mole of a compound undergoes complete combustion with oxygen. 10 Stability of Cycloalkanes: The Baeyer Strain Theory • Baeyer (1885): since carbon prefers to have bond angles of approximately 109°, ring sizes other than five and six may be too strained to exist • Rings from 3 to 30 C’s do exist but are strained due to bond bending distortions and steric interactions 11 Summary: Types of Strain • Angle strain - expansion or compression of bond angles away from most stable • Torsional strain - eclipsing of bonds on neighboring atoms • Steric strain - repulsive interactions between nonbonded atoms in close proximity 12 Heats of Combustion (per CH2 group) Alkane + O2 CO2 + H2O 166.6 164.0 157.4 Long-chain 158.7 158.6 158.3 157.4 13 Cyclopropane • Large ring strain due to angle compression • Very reactive, weak bonds 14 Cyclopropane Torsional strain because of eclipsed hydrogens 15 Cyclobutane • Angle strain due to compression • Torsional strain partially relieved by ringpuckering 16 Cyclopentane • If planar, angles would be 108, but all hydrogens would be eclipsed. • Puckered conformer reduces torsional strain. 17 Cyclohexane • Combustion data shows it’s unstrained. • Angles would be 120, if planar. • The chair conformer has 109.5 bond angles and all hydrogens are staggered. • No angle strain and no torsional strain. 18 Chair Conformer 19 Boat Conformer 20 Conformational Energy 21 Axial and Equatorial Positions 22 Drawing the Axial and Equatorial Hydrogens 23 Monosubstituted Cyclohexanes 24 1,3-Diaxial Interactions • Difference between axial and equatorial conformers is due to steric strain caused by 1,3-diaxial interactions 25 Hydrogen atoms of the axial methyl group on C1 are too close to the axial hydrogens three carbons away on C3 and C5, resulting in 7.6 kJ/mol of steric strain 26 Disubstituted Cyclohexanes 27 Conformational Analysis of Disubstituted Cyclohexanes • In disubstituted cyclohexanes the steric effects of both substituents must be taken into account in both conformations • There are two isomers of 1,2-dimethylcyclohexane. cis and trans • In the cis isomer, both methyl groups are on the same face of the ring, and compound can exist in two chair conformations • Consider the sum of all interactions • In cis-1,2, both conformations are equal in energy 28 Conformational Analysis of Disubstituted Cyclohexanes 29 Trans-1,2-Dimethylcyclohexane • Methyl groups are on opposite faces of the ring • One trans conformation has both methyl groups equatorial and only a gauche butane interaction between methyls (3.8 kJ/mol) and no 1,3-diaxial interactions • The ring-flipped conformation has both methyl groups axial with four 1,3-diaxial interactions • Steric strain of 4 3.8 kJ/mol = 15.2 kJ/mol makes the diaxial conformation 11.4 kJ/mol less favorable than the diequatorial conformation • trans-1,2-dimethylcyclohexane will exist almost exclusively (>99%) in the diequatorial conformation 30 Trans-1,2-Dimethylcyclohexane 31 Cis-Trans Isomers Bonds that are cis, alternate axial-equatorial around the ring. CH3 CH3 32 Bulky Groups • Groups like t-butyl cause a large energy difference between the axial and equatorial conformer. • Most stable conformer puts t-butyl equatorial regardless of other substituents. 33 Example 3 Draw the most stable conformation a) ethylcyclohexane b) isopropylcyclohexane c) t-butylcyclohexane d) cis-1-t-butyl-3-ethylcyclohexane e) trans-1-t-butyl-2-methylcyclohexane f) trans-1-t-butyl-3-(1,1-dimethylpropyl)cyclohexane 34 Example 4 Which of the following is the most strained ring? Least strained? Why? a) c) b) d) 35 Table 4.2 Axial and Equatorial Relationship in Cis and trans Disub Cyclohexanes Cis/trans pattern Axial/Equatorial Relationship 1,2–Cis a,e e,a 1,2-trans a,a e,e 1,3-cis a,a e,e 1,3-trans a,e e,a 1,4-cis a,e e,a 1,4-trans a,a e,e 36