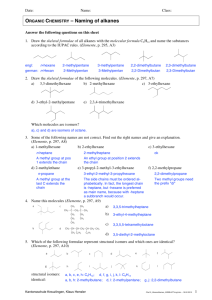
analysis of electron localization/delocalization and aromaticity in
advertisement

27-28/09/2007
UAM
ANALYSIS OF ELECTRON LOCALIZATION/DELOCALIZATION AND AROMATICITY IN MOLECULES
1
ANALYSIS OF ELECTRON
LOCALIZATION/DELOCALIZATION
AND AROMATICITY IN
MOLECULES
Miquel Solà
Institut de Química Computacional
Universitat de Girona
27-28/09/2007
UAM
ANALYSIS OF ELECTRON LOCALIZATION/DELOCALIZATION AND AROMATICITY IN MOLECULES
2
OUTLINE
I.
INTRODUCTION
II.
ELECTRON LOCALIZATION AND DELOCALIZATION
III. AROMATICITY
IV. EXERCICES
27-28/09/2007
UAM
ANALYSIS OF ELECTRON LOCALIZATION/DELOCALIZATION AND AROMATICITY IN MOLECULES
3
INTRODUCTION
Conjugation, hyperconjugation, and aromaticity are
often invoked in organic chemistry textbooks and
research works to explain the structure, stability
and reactivity of molecules.
Structure and stability are important properties
for the design of new materials.
27-28/09/2007
UAM
According to ISI data, during 2005, the words
conjugat* or hyperconjugat* are found in 9863
papers and in every hour two papers appear in which
the term aromatic* is in the title, keywords or the
abstract!
Since 2001, there is a renovated interest in
aromaticity because of the all-metal aromatic species
(e.g. Al4-2,…).
ANALYSIS OF ELECTRON LOCALIZATION/DELOCALIZATION AND AROMATICITY IN MOLECULES
4
INTRODUCTION
Information on electron localization is necessary
to locate lone pairs, bonding pairs, unpaired
electrons, or p-electron subsystems in connection
with the Lewis theory and the VSEPR model.
Electron pair localization tools have been for long
pursued in quantum chemistry to discuss the nature
of the chemical bond.
Electronic localization/delocalization plays also a
key role in the analysis of electron fluctuation and
electron correlation effects (relevant for the
development of new functionals).
27-28/09/2007
UAM
ANALYSIS OF ELECTRON LOCALIZATION/DELOCALIZATION AND AROMATICITY IN MOLECULES
5
INTRODUCTION
Electronic localization/delocalization is not an
observable, and therefore there is no experimental
property that allows measuring it directly. However
electronic delocalization reveals itself in several
chemical phenomena.
Magnetic properties (magnetic susceptibility
and anisotropy)
UV spectra
High symmetry in IR and/or Raman spectra
High stability
Bond length equalization
27-28/09/2007
UAM
ANALYSIS OF ELECTRON LOCALIZATION/DELOCALIZATION AND AROMATICITY IN MOLECULES
6
INTRODUCTION
There is not a unique and widely accepted theoretical
measure of electronic localization/delocalization.
AIM theory (2r(r), e, DI)
Fermi hole maps
Domain-averaged Fermi holes
ELF
27-28/09/2007
UAM
ANALYSIS OF ELECTRON LOCALIZATION/DELOCALIZATION AND AROMATICITY IN MOLECULES
7
OUTLINE
I.
INTRODUCTION
II.
ELECTRON LOCALIZATION AND DELOCALIZATION
III. AROMATICITY
IV. EXERCICES
27-28/09/2007
UAM
ANALYSIS OF ELECTRON LOCALIZATION/DELOCALIZATION AND AROMATICITY IN MOLECULES
8
ELECTRON LOCALIZATION & DELOCALIZATION
Ψ(x1 , x2 , x3 ,..., xN )
x1 r1s1
Born probabilistic interpretation:
2
P[1e (x, x dx)] ψ(x) dx
2
P[1 (x1 , x1 dx1 ),..., N (x N , x N dx N )] ψ(x1 ,x 2 ...) dx1dx 2 ... dx N
Obviously, the probability of electrons being somewhere is 100%:
2
... ψ(x1 ,...) dx 1 dx 2 ...dxN 1
The probability of finding an electron in between x1 and x1+dx1
irrespective of the position of the rest of the electrons is given by:
27-28/09/2007
UAM
dx1 ( x1 ,x2 , xN ) * ( x1 , x2 , xN )dx2 dxN
ANALYSIS OF ELECTRON LOCALIZATION/DELOCALIZATION AND AROMATICITY IN MOLECULES
9
Given that electrons are indistinguishable particles :
r ( x1 )dx1 N dx1 ( x1 ,x2 , xN ) * ( x1 , x2 , xN )dx2 dxN
Density function
Integrating with respect to the spin coordinate we have:
r (r1 ) r ( x1 )ds1 N
( x1 ,x2 , xN ) * ( x1 , x2 , xN )ds1dx2 dxN
Given that is normalised:
Electron density
r1 (r )dr N
It is possible to separate the electron density corresponding
to and b electrons:
r (r1 ) N ( x1 ,x2 , xN ) * ( x1 , x2 , xN )ds1dx2 dxN
r1 (r1 ) N (r1 ,x2 , xN ) * (r1 , x2 , xN )dx2 dxN
27-28/09/2007
UAM
ANALYSIS OF ELECTRON LOCALIZATION/DELOCALIZATION AND AROMATICITY IN MOLECULES
or:
10
Since N = N + Nb we have:
b
r (r1 ) r (r1 ) r (r1 )
S
b
r (r1 ) r (r1 ) r (r1 )
Spin density
The probability of finding two electrons one in between x1 and x1+dx1
and the other in between x2 and x2+dx2, independently of where the
others are found is given by:
dx1dx2 ( x1 ,x2 , xN ) * ( x1 , x2 , xN )dx3 dxN
The second order density is defined as:
G( x1 , x2 ) N ( N 1) ( x1 ,x2 , xN ) * ( x1 , x2 , xN )dx3 dxN
where N(N-1) are the possible electron pairs that can be
formed. G(x1,x2)dx1dx2 consequently gives us the probability
of finding any electron between x1 and x1+dx1 and another
between x2 and x2+dx2.
