Formal Charge & Resonance Structures Chemistry Presentation
advertisement

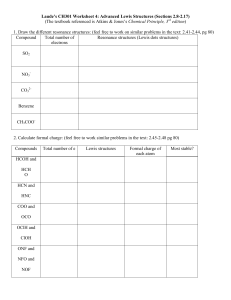
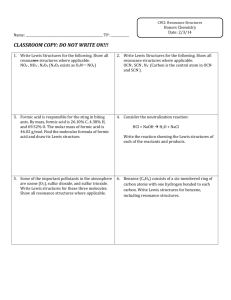
Formal Charge & Resonance Structures These ARE NOT Cornell Notes Learning Objectives • Determine whether a Lewis structure is plausible by calculating formal charges. • Explain why resonance occurs, and identify resonance structures. Review • What is electronegativity? (see notes on page 127 of notebook) • What trend does electronegativity follow in the periodic table? (see labeled periodic table on page 128 of notebook) Question • Is there a way to determine whether our Lewis structure is plausible? Formal Charge • Formal charge is the difference between the number of valence electrons in a free (uncombined) atom and the number of valence electrons assigned to that atom when bonded to others in a Lewis structure. Formal charge = (valence e-) – (# of bonds) – (non-bonding e-) Examples Formal Charge • Usually, the most plausible Lewis structure is one with formal charges of zero on all atoms. Formal Charge • Where non-zero formal charges are required, they should be as small as possible, and negative formal charges should appear on the most electronegative atoms. Formal Charge • Adjacent atoms in a structure should not carry formal charges of the same sign. Formal Charge • The total of formal charges on the atoms in a Lewis structure must be zero for a neutral molecule and must equal the net charge for a polyatomic ion. Example #1 (p174) Draw a Lewis structure for NCl3. Calculate the formal charges on each atom. Example #2 (p174) A student has proposed two condensed structural formulas—H2NOH and H2ONH—for a compound with the molecular formula H3NO. Write a Lewis structure corresponding to each formula, assign formal charges, and select the more plausible Lewis structure. Practice #1 (p 175) • Draw a Lewis structure for CH2O. Calculate the formal charges on each atom. Practice #2 (p175) Draw the Lewis resonance structures for NO2- Resonance • Using the same sequence of atoms, it is possible to have more than one correct Lewis structure when a molecule or polyatomic ion has both a double and a single bond. Resonance • Resonance is a situation in which a molecule or ion can be represented by two or more plausible Lewis structures that differ only in the distribution of electrons. • The true structure is a composite of all possible Lewis structures. • The different plausible structures are called resonance structures. Example #1 Draw the Lewis resonance structures for NO2- Example #2 Draw the Lewis resonance structures for SO2. Practice Draw the Lewis resonance structures for O3.