Lecture 17 - New CombinedPrimary and secondary hemostasis
advertisement

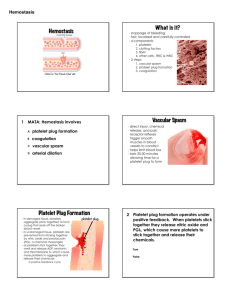
HEMOSTASIS Primary and Secondary Hemostasis HEMOSTASIS Hemostasis The process by which the body stops bleeding upon injury and maintains blood in the fluid state in the vascular compartment Process is rapid and localized HEMOSTASIS The primary players in hemostasis include Blood vessels Platlets Plasma proteins Coagulation proteins – involved in clot formation Fibrinolysis – involved in clot dissolution Serine protease inhibitors Other minor players include Kinin system Complement system HEMOSTASIS Defects In blood vessels, platlets or serum proteins can be corrected by utilization of the other 2 players In 2 of the 3 players results in pathologic bleeding Blood Vessels Platlets Plasma Proteins HEMOSTASIS Hemostasis can be divided into two stages Primary hemostasis Response to vascular injury Formation of the “platelet plug” adhering to the endothelial wall Limits bleeding immediately Secondary Hemostasis Results in formation of a stable clot Involves the enzymatic activation of coagulation proteins that function to produce fibrin as a reinforcement of the platelet plug Gradually the stable plug will be dissolved by fibrinolysis FORMATION OF A STABLE PLUG VASCULAR SYSTEM Smooth and continuous endothelial lining is designed to facilitate blood flow Intact endothelial cells inhibit platelet adherence and blood coagulation Injury to endothelial cells promotes localized clot formation Vasoconstriction Narrows the lumen of the vessel to minimize the loss of blood Brings the hemostatic components of the blood (platelets and plasma proteins) into closer proximity to the vessel wall Enhances contact activation of platlets Von Willebrand factor Collagen fibers Platlet membrane glycoprotein Ib Activated platlets enhance activation of coagulation proteins PRIMARY HEMOSTASIS Platelets Interact with injured vessel wall Interact with each other Produce the primary hemostatic plug Primary platelet plug Fragile Can easily be dislodged from the vessel wall PLATELETS Platelets Small, anucleated cytoplasmic fragments Released from megakaryocytes in the BM Megakaryocyte proliferation is stimulated by thrombopoietin (TPO) Normal platlet count is 150-400 x 109/L Survive 9-12 days Nonviable or aged platelets removed by spleen & liver 2/3 of platelets circulate in the peripheral blood 1/3 are sequestered in the spleen Humoral factor Produced primarily by liver, kidney, spleen, BM Produced at a relatively constant rate These 2 pools are in equilibrium and constantly exchanging Spontaneous hemorrhaging occurs when platlet count gets below 10 x 109/L PLATLETS MATURE MAGAKARYOCYTE PLATLET RELEASE PLATLET FUNCTION Platlets function to Provide negatively charged surface for factor X and prothrombin activation Release substances that mediate vasoconstriction, platlet aggregation, coagulation, and vascular repair Provide surface membrane proteins to attach to other platlets, bind collagen, and subendothelium PLATELETS Are the primary defense against bleeding Circulate in resting state Have minimal interaction with other blood components or the vessel wall Morphology of resting platelet is smooth, discoid When stimulated by endothelial damage, platlets become activated and they Become round and ‘sticky’ Build a hemostatic plug Provide reaction surface for proteins that make fibrin Aid in wound healing Platlet activation and plug formation involves Adhesion Shape change Secretion Aggregation FORMATION OF PRIMARY HEMOSTATIC PLUG PLATELETS AND SECONDARY HEMOSTASIS Primary platelet plug is Unstable and easily dislodged Secondary hemostasis Fibrin formation stabilizes and reinforces the platelet plug Proteins interact to form fibrin assemble on negatively charged membrane phospholipids of activated platelets System mediated by many coagulation factors present in an inactive form in blood. Factors are assigned Roman numerals, I through XIII All are produced in the liver. The von Willebrand factor is also produced in endothelial cells and megakaryocytes. SECONDARY HEMOSTASIS Coagulation factors are divided into three categories based on hemostatic function Substrate –fibrinogen (Factor I), which is the main substrate used to make fibrin Co-factors – accelerate enzymatic reactions Enzymes Coagulation factors are also classified by physical properties Contact proteins Involved in earliest phases of clotting Partially consumed during coagulation Found in serum Prothrombin Group Vitamin-K Dependant Clotting Factors Most are found in serum SECONDARY HEMOSTASIS Fibrinogen Group Thrombin-Sensitive Clotting Factors Some drugs prevent clotting by acting as antagonists to Vitamin K (Warfarin and Coumadin) All are acted upon by thrombin in the process of blood coagulation None found in serum The cascade theory of blood coagulation Involves a series of biochemical reactions Transforms circulating substances into an insoluble gel through conversion of fibrinogen