27-28/09/2007
UAM
ANALYSIS OF ELECTRON LOCALIZATION/DELOCALIZATION AND AROMATICITY IN MOLECULES
11
Integrating over the spin coordinates one obtains the
two-electron density or pair function:
G(r1 , r2 ) G( x1 , x2 )ds1ds2
which gives us the probability of finding any two electrons, one
between r1 and r1 + dr1 and the other between r2 and r2 + dr2,
under any spin combination (,b,b,bb).
Taking into account that:
N(N-1) = (N+Nb)(N+ Nb-1) = N(N-1) + NNb + NbN + Nb(Nb-1):
it is also possible to separate the pair function in the
following four contributions:
b
b
bb
G(r1 , r2 ) G (r1 , r2 ) G (r1 , r2 ) G (r1 , r2 ) G (r1 , r2 )
where:
G (r1 , r2 ) N ( N 1) (r1s1 , r2 s2 , x3 xN ) * (r1s1 , r2 s2 , x3 xN )ds1ds2 dx3 dxN
and:
G (r1 , r2 ) N N b (r1s1 , r2 s2 , x3 xN ) * (r1s1 , r2 s2 , x3 xN )ds1ds2 dx3 dxN
b
27-28/09/2007
UAM
ANALYSIS OF ELECTRON LOCALIZATION/DELOCALIZATION AND AROMATICITY IN MOLECULES
12
Monodeterminantal wavefunctions
Closed-shell:
1
1 (1) 1 (2) 2 (3) N ( N )
N!
2
Electron density
r1 (r1 ) N
1
Pi
Pj
(
1
)
(
1
)
P
(
1
)
(
2
)
(
N
)
P
(
1
)
(
2
)
(
N
)
ds
d
x
d
x
j 1
1 2
i 1
1
N
1
N
N
N! Pi Pj
2
2
N
2
N
2
2
2
1
r1 (r1 ) N
( N 1)!.2 i (r1 ) 2 i (r1 )
N!
i 1
i 1
In the LCAO approximation: i (r1 ) c i (r1 )
N
2
*
*
*
r1 (r1 ) 2 ci ci (r1 ) (r1 ) P (r1 ) (r1 )
i 1
27-28/09/2007
UAM
N
2
P 2 c*i ci
i 1
ANALYSIS OF ELECTRON LOCALIZATION/DELOCALIZATION AND AROMATICITY IN MOLECULES
13
Monodeterminantal wavefunctions
Two-electron density
1
Pi
Pj
G(r1 , r2 ) N ( N 1) (1) (1) Pi 1 (1) 1 (2) N ( N )Pj 1 (1) 1 (2) N ( N )ds1ds2 dx3 dxN
N! Pi Pj
2
2
N
2
N
2
1
1
G(r1,r2 ) N ( N 1) ( N 2)!4 i*(r1 ) *j(r2 ) i (r1 ) j (r2 ) i*(r1 ) *j(r2 ) j (r1 ) i (r2 )
N!
2
i
j
N
2
N
2
1
G(r1,r2 ) 4 i*(r1 ) *j(r2 ) i (r1 ) j (r2 ) i*(r1 ) *j(r2 ) j (r1 ) i (r2 )
2
i
j
In the LCAO approximation:
N
2
N
2
i (r1 ) c i (r1 )
1
G(r1,r2 ) 4 c*i c*j ci cj c*i c*j cj ci * (r1 )* (r2 ) (r1 ) (r2 )
2
i
j
G(r1,r2 ) G* (r1 )* (r2 ) (r1 ) (r2 )
G
27-28/09/2007
UAM
N
2
N
2
1
1
4 c*i c*j ci cj c*i c*j cj ci P P P P
2
2
i
j
ANALYSIS OF ELECTRON LOCALIZATION/DELOCALIZATION AND AROMATICITY IN MOLECULES
Exercices
1 to 4
14
More definitions
G(r1 , r2 ) r (r1 ) r (r2 ) GXC (r1 , r2 )
G(r1 , r2 )
r (r1 ) r (r2 )
GXC (r1 , r2 )
27-28/09/2007
UAM
Probability density of finding simultaneously
an electron in r1 and another one in r2
Probability density of finding an electron in r1
and another one in r2, considering the
electrons as independent particles
Exchange-correlation density
Difference between the conditional and
unconditional probabilities, which is a measure of
the degree to which the density is excluded at
r2 because of the presence of an electron at r1
GXC (r1 , r2 )d r1d r2 N
ANALYSIS OF ELECTRON LOCALIZATION/DELOCALIZATION AND AROMATICITY IN MOLECULES
15
G(r1 , r2 )
P(r1 , r2 )
r (r1 )
P(r1 , r2 )
Conditional pair density
Probability density of finding an electron at
position r2 when one electron is known to
be at reference position r1
G(r1 , r2 ) r (r1 ) P(r1 , r2 )
P(r1 , r2 ) r (r2 ) r XC (r1 , r2 )
r XC (r1 , r2 )
27-28/09/2007
UAM
P(A U B) = P(A) PA(B)
GXC (r1 , r2 )
r XC (r1 , r2 )
r (r1 )
Exchange-correlation hole
Term that must be added to the unconditional
probability of finding an electron at position r2 to
get the conditional pair density
ANALYSIS OF ELECTRON LOCALIZATION/DELOCALIZATION AND AROMATICITY IN MOLECULES
16
b
b
bb
G(r1 , r2 ) G (r1 , r2 ) G (r1 , r2 ) G (r1 , r2 ) G (r1 , r2 )
G (r1 , r2 ) r (r1 ) r (r2 ) GXC (r1 , r2 )
GXC (r1 , r2 )
G (r1 , r2 )
P (r1 , r2 ) r (r2 )
r (r2 ) r XC (r1 , r2 )
r (r1 )
r (r1 )
r XC (r1 , r2 )
r XC r1 , r2 dr2 1
Fermi hole
Negative quantity determining the decrease in
the probability of finding an electron of the same
spin at position r2 when one electron is known to
be at reference position r1
b
GXC (r1 , r2 )
G (r1 , r2 )
b
b
b
b
P (r1 , r2 ) r (r2 )
r (r2 ) r XC (r1 , r2 )
r (r1 )
r (r1 )
b
b
Coulomb hole
r XC (r1 , r2 )
r XC r1 , r2 dr2 0
b
At the HF level:
27-28/09/2007
UAM
r XC (r1 , r2 ) 0
b
ANALYSIS OF ELECTRON LOCALIZATION/DELOCALIZATION AND AROMATICITY IN MOLECULES
17
b
r XC (r1 , r2 ) r XC (r1 , r2 ) r XC (r1 , r2 )
r XC r1 , r2 dr2 1
Sum rule
An important property of the Fermi hole is that its on-top value
(i.e., the Fermi hole when r1=r2) equals r (r2 ), thus ensuring the
complete removal of all like-spin electrons in the position of the
reference electron.
