Biochemistry Questions Jeopardy Review
advertisement

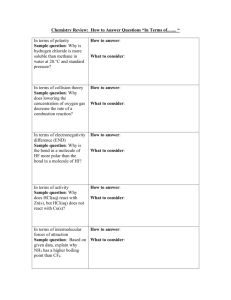
100 200 300 400 Ele500 100 200 300 400 500 100 200 300 400 500 100 200 300 400 500 100 200 300 400 500 Atoms and Subatomic Particles Which subatomic particles are negatively charged? Which two subatomic particles are found in the nucleus? What does “amu” stand for? Carbon 14 has an atomic mass of 14 amu. Carbon 12 has an atomic mass of 12 amu. If both atoms are neutral, what subatomic particle is present in differing numbers between C-12 and C-14? What is created when an atom of chlorine gains an electron and becomes an atom of chloride? Ionic, Covalent and Hydrogen Bonding What type of bond is characterized by the sharing of electrons? Table salt is a combination of Na and Cl. An electron is completely transferred from Na to Cl. What type of bond is formed to create table salt? Of ionic, covalent and hydrogen bonding, which is strongest? What type of bonding keeps two hydrogen atoms bound to an oxygen atom to form water? What sort of bond is created when two pairs of electrons are shared? Water What sort of bonding happens between molecules of water and accounts for water’s unique properties? There’s a saying in chemistry: Like dissolves like. What characteristic of water molecules allows many polar substances to easily dissolve in water? Which property of water allows water to move up a paper towel when one edge of the paper towel gets wet? Cotton and salt both exhibit this trait, as salt dissolves in water and cotton absorbs water without being dissolved. What’s unusually high what allows it to resist changes in temperatures? Acids and Bases What pH is considered neutral? pH levels greater than 7.0 are considered what types of substances? How many times more H+ are in grapefruit juice (pH 3.0) than black coffee (5.0)? How many times more OH- are in sea water (pH 8.0) than ammonia solution (11.0)? What sort of solution has a greater concentration of OH- than H+? Experimental Design The title of an experiment is: The color of hospital nurseries impacts the amount of time newborn humans spend crying. What is the independent variable? The title of an experiment is: The color of hospital nurseries impacts the amount of time newborn humans spend crying. What is the dependent variable? The title of an experiment is: The color of hospital nurseries impacts the amount of time newborn humans spend crying. Name three things that should be held constant (and could realistically be held constant) in this experiment. What is the maximum number of independent variables an experiment can have? On which axis of a graph will you usually find the independent variable? Electrons Protons and Neutrons Atomic Mass Unit Isotopes Table Salt Covalent Ionic Covalent Covalent Double Bond Hydrogen Water is polar Adhesion Hydrophilic Specific Heat 7.0 Bases 100x 1000x Basic Color of the Nursery Time newborns spend crying Age of babies, health of babies, time of day, noise level in nursery, etc. One X axis 100 200 300 400 500 100 200 300 400 4500 100 200 300 400 500 100 200 300 400 500 100 200 300 Atoms and Subatomic Particles Which subatomic particle determines the identity of the element? How many neutrons must an atom of carbon have, if the atom has an atomic mass of 13 amu? What will the charge of a magnesium ion be if it becomes stable by filling an outer ring in the easiest way possible for the atom? Neon does not easily for ions. How many valence electrons must it have when it is neutral? How many electrons does it take to fill up the first and second energy shells, respectively? Ionic, Covalent and Hydrogen Bonding Which subatomic particles are involved in bonding? Which bonds are formed by the complete transfer of an electron, resulting in an attraction between oppositely charged atoms? Of ionic, covalent and hydrogen bonding, which bonds are weakest? How many valence electrons does a neutral atom of Carbon have? Which type of bonds generally do not involve metals? Water Give the chemical formula for water. A water molecule exhibits polarity. Which atom or atoms of a water molecule accumulate electrons, generating a partial negative charge? What property of water allows it (as a component of your blood) to move through the smallest of capillaries? What is the atomic mass of a typical water molecule? Round to the nearest hundredth. What word describes nonpolar compounds that do not dissolve in water and do not absorb water? Acids and Bases Substances with a pH less than 7.0 are considered what? The pH scale is logarithmic. This means there are _________ the number of H+ or OH- ions between any two integers. If a solution has a concentration of OH- greater than H+ ions, would the solution be considered an acid, a base or neutral? I mixed 10 mL liquid hydrochloric acid with 1 L distilled water. What is the solute in this example? What number on the pH scale will have the highest concentration of OHions? Experimental Design A colder winter results in fewer mosquitoes during the following summer. What is the independent variable? A colder winter results in fewer mosquitoes during the following summer. What is the dependent variable? A colder winter results in fewer mosquitoes during the following summer. What things would be considered constants? 400 Give two qualitative observations about the weather today. 500 Give two quantitative observations about the weather today. Proton 6 +2 8 2 and 8 Electrons Ionic Hydrogen 4 Covalent H2O Oxygen Adhesion 18.01 amu hydrophobic Acidic 10 times Basic Hydrochloric acid 14 Temperature during winter Number of mosquitoes Region, proximity of water, use of pesticides, etc. It’s humid, it’s sunny, it’s cloudy, there is fog, etc It’s 87 degrees F, the humidity is 60%, etc.