Periodic Table and Chemical Families

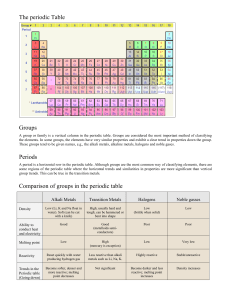
Periodic Table
Periodic Table and
Chemical Families
Science
Learning Presentation
8 th Grade
Success Oriented Lesson Plans
“Organizing” the elements !
We have seen the different elements have different characteristics.
Is there any connection between the element ’s place in the periodic table and its characteristic?
“Organizing” the elements !
Dmitri Mendeleev
1834-1907
Mendeleev (a Russian chemist) organized in
1869 the elements in columns and rows. He integrated
63 elements recognized in his time.
Mendeleev’s Greatness
It is amazing to see that the updated table of
2007
, in which
115 element are written (
23 of them are man made), is not so different from the one Dmitri
Mendeleev invented in
1869
.
It is important to mention that the scientific world at
Mendeleev ’s period knew only 63 elements and scientists knew nothing about the atom ’s structure.
Only genius and daring caused this young scientists to create the connections.
His grate victory was by predicting elements that were not discovered yet!
He left empty spaces with question marks for these elements, and added detailed characteristics he believed these elements will have.
The elements were organized according to two criterions:
1.
According to an atomic number (the number of protons in the element ’s atom).
2.
According to the common characteristics of groups of elements.
Question 1
What is Mendeleev ’s greatness?
A.
He organized the elements in a table according to their atomic number
B.
He organized the elements in a table according to their characteristics
C.
He predicted and left room for elements yet to be discovered
D.
All of the above
Studying the periodic table shows that the elements are organized in columns and rows.
Rows – The elements are organized according to an atomic number (rises from left to right).
Columns – The elements are organized according to common characteristics.
Who was Marie Curie?
Marie Curie was the first woman to win the
Nobel price.
In 1903 she won (along with her husband) the
Nobel price for Physics and in 1911 she won the Nobel price for Chemistry.
(Her daughter, Irène Curie, also won the Nobel price for Physics in 1935).
The connection between Marie
Curie and the Periodic Table:
Marie Curie discovered, along with her husband, the X-rays radiation.
Think – Where will we be without this discovery!
Her second scientific discovery was the discovery of the uranium and the radioactive radiation emitted by the uranium atoms.
Marie Curie died of cancer – a by product of long exposure to radioactive materials.
The elements in the periodic table are also organized in “big” groups:
In addition to the columns and rows we can see that the elements are also organized in bigger chunks – the metals chunk and the nonmetals chunk.
Metals Nonmetals
Question 2
The nonmetals elements are located on the left side of the periodic table.
This sentence is:
A. True
B. False
Let ’s learn about
“Chemical Families”.
“Chemical Family”
A group of like characteristics located at the same column in the periodic table.
Let ’s learn about
“Chemical Families”.
To better understand the concept, we will focus on
3 chemical families:
1.
The Halogen family.
2.
The Alkali metals family.
3.
The Noble gasses family.
What is a “Chemical Family”?
Question 3
A.
A group of elements with the same color.
B.
A group of elements located at the same row in the periodic table.
C.
A group of elements located at the same column in the periodic table.
D.
A group of elements with the same characteristics.
E.
Answers (C) and (D) are correct.
When looking for a replacement for a certain element, it is best to go to the column in which it is located in the periodic table.
The Alkali metals
The Alkali metals are located on the first column on the left (except hydrogen).
The Alkali metals
Common characteristics:
Solid in room temperature, electrical conductivity, soft, low-density, floats in water, react readily with halogens to form ionic salts, and with water to form strongly alkaline (basic) hydroxides.
Examples: sodium, potassium, lithium.
Uses: Mainly batteries.
Question 4
One of the Alkali metals characteristics is their readily reaction with water and nonmetals.
What is the recommended way to store Alkali metal?
A.
With oil
B.
With water
C.
With air
D.
With another metal
The Noble Gasses family
The Nobel Gasses are located on the most right column (number
8
).
The Noble Gasses family
Common characteristics:
These elements do no tend to create compounds, thus they exist in nature as elements. While in room temperature, they are in a state of gas.
Examples: helium, neon, argon.
Uses: lighting, welding, space technology.
Question 5
The hydrogen is a gas lighter than air and is a flammable material.
In airships, the hydrogen was to be replaced with a gas lighter than air, does not readily ignite and does not react to other materials.
Which gas is recommended for usage?
A.
Oxygen, which weigh as air
B.
Nitrogen, which does not react with other materials
C.
The noble gas helium, which meet all the requirements
D.
No gas can replace the hydrogen gas
Question 6
At a factory for manufacturing light bulbs, the argon gas which is used as an inert atmosphere for the inside of filament light bulb was exhausted.
A replacement can be found among the elements belonging to a chemical family called:
A.
The halogen family
B.
The noble gasses family
C.
The alkali metals family
D.
There is no suitable chemical family
Question 7
Why the elements in this “chemical family ” are suitable to fill the light bulbs?
Glass bulb filament
A.
They do not conduct heat
B.
They are colorless
C.
They do not burn in oxygen
D.
They do not conduct electricity
E.
All of the above
Stem
(glass mount)
Cap
(sleeve)
Insulation
Electrical contact
The Halogen Family
The Halogen family is located on the
7 th column from the right.
The Halogen Family
Common characteristics:
Highly reactive and can be harmful or lethal, create salt compounds with other metals, due to their chemical activity they do not appear as elements in nature, toxic.
Examples: fluorine, chlorine, bromine, iodine.
Uses: disinfection, utensils coating, cleaning and bleaching .
Let ’s expound on the 4 halogens.
Owing to their high reactivity, the halogens are found in nature only in compounds.
Fluorine (F) – a poisonous, pale, yellowish brown gas. Used in toothpaste to prevent dental cavities.
Bromine (Br) – a red volatile liquid at standard room temperature. Its vapors are corrosive and toxic.
Used for manufacturing paint, photography materials, medications and fire-resistance cloths.
Additional Halogens:
Chlorine (Cl) – it is a pale green gas and very poisonous. It has a disagreeable, suffocating odor. Was use as a weapon.
Used in swimming pools to keep them clean and sanitary. Known as the cooking salt NaCl.
Iodine (I) – a dark-purple/dark-brown solid. Turns into a violet-pink gas at standard temperatures. Used to disinfect wounds. It is also can be found in small quantities in the human body, and it is important for growth and development.
Question 8
The following are
3 elements characteristics.
Match the appropriate chemical family to the characteristic.
1.
We use it for disinfection in everyday life.
2.
It is used for filling light bulbs.
3.
It is soft and react readily with water to form strongly alkaline (basic) hydroxides.
A.
(1) Alkali metals, (2) Noble gasses, (3) Halogens
B.
(1) Noble gasses, (2) Halogens, (3) Alkali metals
C.
(1) Halogens, (2) Noble gasses, (3) Alkali metals
Concepts Mapping
Elements
Nonmetals
Halogens
Nobel
Gasses
Alkali
Metals
Metals
Other
Metals
Fluorine Helium Potassium Copper
Chlorine Neon Sodium Iron
Question 9
What characteristics we should look for when looking for an element to be used as a filament?
A.
Electricity conductor and low melting temperature
B.
Not an electricity conductor and low melting temperature
C.
Electricity conductor and high melting temperature
D.
Electricity conductor and low melting temperature