G (r1 , r1 ) 0
r XC (r1 , r1 ) r (r1 )
Exercices
5 to 8
If this total exclusion of the same-spin density persists as r2 is
displaced from r1, then the hole describes a region of space from
which all other same-spin electrons are excluded (localized Fermi
hole).
27-28/09/2007
UAM
ANALYSIS OF ELECTRON LOCALIZATION/DELOCALIZATION AND AROMATICITY IN MOLECULES
18
Monodeterminantal wavefunctions
Exchange-correlation density
N
2
N
2
1
G(r1,r2 ) 4 i*(r1 ) *j(r2 ) i (r1 ) j (r2 ) i*(r1 ) *j(r2 ) j (r1 ) i (r2 )
2
i
j
N
2
2
r1 (r1 ) 2 i (r1 )
i 1
GXC ( r1 , r2 ) G( r1 , r2 ) r ( r1 ) r ( r2 )
N
2
N
2
i
j
*
*
GXC(r1,r2 ) 2 i (r1 ) j (r2 ) j (r1 ) i (r2 )
GXC (r1 , r2 )
r XC (r1 , r2 )
r (r1 )
27-28/09/2007
UAM
r XC(r1,r2 )
N
2
N
2
i
j
*
2 (r1 ) j (r2 ) j (r1 ) i (r2 )
*
i
r1 r1
ANALYSIS OF ELECTRON LOCALIZATION/DELOCALIZATION AND AROMATICITY IN MOLECULES
19
H2 molecule
RH-H=5.0 bohr
RH-H=1.4 bohr
0,05
0,05
0
1
2
0,00
Fermi
Hole
3
*
4
5
6
7
8
-0,05
-0,05
-0,10
-0,10
-0,15
-0,15
-0,20
-0,20
-0,25
-0,25
0,05
1
2
0,00
3
*
4
5
6
7
2
3
4
5
6
7
8
8
0,10
-0,05
0,05
-0,10
0,00
-0,15
-0,05
-0,20
-0,10
-0,25
-0,15
0,05
0
1
2
3
4
5
6
7
8
0
1
2
3
4
5
6
7
8
0,05
0
0,00
1
0,15
0
Coulomb
Hole
0
0,00
1
2
3
*
4
5
6
7
8
0,00
-0,05
-0,05
Total
Hole
-0,10
-0,10
-0,15
-0,15
-0,20
-0,20
-0,25
-0,25
27-28/09/2007
UAM
-0,30
ANALYSIS OF ELECTRON LOCALIZATION/DELOCALIZATION AND AROMATICITY IN MOLECULES
20
• Fermi holes for N2
A
B
A Reference electron at
bond critical point
B Reference electron at
N nucleus
C
C Reference electron at
one N lone pair
The spatial localization of the corresponding hole
density function will be related to the degree of
localization of the reference electron.
In a closed-shell molecule, the spatial localization of a given
electron in a certain region implies the same behavior for an
electron of opposite spin, thus forming a localized ,b pair.
27-28/09/2007
UAM
ANALYSIS OF ELECTRON LOCALIZATION/DELOCALIZATION AND AROMATICITY IN MOLECULES
21
r XC r1, r2dr2 1
r (r1 ) r XC (r1 , r2 ) GXC (r1 , r2 )
r (r1 ) r XC r1 , r2 dr2 r (r1 )
A r (r1 ) r XC r1, r2 dr1dr2 A GXC (r1, r2 )dr1dr2 N A
Atomic XC hole
r
(
r
)
r
r
,
r
d
r
1 XC 1 2 1
Domain averaged Fermi hole
A
GXC (r1, r2 )dr1dr2 r (r1 ) r XC r1, r2 dr1dr2 r (r1 )dr1 N A
A
A
A
A hole density function integrates to the negative of the electron density that
it excludes. The usual point hole functions fix an electron in a point of space,
and thus integrate to -1. On the other hand, atomic holes exclude the electron
density associated to a whole atom, and they must integrate to the negative of
27-28/09/2007 the population of that atom.