to fibrin Requires Plasma proteins Phospholipids calcium CASCADE THEORY OF COAGULATION Each coagulation factor is converted to an active form by the preceeding factor in the cascade Calcium participates in some of the reactions as a co-factor The blood coagulation cascade occurs on cell surface membranes. The membrane localizes the reaction to the site of injury SECONDARY HEMOSTASIS Three different complexes assemble on the phospholipid membrane The pathways for the formation of these complexes are Intrinsic Extrinsic Common -Both intrinsic and extrinsic pathways converge to share factors in the common pathway Both intrinsic and extrinsic pathways require initiation Intrinsic - all factors involved in clot formation are in the vascular compartment Extrinsic- is initiated when a tissue factor not found in blood enters the vascular system COMPLEXES ON MEMBRANE Extrinsic pathway Intrinsic pathway Common pathway Fibrin formation EXTRINSIC PATHWAY INTRINSIC PATHWAY COMMON PATHWAY Intrinsic and extrinsic pathways Converge on the common pathway In the final steps thrombin converts fibrinogen to soluble fibrin and the fibrin monomers are crosslinked to form a stable fibrin polymer. COMMON PATHWAY COAGULATION CASCADE INHIBITION OF COAGULATION Antithrombin (AT) is a potent physiologic inhibitor of thrombin, and several other factors involved in coagulation In the presence of heparin, the inactivation of thrombin by AT is significantly increased INHIBITOR PATHWAY OF COAGULATION SUMMARY OF PRIMARY AND SECONDARY HEMOSTASIS Sequence after vessel injury Vasoconstriction Platelet adhesion Controlled by vessel smooth muscle; enhanced by chemicals secreted by platelets Adhesion to exposed subendothelial connective tissue Platelet aggregation Interaction and adhesion of platelets to one another to form initial plug at injury site SUMMARY OF PRIMARY AND SECONDARY HEMOSTASIS Sequence cont’d Fibrin-platelet plug Coagulation factors interact on platelet surface to produce fibrin; fibrin-platelet plug then forms at site of vessel injury Fibrin stabilization Fibrin clot must be stabilized by F-XIIIa FIBRINOLYSIS Activation of coagulation also activates fibrin lysis Fibrinolysis results in a gradual enzymatic cleavage of fibrin to soluble fragments Due to the activity of plasmin which is responsible for degradation of fibrin Limits the extent of the hemostatic process Reestablishes normal blood flow PLASMIN ACTION FDP= fibrin degradation products KININ AND COMPLEMENT SYSTEMS The kinin system is also activated by both coagulation and fibrinolytic systems The kinin system is important in inflammation, vascular permeability, and chemotaxis The complement system is activated by plasmin INTERRELATIONSHIP OF COAGULATION, FIBRINOLYTIC, KININ, AND COMPLEMENT SYSTEMS HEMOSTATIC BALANCE The regulation of hemostatic and fibrinolytic processes is dynamic Balance between Pro- and anti-hemostatic mediators Pro- and anti-fibrinolytic mediators Balance can be upset if any components are Inadequate Excessive Development of thrombi Excessive local or systemic activation of coagulation Sustained bleeding Excessive local or systemic fibrinolytic activity HEMOSTATIC BALANCE When hemostasis is delayed Either platelet disorder or a coagulation defect Bleeding episode may be prolonged Imbalance created between An abnormally slow hemostatic rate A normal rate of fibrinolysis An inadequate fibrinolytic response May retard lysis of a thrombus and even contribute to its extension BALANCE OF CLOTTING AND FIBRINOLYSIS DIAGNOSIS OF BLEEDING PROBLEMS Questions to address: Is a bleeding tendency present? Is the condition familial or acquired? Is the disorder one affecting Primary hemostasis (platelet or blood vessel wall problems) Secondary hemostasis (coagulation problems) Is there another disorder present that could be the cause of or might exacerbate any bleeding tendency? Principal Presentations of bleeding disorders Easy bruising Spontaneous bleeding from mucous membranes Menorrhagia – excessive bleeding during menstruation Excessive bleeding after trauma LABORATORY EVALUATION OF HEMOSTASIS Three different categories of disorders may be found Vascular and platlet disorders Coagulation factor deficiencies or specific inhibitors Fibrinolytic disorders Bleeding disorders present differently depending upon the causative problem Platlet disorders present as petechiae and bleeding into mucous membranes because of failure to form the platlet plug Patients with coagulation defects (includes those with hemophilia) may develop deep spreading hematomas and bleeding into the joints with evident hematuria because of failure to reinforce the platlet LABORATORY EVALUATION Tests to differentiate between these include Platlet count Peripheral blood smear evaluation Ivy bleeding time (N=2.5-9.5 min) or platlet function analyzer (PFA) Prothrombin time (PT) – test contains thromboplastin and calcium chloride and measures measures the extrinsic and common pathways (Normal=11-13 sec) Activated