CHAPTER 5 The Working Cell
advertisement

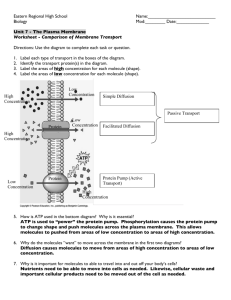
CHAPTER 5 The Working Cell Overview: Energy Def Laws Chemical Reactions ATP Enzymes Def Activity Membrane Structure Function Transport (passive, active, Osmosis, exo- and endocytosis) – What is energy? • Energy is defined as the capacity to do work • All organisms require energy to stay alive • Energy makes change possible • Kinetic energy is the energy of motion • Potential energy is stored energy Two laws govern energy conversion • First law of thermodynamics: Energy can be changed from one form to another – However, energy cannot be created or destroyed • Second law of thermodynamics: Energy changes are not 100% efficient – Energy conversions increase disorder, or entropy – Some energy is always lost as heat • There are two types of chemical reactions: – Endergonic reactions absorb energy and yield products rich in potential energy – Exergonic reactions release energy and yield products that contain less potential energy than their reactants • Cellular respiration – Is the energy-releasing chemical breakdown of fuel molecules – Provides energy for the cell to do work Chemical reactions either store or release energy • Cells carry out thousands of chemical reactions – The sum of these reactions constitutes cellular metabolism ATP shuttles chemical energy within the cell • The chemical energy of organic molecules is released in cellular respiration to make ATP in the mitochondria • In cellular respiration, some energy is stored in ATP molecules • ATP powers nearly all forms of cellular work • ATP molecules are the key to energy coupling The Structure of ATP • ATP (adenosine triphosphate) – Consists of adenosine plus a tail of three phosphate groups – Is broken down to ADP, accompanied by the release of energy • When the bond joining a phosphate group to the rest of an ATP molecule is broken by hydrolysis, the reaction supplies energy for cellular work • ATP can energize other molecules by transferring phosphate groups ENZYMES Enzymes speed up the cell’s chemical reactions by lowering energy barriers • For a chemical reaction to begin, reactants must absorb some energy – This energy is called the energy of activation (EA) – This represents the energy barrier that prevents molecules from breaking down spontaneously • Activation energy – Is the energy that activates the reactants – Triggers a chemical reaction • Enzymes – Lower the activation energy for chemical reactions A specific enzyme catalyzes each cellular reaction • Enzymes are selective – This selectivity determines which chemical reactions occur in a cell • Each enzyme recognizes a specific substrate – The active site fits to the substrate, and the enzyme changes shape slightly – This interaction is called induced fit The cellular environment affects enzyme activity • Enzyme activity is influenced by – temperature – concentration – pH • Some enzymes require nonprotein cofactors – Some cofactors are organic molecules called coenzymes Enzyme Inhibitors • Enzyme inhibitors – Can inhibit a metabolic reaction – Bind to the active site, as substrate impostors • Other inhibitors – Bind at a remote site, changing the enzyme’s shape – In some cases, this is called feedback regulation MEMBRANE STRUCTURE AND FUNCTION Membranes organize the chemical activities of cells • Membranes organize the chemical reactions making up metabolism Cytoplasm • Membranes are selectively permeable – They control the flow of substances into and out of a cell • Membranes can hold teams of enzymes that function in metabolism Membrane phospholipids form a bilayer • Phospholipids are the main structural components of membranes • They each have a hydrophilic head and two hydrophobic tails • In water, phospholipids form a stable bilayer – The heads face outward and the tails face inward The membrane is a fluid mosaic of phospholipids and proteins • Phospholipid molecules form a flexible bilayer – Cholesterol and protein molecules are embedded in it – Carbohydrates act as cell identification tags Proteins make the membrane a mosaic of function • Some membrane proteins form cell junctions • Others transport substances across the membrane • Many membrane proteins are enzymes • Some proteins function as receptors for chemical messages from other cells – The binding of a messenger to a receptor may trigger signal transduction Passive transport is diffusion across a membrane • In passive transport, substances diffuse through membranes without work by the cell – They spread from areas of high concentration to areas of lower concentration • Diffusion is one result of the movement of molecules – Molecules tend to spread into the available space – Diffusion is passive transport; no energy is needed • Another type of passive transport is facilitated diffusion, the transport of some substances by specific transport proteins that act as selective corridors Osmosis is the passive transport of water • In osmosis, water travels from an area of lower solute concentration to an area of higher solute concentration Water balance between cells and their surroundings is crucial to organisms • Osmosis causes cells to shrink in a hypertonic (has a higher concentration of solute) solution and swell in a hypotonic (has a lower concentration of solute) solution The control of water balance (osmoregulation) is essential for organisms Transport proteins facilitate diffusion across membranes • Small nonpolar molecules diffuse freely through the phospholipid bilayer • Many other kinds of molecules pass through selective protein pores by facilitated diffusion Active Transport: the Pumping of Molecules Across Membranes • Active transport requires energy to move molecules across a membrane against a concentration gradient • Active transport in two solutes across a membrane Exocytosis and endocytosis transport large molecules • To move large molecules or particles through a membrane – a vesicle may fuse with the membrane and expel its contents (exocytosis: Secretes substances outside of the cell) – or the membrane may fold inward, trapping material from the outside (endocytosis: takes material into the cell) • Three kinds of endocytosis • Receptor-mediated endocytosis – Is triggered by the binding of external molecules to membrane proteins • In phagocytosis (“cellular eating”) a cell engulfs a particle and packages it within a food vacuole • In pinocytosis (“cellular drinking”) a cell “gulps” droplets of fluid by forming tiny vesicles Pseudopod of amoeba Food being ingested Chloroplasts and mitochondria make energy available for cellular work • Enzymes and membranes are central to the processes that make energy available to the cell • Chloroplasts carry out photosynthesis, using solar energy to produce glucose and oxygen from carbon dioxide and water • Mitochondria consume oxygen in cellular respiration, using the energy stored in glucose to make ATP • Nearly all the chemical energy that organisms use comes ultimately from sunlight • Chemicals recycle among living organisms and their environment