UAM
ANALYSIS OF ELECTRON LOCALIZATION/DELOCALIZATION AND AROMATICITY IN MOLECULES
22
N2
r XC (r1 , r2 ) r (r1 ) r XC r1 , r2 dr1
A
A
A XC hole: reference
electron at one N lone pair
B
B Atomic XC hole: basin
of a N atom as a
reference
Localization index
A GXC r1 , r2 dr1dr2
AA
Delocalization index
A, B GXC r1 , r2 dr1dr2 GXC r1 , r2 dr1dr2 2 GXC r1 , r2 dr1dr2
A B
B A
GXC (r1, r2 )dr1dr2 N A
AB
1
δA, B N A
2 AB
A
R.F.W. Bader and M.E. Stephens J. Am. Chem. Soc. 1975, 97, 7391
δA, B 0
27-28/09/2007
UAM
λ(A)
X. Fradera, M.A. Austen and R.F.W. Bader J. Phys. Chem. A 1999, 103, 304
ANALYSIS OF ELECTRON LOCALIZATION/DELOCALIZATION AND AROMATICITY IN MOLECULES
23
Statistical interpretation
r (r1 )dr1 N A N A
A
2
G
(
r
,
r
)
d
r
d
r
(
r
,
r
,
r
)
N
N
*
(
r
,
r
,
r
)
d
r
d
r
d
r
d
r
1 2
N
1 2
3
N
1 2 1 2 1 2 N
A A
AA
2
2
G
r
,
r
d
r
d
r
N
N
N
A
A
1 2 1 2
AA
N A N
2
2
A
N
A
N
A
2
A
N A G r1 , r2 d r1d r2 N A r (r1 )dr1 r (r2 )dr2
2
AA
A
A
N A G r1 , r2 r (r1 ) r (r2 )d r1d r2 N A
2
AA
27-28/09/2007
UAM
ANALYSIS OF ELECTRON LOCALIZATION/DELOCALIZATION AND AROMATICITY IN MOLECULES
24
Statistical interpretation
N A GXC r1 , r2 d r1d r2 N A
2
AA
A GXC r1 , r2 d r1d r2
AA
2 N A A N A
1
λ(A) δA, B N A
2 AB
1
N A ( A, B)
2 B A
2
27-28/09/2007
UAM
ANALYSIS OF ELECTRON LOCALIZATION/DELOCALIZATION AND AROMATICITY IN MOLECULES
25
A GXC r1 , r2 dr1dr2
AA
A, B 2 GXC r1 , r2 dr1dr2
AB
G(r1 , r2 ) r (r1 ) r (r2 ) GXC (r1 , r2 )
G(r1 , r2 )
*
D r1 j r1 k r2 l r2
*
ijkl i
i , j , k ,l
A
A, B 2
Dijkl Sij AS kl A N A
2
i , j , k ,l
D
ijkl
Sij AS kl B 2 N AN B
i , j , k ,l
At the HF (or KS) level
N N
*
*
G xc (r1 , r2 ) i (r1 )i (r2 ) j (r1 ) j (r2 )
i 1 j 1
2
A Sij A
i, j
; A, B 2 Sij A Sij B
i, j
The sums are over occupied molecular spin-orbitals.
27-28/09/2007
UAM
ANALYSIS OF ELECTRON LOCALIZATION/DELOCALIZATION AND AROMATICITY IN MOLECULES
26
A GXC r1 , r2 dr1dr2
A, B 2 GXC r1 , r2 dr1dr2
AA
AB
Fulton bond orders
N N
*
1/ 2 1/ 2 *
G xc (r1 , r2 ) ni n j i (r1 )i (r2 ) j (r1 ) j (r2 )
i 1 j 1
A. M. K. Müller Phys. Rev. Lett. 1984, 105, 446
M. A. Buijse and E. J. Baerends Mol. Phys. 2001, 100, 401
A ni Sij A 2
i, j
A, B 2 ni1/ 2 n1j / 2 Sij A Sij B
i, j
R. L. Fulton J. Phys. Chem. 1993, 97, 7516
27-28/09/2007
UAM
ANALYSIS OF ELECTRON LOCALIZATION/DELOCALIZATION AND AROMATICITY IN MOLECULES
27
2
A Sij A
i, j
; A, B 2 Sij A Sij B
i, j
Sij A i * (r ) j (r )dr
A
Localization and delocalization indices
can be separated into and p
contributions (disconnected orbitals).
A, B [2 Sij A Sij B] i A, B
i
j
i
Al422p e
2 e
4 e
2 e
For monodeterminantal WF, localization and delocalization
indices can be separated into and b contributions.
27-28/09/2007
UAM
ANALYSIS OF ELECTRON LOCALIZATION/DELOCALIZATION AND AROMATICITY IN MOLECULES
28
N-center DIs
Three-center DI
A, B, C 12 Sij A S jk BSki C
Exercices
9 and 10
i , j ,k
Six-center DI
A, B, C, D, E, F 24
S A S BS CS DS E S F
ij
i , j , k ,l , m , n
n-center DI
jk
kl
lm
mn
ni
A = {A1, A2, …, AN}
( A) 4N
occ. MO
S A S A S A
i1i2
1
i2i3
2
i N i1
N
i1 ,i2 ,,i N
27-28/09/2007
UAM
The factor 4N comes from 2N ways to follow the ring (starting from
each of the N atoms and moving clockwise or anticlockwise) and 2
for and b in closed-shell species.