partial thromboplastin time (APTT) contact activators and a platlet substitute and calcium chloride are added to measure the intrinsic and common pathways (Normal usually 23-35 sec, may vary depending upon analyzer used, reagents used, and patient population) LABORATORY EVALUATION Thrombin time (TT) – add thrombin and measure the time required for thrombin to convert fibrinogen to fibrin (common pathway) (N=15-22 sec) Mixing studies with PT and APTT abnormal results patient plasma is mixed with normal plasma to distinguish between factor deficiencies and coagulation inhibitors If assay is corrected – due to factor deficiency If partially corrected or uncorrected – due to inhibitor Coagulation factor assays Assays for fibrin degradation products – evidence of fibrinolysis INHERITED QUALITATIVE PLATELET DISORDERS Defects in platelet-vessel wall interaction Most common disorder is von Willebrand disease Deficiency or defect in plasma VWF Defects in platelet-platelet interaction Defects of platelet secretion and signal transduction Abnormalities of platelet granules Defects in platlet coagulant activity LAB TESTS IN DISORDERS OF PRIMARY HEMOSTASIS Platlet count PT APTT Bleeding time Vascular disorder Normal Normal Normal Normal or abnormal Thrombocytopenia Decreased Normal Normal Abnormal Platlet Dysfunction Usually Normal Normal Normal or Abnormal Normal DRUGS THAT ALTER PLATELET FUNCTION A variety of drugs alter platelet function Some are used therapeutically for their antithrombotic activity For others, abnormal platelet function is an unwanted side effect Effect on platelet function Defined by an abnormality of bleeding time or platelet aggregation Aspirin Inhibits platlet aggregation Inhibits platlet secretion DISORDERS OF SECONDARY HEMOSTASIS Hereditary vs acquired Quantitative vs qualitative deficiencies Laboratory screening tests (PT, APTT) Does not differentiate quantitative vs qualitative disorders Qualitative abnormal proteins will Prolong clotting test Be recognized by immunologically-based procedures Activity assays Essential when screening for deficiencies VON WILLEBRAND DISEASE Inherited hemorrhagic disorder Genetically and clinically heterogeneous Caused by a deficiency/dysfunction of VWF Most common hereditary bleeding disorder VWF Multimeric blood protein Performs two major roles in hemostasis Mediates adhesion of platelets to sites of vascular injury Is a carrier protein for F-VIII Inherited defects in VWF may Interfere with biosynthetic processing or disrupt specific ligand binding sites Cause bleeding by impairing either platelet adhesion or blood clotting HEMOPHILIAS Hemophilia A Factor VIII Deficiency Hemophilia B Factor IX Deficiency Antihemophilic Factor X-linked recessive disorder Most common type of hemophilia Christmas Factor (from family of first patients diagnosed with the disorder) X-linked recessive disorder Hemophilia C Factor XI Deficiency Autosomal recessive disorder seen primarily in the Ashkenazi Jewish population Symptoms range from mild to severe HEMOPHILIA Insufficient generation of thrombin by F-IXa/VIIIa complex through the intrinsic pathway of coagulation cascade Bleeding severity complicated by excessive fibrinolysis Clinical severity corresponds with level of factor activity Severe hemophilia Factor coagulant activity <1% of normal Frequent spontaneous bleeding into joints and soft tissues Prolonged bleeding with trauma or surgery HEMOPHILIA Moderate hemophilia Factor coagulant activity 1-5% of normal Occasional spontaneous bleeding Excessive bleeding with surgery or trauma Mild hemophilia Factor coagulant activity >5% of normal Usually no spontaneous bleeding Excessive bleeding with surgery or trauma HEMOPHILIA – CLINICAL PRESENTATION Readily diagnosed In severe disease and patients with prior family history Diagnosis based on Unusual bleeding symptoms early in life Age of first bleeding varies with severity of disease Family history Physical exam Laboratory evaluation HEMOPHILIA – TREATMENT Replacement of clotting factor to achieve hemostasis Annual cost for patient with severe hemophilia $20,000-100,000 Various products available Plasma-derived low, intermediate and high purity products Plasma-derived ultrapure products Ultrapure recombinant products Replacement products – benefits vs risks Blood-born pathogens Hepatitis A, B, C, G; HIV, Parvovirus B-19 Thrombotic complications with some F-IX concentrations Development of alloantibody inhibitors Neutralize coagulant effects of replacement therapy COAGULATION SCREENING TESTS IN CONGENITAL DEFICIENCIES Platlet PT count APTT PFA TT Congenital Deficiency N N N N N XIII, mild deficiency of any factor, plasminogen activator inhibitor-1, α2 antiplasmin N A N N N VII – (extrinsic pathway) N N A N N XII, XI, IX, VIII, prekallikrein, high molecular weight kininogen (intrinsic pathway – includes hemophilias) N A A N N X, V, II (common pathway) N A A N A Fibrinogen (last part of common pathway) N N A or N A or N N Von Willibrands