Biochemistry - Green Local Schools
advertisement

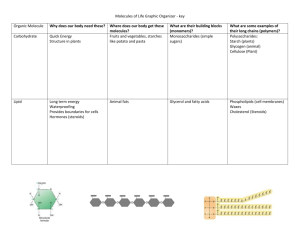
Biochemistry Chapter 3 Water Section 2.3 Structure of Water Most abundant molecule Held together by covalent bonds 2 atoms of H, 1 atom of O Water is a Polar Molecule Definition: a molecule with an uneven distribution of charge but a net charge of zero Water bonds at an angle Water’s ability to dissolve “Like Dissolves Like” Polar substances can dissolve polar substances, nonpolar substances can dissolve nonpolar substances “Universal solvent” Questions: Why don’t oil and water mix? Why can water dissolve sugar or salt? How does water dissolve a polar substance? Answer: The + end of water attracts to the – ion of the substance The – end of water attracts to the + ion of the substance This breaks the ionic bond of the substance thus dissolving it Water’s special bond: Hydrogen Bond: a weak chemical bond that exists between the H atoms and a – charged part of a different molecule Water’s special properties: Due to H-bonds: 1. Water can cling to itself and other substances 2. Water is able to absorb large amounts of energy without changing temperatures quickly (high boiling point) Cohesion: When like molecules are attracted to each other Ex) surface tension in water Adhesion: Definition: the attractive forces between unlike substances Capillarity: Definition: cohesion and adhesion working together to move water molecules up a narrow tube against the force of gravity Homeostasis? 1. 2. 3. The H-bonds in water are the first bonds to break when an increase in energy (temp) is applied It takes a LARGE amount of energy to break/move water molecules Thus, the temperature of water stays fairly constant in a cell even though there might be a drastic temperature change outside Lab time….yeah! Carbon Compounds SECTION 3.1 Organic Compounds Organic compounds contain carbon atoms Form covalent bonds with other C atoms or to other elements N, H, and O. Carbon Bonding C readily covalently bonds (a bond forms when electrons are shared) Each C forms 4 covalent bonds Large Carbon Molecules Polymers are made up of many small, repeating molecules called monomers. Macromolecules- large polymers Condensation Reaction A chemical reaction that links monomers to form polymers One water molecule is produced A.k.a. dehydration synthesis Ex) Glucose and Fructose combine to make Sucrose, table sugar Condensation Reaction: The formation of larger molecules by removing a H+ from one monomer and a OH- from the other monomer. The H+ and the OH- combine to form the bi-product H2O. Hydrolysis A chemical reaction that breaks down polymers Reversal of condensation reaction Usually done by adding water ATP – Adenosine Triphosphate All of life’s functions require energy Energy compounds found in cells Biochemistry The stuff life is made of! Section 3.3 The Four Molecules of Life 1. 2. 3. 4. Carbohydrates Proteins Lipids Nucleic Acids All of your body is made up of these four types of molecules! The Building Blocks Each type of molecules is made up of smaller parts called monomers. When 3 or more monomers are linked together, they form a polymer. How to make a macromolecule. How do you make a polymer from many monomers? CONDENSATION REACTION! What will break up a large molecule? The opposite reaction of condensation…… HYDROLYSIS REACTION! The use of water to break apart polymers back into monomers. Condensation/Hydrolysis Monomers of the 4 Life Molecules 1. 2. 3. 4. Carbohydrates - Monosaccharide Proteins – Amino Acids Lipids – Fatty Acids Nucleic Acids - Nucleotides How do you make macromolecules of each monomer? CONDENSATION REACTION 1. Carbohydrates Used for “fuel” and structural material Monosaccharide = monomer, simple sugar, ex) fructose, galactose, & glucose C6H12O6 Disaccharide = 2 monomers, double sugar ex) sucrose (table sugar) Examples) ________________________________________ Polysaccharide = too many to count ex) starch & cellulose (in plants) glycogen (in animals) 2. Protein Amino Acid = monomer, 20 different types Peptide bond holds amino acids together Polypeptide = chains of amino acids Protein = the final functional form, 1 or more polypeptides Most DIVERSE group of molecules: •structures such as antibodies, hormones, muscles, skin, hair, and biological catalysts (enzymes) are made of proteins. Enzymes Protein molecules that catalyze (increase the rate of) biochemical reactions How Do Enzymes Work? Animation: How Enzymes Work 3. Lipids Fatty Acids = monomer •Hydrophilic carboxyl head polar •Hydrophobic fatty acid tail – nonpolar, not soluble in water Ex) saturated (solid) and unsaturated (liquid) Complex Lipids Triglycerides (fats) Phospholipids Ex) cell membrane Waxes Ex) butter Ex) earwax Steroids Ex) testosterone, cholesterol 4. Nucleic Acids Nucleotide = monomer Found in the genetic material DNA & RNA carries out all of the functions of the cell