ANALYSIS OF ELECTRON LOCALIZATION/DELOCALIZATION AND AROMATICITY IN MOLECULES
29
λ(A) δA, B N
A
A
A B
Saa A Saa B
1
2
A, B 4 Saa ASaa B 1
2p electron systems (Dnh)
A 2(Saa A )2 0.5
Saa A Saa B
1
4
A, B 4 Saa ASaa B 0.25
A 2(Saa A )2 0.125
Saa A Saa B
1
6
A, B 4 Saa ASaa B 1 / 9
A 2(Saa A )2 1 / 18
Saa A Saa B
n edges (Dnh)
A, B 4 Saa A Saa B 4 / n
A 2(Saa A ) 2 / n
2
27-28/09/2007
UAM
1
n
1 e localized
1 e delocalized
0.5 e localized
1.5 e delocalized
1/3 e localized
5/3 e delocalized
2/n e localized
2
2(n-1)/n e delocalized
2
ANALYSIS OF ELECTRON LOCALIZATION/DELOCALIZATION AND AROMATICITY IN MOLECULES
30
The partition of the molecular space
Sij A i * (r ) j (r )dr
Mulliken partition
A
GXC (r1 , r2 )d r1d r2 N
N N
*
*
G xc (r1 , r2 ) i (r1 )i (r2 ) j (r1 ) j (r2 )
i 1 j 1
i (r1 ) Ci (r1 )
P
* *
ci r1 ci r2 cj r1 cj r2 dr1dr2 N
N
N
*
i 1 j 1
*
P
27-28/09/2007
UAM
N
P Ci C*i
i
ANALYSIS OF ELECTRON LOCALIZATION/DELOCALIZATION AND AROMATICITY IN MOLECULES
31
*
P P r1 r2 r1 r2 dr1dr2 N
*
*
P P r1 r1 dr1 r2 r2 dr2 N
*
P P S S
N
P S P S N
PS PS N
PS PS N
PS PS
Number of electrons shared by μ and ν
BAB 2 PS PS
A B
I. Mayer, Int. J. Quantum Chem. 1986, 29, 477
27-28/09/2007
UAM
BAB ( A, B)
ANALYSIS OF ELECTRON LOCALIZATION/DELOCALIZATION AND AROMATICITY IN MOLECULES
32
The Atoms-in-Molecules (AIM) partition
At the HF or DFT level:
N
2
r1 (r1 ) i (r1 )
i 1
At a correlated level (Configuration Interaction):
l
l
*
r1 (r1 ) i (r1 ) rij j (r1 )
i 1 j 1
M
M
rij cK cL* ijKL
K
L
ijKL
(1) p q if K and L differ only in i and j spinorbita ls
1
if K L and i j
0
other cases
Experimental densities: X-ray diffraction at low temperatures using
nonspherical multipolar refinement procedure and synchrotron
radiation.
27-28/09/2007
UAM
ANALYSIS OF ELECTRON LOCALIZATION/DELOCALIZATION AND AROMATICITY IN MOLECULES
33
0.01
0.02
0.03
0.04
LiF
The use of r(r),r(r), and 2r(r) allows:
o To partition the space defining atoms in molecules
o To define the molecular graph
o To determine the physical properties of the defined atoms
o To analyze the nature of the chemical bonds
27-28/09/2007
UAM
ANALYSIS OF ELECTRON LOCALIZATION/DELOCALIZATION AND AROMATICITY IN MOLECULES
34
, ,
x y z
r (r )
From the gradient of the density you
can obtain gradient lines and the critical
points
r .n r
r .n 0
Gradient density flux
Zero flux surface
Quantum mechanics can be generalized to a
subsystem if the subsystem is bounded by
zero flux surfaces (open systems)
r (r ) 0
27-28/09/2007
UAM
Critical points
ANALYSIS OF ELECTRON LOCALIZATION/DELOCALIZATION AND AROMATICITY IN MOLECULES
35
Gradient lines do not cross each other
Gradient lines always originate and terminate at a critical point
(which may be infinite)
All gradient lines in the vicinity of a nucleus terminate at that
nucleus. A nucleus is said to behave as an attractor
The portion of the 3D space containing all gradient lines that
finish in a given nuclei is the so-called the basin of this atom
27-28/09/2007
UAM
ANALYSIS OF ELECTRON LOCALIZATION/DELOCALIZATION AND AROMATICITY IN MOLECULES
36
The point of minimum electron density following the ridge
between the two nuclei is a critical point named the bond
critical point (BCP)
There are BCPs between some but not
all pairs of nuclei in a molecule, only in
those where the electronic charge is
accumulated between them.
27-28/09/2007
UAM
ANALYSIS OF ELECTRON LOCALIZATION/DELOCALIZATION AND AROMATICITY IN MOLECULES
37
Partitioning of the 3D space allows to define atomic properties
inside molecules. For instance, atomic charges:
N A r r dr
q A Z A r r dr
A
2 N A N 2
A
N
2
A
A
N A N
A
Variance (fluctuation of charge)
Sij A i * (r ) j (r )dr
A
Dipole moments:
A r r r dr
A
A A
…and atomic polarizabilities, hyperpolarizabilities, magnetic
susceptibilities….and energies:
2
G (r )
N ' r (r , r ' )dr '
2m
G( A) G(r )dr
27-28/09/2007
UAM
Local kinetic density
Atomic kinetic energy
A
ANALYSIS OF ELECTRON LOCALIZATION/DELOCALIZATION AND AROMATICITY IN MOLECULES
38
Fuzzy-Atom & AIM partition
1
0,9
0,8
w ( r ) 1
wA r 0
0,7
0,6
A
A
0,5
0,4
0,3
0,2
0,1
0
C
LI:
DI:
27-28/09/2007
UAM
O
Sij A wA r i * (r ) j (r )dr
PA r
wA r
PB r
B
A
Bader’s partition
r A
1
wA (r )
0 otherwise
2
r r
rA rB
A
B
PA r 0.5 1
a AB
RAB
B A
RAB
I. Mayer, P. Salvador Chem. Phys. Lett. 2004, 383, 368
ANALYSIS OF ELECTRON LOCALIZATION/DELOCALIZATION AND AROMATICITY IN MOLECULES
39
AIM results
HF/6-311++G(2d,2p)
• H2 g2
Molecule Atom A (A) (A,B)
1.000 0.500 1.000
H2
H
7.000 5.479 3.042
N2
N
9.000 8.358 1.283
F2
F
LiF
Li 2.060 1.971 0.178
9.940 9.851
F
4.647 3.860 1.574
CO
C
9.354 8.567
O
5.227 4.121 2.210
CN
C
8.773 7.668
N
+
5.525 4.323 2.405
NO
N
8.475 7.273
O
g
Lewis model
u
Sii A Sii B
1
; Sij A Sij B 0
2
A Sij A 2 ; A, B 2 Sij A Sij B
i, j
g2 = u2
i, j
A,B cannot be considered a bond order.
X. Fradera, J. Poater, S. Simon, M. Duran and M. Solà, Theor. Chem. Acc. 2002, 108, 214.
27-28/09/2007
UAM
ANALYSIS OF ELECTRON LOCALIZATION/DELOCALIZATION AND AROMATICITY IN MOLECULES
40
Correlation effects / 6-311++G(2d,2p) basis set
molecule
H2
N2
LiF
CO
atom
H
N
Li
F
C
O
HF
0.500
5.479
1.971
9.851
3.860
8.567
(A)
DFT
0.500
5.477
1.969
9.810
3.946
8.240
CI
0.575
5.891
1.973
9.838
4.072
8.484
HF
1.000
3.042
0.178
(A,B)
DFT
1.000
3.046
0.221
CI
0.849
2.219
0.193
1.574
1.814
1.443
(A,B)
Species
CISD Fulton
H2
0.844
N2
2.419
CO
1.474
In equally shared system, Coulomb correlation causes electron
density to be removed from the vicinity of the interatomic surface
and to be concentrated in each atomic basin, with a decrease in the
number of electron pairs shared between the two atoms.
The introduction of Coulomb correlation is essential in the H2
dissociation process.
27-28/09/2007
UAM
DFT calculations are performed with the HF expressions using the
KS orbitals. DFT results are not better than the HF ones. DFT twoelectron density is calculated using the HF formalism.
ANALYSIS OF ELECTRON LOCALIZATION/DELOCALIZATION AND AROMATICITY IN MOLECULES
41
• Correlation effects – CISD/STO-3G
The introduction of Coulomb correlation is essential in the H2
dissociation process.
At the HF level, (A)=1/2 and (A,B)=1 irrespective of the H-H bond
length and basis set considered.
Exercice 14
u*
localization/delocalization
1,0
1sA H
0,8
H 1sB
g
0,6
H , H ' 1 8cocD S12 H 2
0,4
0,2
0,0
0,7
1,1
1,5
1,9
2,3
2,7
3,1
3,5
r(H-H) Å
At the CI level, the localizability of the electrons in the system turns
into the intuitive scheme expected for homolytic dissociation.
27-28/09/2007
UAM
E. Matito, M. Duran, M. Solà J. Chem. Educ. 2006, 83, 1243.
ANALYSIS OF ELECTRON LOCALIZATION/DELOCALIZATION AND AROMATICITY IN MOLECULES
42
Results from AIM-Mulliken-fuzzy partitions
179 different C–C bonds
27-28/09/2007E.
UAM
Matito, J. Poater, M. Solà, M. Duran, P. Salvador J. Chem. Phys. A 2005, 109, 9904
43
ANALYSIS OF ELECTRON LOCALIZATION/DELOCALIZATION AND AROMATICITY IN MOLECULES
Results from AIM-Mulliken-fuzzy partitions
27-28/09/2007
UAM
ANALYSIS OF ELECTRON LOCALIZATION/DELOCALIZATION AND AROMATICITY IN MOLECULES
44
Excited states and open-shell systems
H2
g
Saa A Saa B Sbb A Sbb B
1
2
Sab A Sba A Sab B Sba B
a2
b2
Singlet a1b1
u
A, B 2 Sij A Sij B
i, j
A, B 4 Saa ASaa B 1
A, B 4 Sbb ASbb B 1
A, B 2[Saa ASaa B Sbb ASbb B] 1
Triplet a1b1
A, B 2[Saa A Saa B Sbb A Sbb B Sab A Sab B Sba A Sba B ] 1 4(Sab A ) 2
H2- ground state a2b1
A, B 2[2 Saa A Saa B Sbb A Sbb B Sab A Sab B Sba A Sba B ] 1.5 4(Sab A ) 2
27-28/09/2007
UAM
ANALYSIS OF ELECTRON LOCALIZATION/DELOCALIZATION AND AROMATICITY IN MOLECULES
45
Excited states and open-shell systems
O2
9 and 7b electrons
symmetry
total
p
total
6.853
4.480
2.372
(O)
4.113
2.240
1.872
b
2.740
2.240
0.500
total
2.294
1.038
1.255
(O,O’)
0.775
0.519
0.255
b
1.519
0.519
1.000
The contribution to the DI is 1.04, corresponding to a perfectly delocalized
electron pair. The two b electrons with p symmetry are also perfectly shared. In
contrast, the DI for the four electrons is 0.26 electrons.
27-28/09/2007
UAM
X. Fradera and M. Solà J. Comput. Chem., 2002, 23, 1347
ANALYSIS OF ELECTRON LOCALIZATION/DELOCALIZATION AND AROMATICITY IN MOLECULES
46
The ground state of Cu+2-H2O
a1*
a1*
b1*
b2 (3dxz)
a2 (3dxy)
b1*
x
z
a1 (3dx2-y2)
b2 (3dxz)
a2 (3dxy)
a1 (3dx2-y2)
y
b1
a1
2A
1
27-28/09/2007
UAM
b1
a1
2B
1
ANALYSIS OF ELECTRON LOCALIZATION/DELOCALIZATION AND AROMATICITY IN MOLECULES
47
The ground state of Cu+2-H2O
Cu, [10s7p4d1f] of Wachters; O, H, 6-311++G(d,p)
State
2
A1
2
B1
LSDA
E XC = E X
BLYP
B3LYP
BHLYP
CCSD(T)
0.0
0.0
0.0
0.0
-10.7
-2.3
5.8
5.6
VWN
+ ao ( Eexact
- E LSDA
)+ a x E B88
+ ac ( ECLYP - EVWN
)
X
X
X + EC
C
Parameter Set
State
2
A1
2
B1
1
2
3
4
5
6
7
8
9
0.0
0.0
0.0
0.0
0.0
0.0
0.0
0.0
0.0
-6.6
-2.3
1.9
5.1
6.8
7.4
7.6
7.6
7.5
PS {i}: ao=0.i, ax=ac=1-0.i
27-28/09/2007
UAM
ANALYSIS OF ELECTRON LOCALIZATION/DELOCALIZATION AND AROMATICITY IN MOLECULES
48
The ground state of Cu+2-H2O
1,200
1,000
2
2
A1
0,800
0,600
2
B1
0,400
B1
0,800
Cu, O)
Spin density
1,000
2
0,600
A1
0,400
0,200
0,200
0,000
0,000
0,0
0,2
0,4
0,6
0,8
0,0
1,0
0,2
0,4
B1
(Cu,O)
(Cu,O) b
2
2
A1
0
0,2
0,4
0,6
ao
27-28/09/2007
UAM
0,8
1,0
0,6
0,8
1
ao
ao
1,000
0,900
0,800
0,700
0,600
0,500
0,400
0,300
0,200
0,100
0,000
0,6
0,8
1
1,000
0,900
0,800
0,700
0,600
0,500
0,400
0,300
0,200
0,100
0,000
2
A1
2
B1
0
0,2
0,4
ao
ANALYSIS OF ELECTRON LOCALIZATION/DELOCALIZATION AND AROMATICITY IN MOLECULES
49
The ground state of Cu+2-H2O
u*
a1*
g
b1*
H, H 1
H, H 1.5 4(Sab A ) 2
b2 (3dxz)
H, H 0.5 4(Sab A ) 2
2
a2 (3dxy)
a1 (3dx2-y2)
b1
B1
0,200
a1
0,150
Sa1a1* 0,100
2A
0,050
0,000
0
0,2
0,4
0,6
0,8
1
1
Sb1b1*(Cu) = ±0.24
Small increase in
…but more localized when ao increases!
ao
2B
1
Sa1a1*(Cu) = ±0.04
Large increase in
More delocalized!
J. Poater, M. Solà, A. Rimola, L. Rodríguez-Santiago, M. Sodupe, J. Phys. Chem A 2004, 108, 6072.
27-28/09/2007
UAM
ANALYSIS OF ELECTRON LOCALIZATION/DELOCALIZATION AND AROMATICITY IN MOLECULES
50
3c-2e and 3c-4e systems
(A)
molecule
CO2
B2H6
ato
m
C
O
HF
2.187
8.660
CI
2.443
8.547
B
H3
H5
2.103
1.257
1.103
2.207
1.235
1.100
H3
H7
B1
H4
H5
B2
H8
(A,B)
pair
C,O
O,O
B1,B2
B1,H3
B1,H5
B1,H7
H3,H4
H5,H6
H3,H5
H3,H7(c)
H3,H8(t)
HF
CI
1.056 1.075
0.380 0.313
0.047
0.473
0.264
0.017
0.134
0.237
0.116
0.011
0.015
0.033
0.477
0.282
0.009
0.120
0.194
0.103
0.021
0.017
pu*
pg
pu
The large (O,O’) of 0.3 electrons arises mainly from the pg orbital. A
large DI between nonbonded atoms is an indication of 3c-4e bonds.
FHF- and FFF- show also large (F,F’) between terminal fluorine atoms.
In B2H6, (B,H) is larger for terminal hydrogens than for bridge
hydrogens. (H,H’) between bridge hydrogens is larger than (B,B’).
27-28/09/2007
UAM
ANALYSIS OF ELECTRON LOCALIZATION/DELOCALIZATION AND AROMATICITY IN MOLECULES
51
3c-2e and 3c-4e systems
H3+:
4*3*1/3*1/3*1/3=0.444
For 3c-2e species, the 3-center DI is positive and for the 3c-4e
species it is negative.
R. Bochicchio, L. Lain, A. Torre, R. Ponec J. Math. Chem. 2000, 28, 83
27-28/09/2007
UAM
R. Ponec, I. Mayer J. Phys. Chem. A 1997, 101, 1738
ANALYSIS OF ELECTRON LOCALIZATION/DELOCALIZATION AND AROMATICITY IN MOLECULES
52
Bielectronic indices in reactivity
DIELS-ALDER
• Evolution of the most relevant delocalization indices along
the IRC of the Diels-Alder cycloaddition reaction between
butadiene and ethylene computed at the HF/6-31G* level:
27-28/09/2007
UAM
ANALYSIS OF ELECTRON LOCALIZATION/DELOCALIZATION AND AROMATICITY IN MOLECULES
53
Bielectronic indices in reactivity
2
(C1,C2)
(C1,C6)
(C4,C5)
(C5,C6
TS
1,8
1,6
1,4
1,2
1
0,8
0,6
0,4
0,2
0
4
5
3
6
2
27-28/09/2007
UAM
-5
-4
-3
-2
-1
0
1
2
3
4
Rx
1
4
5
3
6
2
ANALYSIS OF ELECTRON LOCALIZATION/DELOCALIZATION AND AROMATICITY IN MOLECULES
5
1
54
Bielectronic indices in reactivity
2
(C1,C2)
(C1,C6)
(C4,C5)
(C5,C6
TS
1,8
1,6
1,4
1,2
1
0,8
0,6
0,4
0,2
0
4
5
3
-5
6
2
1
-4
-3
-2
-1
0
Rx
1
2
3
4
4
5
5
3
6
2
1
• Aromatic TS, whose structure is nearly equivalent to that
of benzene from an electronic point of view, with the
difference that there is no s bonding associated to the C3C4 and C5-C6 pairs.
• Main changes in electron-pair reorganization take place
mainly in a small region around the TS.
27-28/09/2007
UAM
ANALYSIS OF ELECTRON LOCALIZATION/DELOCALIZATION AND AROMATICITY IN MOLECULES
55
The electron localization function (ELF)
GXC (r1 , r2 )
G (r1 , r2 )
P (r1 , r2 ) r (r2 )
r (r2 ) r XC (r1 , r2 )
r (r1 )
r (r1 )
P (r1 , r2 )dr2 r1 (r2 )dr2 r XC (r1 , r2 )dr2 N 1
If there is an electron located in r1, the probability to find another one with
the same spin in the whole space is N-1.
P (r1 , r1 ) 0
Becke proposes to make the spherical average of the conditional probability:
P (r1 , r2 ) P (r , r s )
s
r
27-28/09/2007
UAM
A. D. Becke, K. E. Edgecombe J. Chem. Phys. 1990, 92, 5397
ANALYSIS OF ELECTRON LOCALIZATION/DELOCALIZATION AND AROMATICITY IN MOLECULES
56
The electron localization function (ELF)
Let’s now expand the following function in a Taylor series around s=0:
s 1
P (r , r s ) e P (r , r1 ) |r r1
1 2
e 1 x x ...
2!
x
And then one does the spherical average of the s coordinate:
e
s 1
1
1
s 1
e d
4p
4p
e
27-28/09/2007
UAM
s 1
1
4p
2p p
e
0 0
s 1
1
sin dd
4p
2p p
s1 cos
e
sin dd
0 0
s1
s1
e
e
sinh s1
s1 '
0 d 1e d ' 2s1 s1
2p
1
ANALYSIS OF ELECTRON LOCALIZATION/DELOCALIZATION AND AROMATICITY IN MOLECULES
57
The electron localization function (ELF)
e x e x
x3 x5 x7
sinh x
x ...
2
3! 5! 7!
e
s 1
1 2 2 1 4 4 1 6 6
1 s 1 s 1 s 1 ....
3!
5!
7!
Then inserting this result in the previous expression, we have:
1 2 2
P (r , s) 1 s 1 P (r , r1 ) |r r1
6
1 2 2
1 2 2
P (r , s) P (r , r ) s 1 P (r , r1 ) |r r1 s 1 P (r , r1 ) |r r1
6
6
27-28/09/2007
UAM
ANALYSIS OF ELECTRON LOCALIZATION/DELOCALIZATION AND AROMATICITY IN MOLECULES
58
The electron localization function (ELF)
As we have shown before:
P (r , r1 ) r 1 (r1 ) r XC (r , r1 )
So we are left with:
1 2 2
2
P (r , s) s r (r ) 1 r xc (r , r1 ) |r r1
6
For monodeterminantal WFs:
*
i (r )i (r1 ) j (r ) j (r1 )
r xc (r , r1 )
0
r1 (r )
i 1 j 1
N N
*
2
1 r
2
r xc (r , r1 ) |r r1 r 2
2 r
2
1
27-28/09/2007
UAM
N
i
2
i 1
ANALYSIS OF ELECTRON LOCALIZATION/DELOCALIZATION AND AROMATICITY IN MOLECULES
59
The electron localization function (ELF)
Therefore we finally get:
r
1
1
P (r , s) s 2
3
4 r
2
This function yields the probability of finding an electron of the same spin at a
distance s from the position of the reference electron. In this sense, it gives a
measure of localization. The lower the value of the P(r,s) function, the higher
the localization of the reference electron.
Becke used the alternative function:
2
1
1 r
D
2
8 r
To define the electron localization function (), ELF, as:
1
1 2
D
o
D
3
D
6p 2
10
o
2/3
r5 / 3
0 < < 1, ELF is close to 0 in locations where electrons are delocalized and
becomes close to 1 in well-localized electronic regions
27-28/09/2007
UAM
ANALYSIS OF ELECTRON LOCALIZATION/DELOCALIZATION AND AROMATICITY IN MOLECULES
60
The electron localization function (ELF)
From the ELF basins one can also get:
N A r r dr
2 N A N 2
Charges
A
A
N
2
A
Variance (fluctuation of charge)
Sij A i * (r ) j (r )dr
A
A
27-28/09/2007
UAM
2 A
N A
Relative fluctuation
ANALYSIS OF ELECTRON LOCALIZATION/DELOCALIZATION AND AROMATICITY IN MOLECULES
61
The electron localization function (ELF)
0.85
C(O)
V(OH)
V(O)
N(i)
2.13
1.69
2.25
2
0.33
0.77
0.07
1.04
A change of topological type that occurs at a critical value of the function it is
called a bifurcation, i.e., a value of the ELF at which tow (or more) basins merge,
or separate, from one another.
27-28/09/2007
UAM
ANALYSIS OF ELECTRON LOCALIZATION/DELOCALIZATION AND AROMATICITY IN MOLECULES
62
The electron localization
function (ELF)
27-28/09/2007
UAM
F. Fuster, B. Silvi, Theor. Chem. Acc. 2000, 104, 13
ANALYSIS OF ELECTRON LOCALIZATION/DELOCALIZATION AND AROMATICITY IN MOLECULES
63
The electron localization function (ELF)
B. Silvi, I. Fourré, M. E. Alikhani
Monat. Chem. 2005, 136, 855
27-28/09/2007
UAM
ANALYSIS OF ELECTRON LOCALIZATION/DELOCALIZATION AND AROMATICITY IN MOLECULES
64
The electron localization function (ELF)
Multicenter bonding
Polysynaptic basins
27-28/09/2007
UAM
ANALYSIS OF ELECTRON LOCALIZATION/DELOCALIZATION AND AROMATICITY IN MOLECULES
65
The electron localization function (ELF)
CH3Li
27-28/09/2007
UAM
E. Matito, J. Poater, F. M. Bickelhaupt, M. Solà J. Phys. Chem. A 2006, 110, 7189
ANALYSIS OF ELECTRON LOCALIZATION/DELOCALIZATION AND AROMATICITY IN MOLECULES
66
The electron localization function (ELF)
(CH3Li)4
27-28/09/2007
UAM
ANALYSIS OF ELECTRON LOCALIZATION/DELOCALIZATION AND AROMATICITY IN MOLECULES
67
Programs
Delocalization indices and related aromaticity descriptors
Gaussian 03: http://www.gaussian.com/ → out=wfn
AIMPAC: http://www.chemistry.mcmaster.ca/aimpac/
ESI-3D: http://iqc.udg.edu/~eduard/ESI
ELF calculations
Gaussian 03: http://www.gaussian.com/ → out=wfn
ToPMoD: http://www.lct.jussieu.fr/pagesperso/silvi/topmod_english.html
Vis5D: http://www.ssec.wisc.edu/~billh/vis5d.html
27-28/09/2007
UAM
ANALYSIS OF ELECTRON LOCALIZATION/DELOCALIZATION AND AROMATICITY IN MOLECULES
68